Abstract
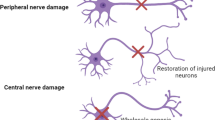
Phantom limb pain (PLP) is a chronic neuropathic pain occurring in 45–85% of patients who undergo major amputations of the upper and lower extremities. Chronic pain is physically and mentally debilitating, affecting an individual’s potential for self-care and the performance of daily living activities essential for personal and economic independence. In addition, chronic pain may lead to depression and feelings of hopelessness. A National Center for Biotechnology Information study found that in the USA alone, the annual cost of dealing with neuropathic pain is more than $600 billion, with an estimated 20 million people in the USA suffering this condition. PLP manifest predominantly during two time frames post-amputation: during days to a month and again at around 1 year. In most patients, the frequency and intensity of the chronic neuropathic pain diminish over time, but severe pain persists in about 5–10% of patients. The development and maintenance of neuropathic pain is attributed to extremity amputations causing changes in peripheral axon properties and neuronal circuitry in both the peripheral and central nervous systems: peripheral axons, dorsal root ganglia, the spinal cord, and the cortex. However, it is not clear how the changes in neuronal properties in these different locations affect neuropathic pain. Is pain initiated by one set of post-amputation changes while the pain is maintained by another set of changes? If one set of amputation-induced changes, such as those of peripheral axons, are reverted to normal, is the chronic pain reduced or eliminated, while reversing another set of neuronal changes and neuronal circuits to normal do not reduce or eliminate the pain? Or, must all the amputation-induced changes be reverted to normal for pain to be eliminated? While this review examines the mechanisms underlying the induction or maintenance of PLP, it is beyond its scope to examine the mechanisms that may permanently reduce or eliminate neuropathic pain. This paper is the first of two reviews in this journal and deals with the causes of chronic PLP development and maintenance, while the second review examines potential mechanisms that may be responsible for promoting the capacity to coping with PLP by reducing or eliminating it.
Similar content being viewed by others
References
Kooijman CM, Dijkstra PU, Geertzen JH et al (2000) Phantom pain and phantom sensations in upper limb amputees: an epidemiological study. Pain 87:33–41
Probstner D, Thuler LC, Ishikawa NM et al (2010) Phantom limb phenomena in cancer amputees. Pain Pract 10:249–256
Schley MT, Wilms P, Toepfner S et al (2008) Painful and nonpainful phantom and stump sensations in acute traumatic amputees. J Trauma 65:858–864
Diers M, Christmann C, Koeppe C et al (2010) Mirrored, imagined and executed movements differentially activate sensorimotor cortex in amputees with and without phantom limb pain. Pain 149:296–304
McCabe CS, Haigh RC, Halligan PW et al (2005) Simulating sensory-motor incongruence in healthy volunteers: implications for a cortical model of pain. Rheumatology (Oxford) 44:509–516
Loeser JD, Treede RD (2008) The Kyoto protocol of IASP basic pain terminology. Pain 137:473–477
Ziegler-Graham K, MacKenzie EJ, Ephraim PL et al (2008) Estimating the prevalence of limb loss in the United States: 2005 to 2050. Arch Phys Med Rehabil 89:422–429
Reiber GE, McFarland LV, Hubbard S et al (2010) Servicemembers and veterans with major traumatic limb loss from Vietnam war and OIF/OEF conflicts: survey methods, participants, and summary findings. J Rehabil Res Dev 47:275–297
Richardson C, Glenn S, Nurmikko T et al (2006) Incidence of phantom phenomena including phantom limb pain 6 months after major lower limb amputation in patients with peripheral vascular disease. Clin J Pain 22:353–358
Aldington D, Small C, Edwards D et al (2014) A survey of post-amputation pains in serving military personnel. J R Army Med Corps 160:38–41
Le Feuvre P, Aldington D (2014) Know pain know gain: proposing a treatment approach for phantom limb pain. J R Army Med Corps 160:16–21
Tilak M, Isaac SA, Fletcher J et al (2015) Mirror therapy and transcutaneous electrical nerve stimulation for Management of Phantom Limb Pain in amputees—a single blinded randomized controlled trial. Physiother Res Int
Davidson JH, Khor KE, Jones LE (2010) A cross-sectional study of post-amputation pain in upper and lower limb amputees, experience of a tertiary referral amputee clinic. Disabil Rehabil 32:1855–1862
Hirsh AT, Dillworth TM, Ehde DM et al (2010) Sex differences in pain and psychological functioning in persons with limb loss. J Pain 11:79–86
Richardson C, Crawford K, Milnes K et al (2015) A clinical evaluation of postamputation phenomena including phantom limb pain after lower limb amputation in Dysvascular patients. Pain Manag Nurs 16:561–569
Hanley MA, Jensen MP, Smith DG et al (2007) Preamputation pain and acute pain predict chronic pain after lower extremity amputation. J Pain 8:102–109
Zeng Y, Wang X, Guo Y et al (2016) Coblation of femoral and sciatic nerve for stump pain and phantom limb pain: a case report. Pain Pract 16:E35–E41
Eldabe S, Burger K, Moser H et al (2015) Dorsal root ganglion (DRG) stimulation in the treatment of phantom limb pain (PLP). Neuromodulation 18:610–616 discussion 616-617
Azhary H, Farooq MU, Bhanushali M et al (2010) Peripheral neuropathy: differential diagnosis and management. Am Fam Physician 81:887–892
Knotkova H, Cruciani RA, Tronnier VM et al (2012) Current and future options for the management of phantom-limb pain. J Pain Res 5:39–49
De Graaf JB, Jarrasse N, Nicol C et al (2016) Phantom hand and wrist movements in upper limb amputees are slow but naturally controlled movements. Neuroscience 312:48–57
Santello M, Nevian T (2015) Dysfunction of cortical dendritic integration in neuropathic pain reversed by serotoninergic neuromodulation. Neuron 86:233–246
Raffin E, Richard N, Giraux P et al (2016) Primary motor cortex changes after amputation correlate with phantom limb pain and the ability to move the phantom limb. NeuroImage 130:134–144
Jutzeler CR, Curt A, Kramer JL (2015) Relationship between chronic pain and brain reorganization after deafferentation: a systematic review of functional MRI findings. Neuroimage Clin 9:599–606
Sugimine S, Ogino Y, Kawamichi H et al (2016) Brain morphological alternation in chronic pain patients with neuropathic characteristics. Mol Pain 12:1–7
MacIver K, Lloyd DM, Kelly S et al (2008) Phantom limb pain, cortical reorganization and the therapeutic effect of mental imagery. Brain 131:2181–2191
Roux FE, Ibarrola D, Lazorthes Y et al (2008) Chronic motor cortex stimulation for phantom limb pain: a functional magnetic resonance imaging study: technical case report. Neurosurgery 62:978–985
Weeks SR, Anderson-Barnes VC, Tsao JW (2010) Phantom limb pain: theories and therapies. Neurologist 16:277–286
Baron R, Binder A, Wasner G (2010) Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol 9:807–819
Borghi B, D'Addabbo M, White PF et al (2010) The use of prolonged peripheral neural blockade after lower extremity amputation: the effect on symptoms associated with phantom limb syndrome. Anesth Analg 111:1308–1315
Ramachandran VS, Brang D, McGeoch PD (2010) Dynamic reorganization of referred sensations by movements of phantom limbs. Neuroreport 21:727–730
Vartiainen N, Kirveskari E, Kallio-Laine K et al (2009) Cortical reorganization in primary somatosensory cortex in patients with unilateral chronic pain. J Pain 10:854–859
Giummarra MJ, Gibson SJ, Georgiou-Karistianis N et al (2008) Mechanisms underlying embodiment, disembodiment and loss of embodiment. Neurosci Biobehav Rev 32:143–160
Oelschlager M, Pfannmoller J, Langner I et al (2014) Usage of the middle finger shapes reorganization of the primary somatosensory cortex in patients with index finger amputation. Restor Neurol Neurosci 32:507–515
McGeoch PD, Brang D, Huang M et al (2015) Primary somatosensory cortex hand representation dynamically modulated by motor output. Neurocase 21:103–105
Tyler DJ (2015) Neural interfaces for somatosensory feedback: bringing life to a prosthesis. Curr Opin Neurol 28:574–581
Baron R (2006) Mechanisms of disease: neuropathic pain—a clinical perspective. Nat Clin Pract Neurol 2:95–106
Bee LA, Dickenson AH (2008) Descending facilitation from the brainstem determines behavioural and neuronal hypersensitivity following nerve injury and efficacy of pregabalin. Pain 140:209–223
Costigan M, Scholz J, Woolf CJ (2009) Neuropathic pain: a maladaptive response of the nervous system to damage. Annu Rev Neurosci 32:1–32
Arbat-Plana A, Torres-Espin A, Navarro X et al (2015) Activity dependent therapies modulate the spinal changes that motoneurons suffer after a peripheral nerve injury. Exp Neurol 263:293–305
Berg A, Zelano J, Thams S et al (2013) The extent of synaptic stripping of motoneurons after axotomy is not correlated to activation of surrounding glia or downregulation of postsynaptic adhesion molecules. PLoS One 8:e59647
Walters ET (2012) Nociceptors as chronic drivers of pain and hyperreflexia after spinal cord injury: An adaptive-maladaptive hyperfunctional state hypothesis. Front Physiol 3:309
Bai L, Wang X, Li Z et al (2016) Upregulation of chemokine CXCL12 in the dorsal root ganglia and spinal cord contributes to the development and maintenance of neuropathic pain following spared nerve injury in rats. Neurosci Bull 32:27–40
Bedi SS, Yang Q, Crook RJ et al (2010) Chronic spontaneous activity generated in the somata of primary nociceptors is associated with pain-related behavior after spinal cord injury. J Neurosci 30:14870–14882
Yang Q, Wu Z, Hadden JK et al (2014) Persistent pain after spinal cord injury is maintained by primary afferent activity. J Neurosci 34:10765–10769
Dickinson BD, Head CA, Gitlow S et al (2010) Maldynia: pathophysiology and management of neuropathic and maladaptive pain—a report of the AMA Council on science and public health. Pain Med 11:1635–1653
Laedermann CJ, Cachemaille M, Kirschmann G et al (2013) Dysregulation of voltage-gated sodium channels by ubiquitin ligase NEDD4-2 in neuropathic pain. J Clin Invest 123:3002–3013
Nikolajsen L, Black JA, Kroner K et al (2010) Neuroma removal for neuropathic pain: efficacy and predictive value of lidocaine infusion. Clin J Pain 26:788–793
Thakor DK, Lin A, Matsuka Y et al (2009) Increased peripheral nerve excitability and local NaV1.8 mRNA up-regulation in painful neuropathy. Mol Pain 5:14
Waxman SG, Cummins TR, Dib-Hajj SD et al (2000) Voltage-gated sodium channels and the molecular pathogenesis of pain: a review. J Rehabil Res Dev 37:517–528
Zimmermann M (2001) Pathobiology of neuropathic pain. Eur J Pharmacol 429:23–37
Karanikolas M, Aretha D, Tsolakis I et al (2011) Optimized perioperative analgesia reduces chronic phantom limb pain intensity, prevalence, and frequency: a prospective, randomized, clinical trial. Anesthesiology 114:1144–1154
Flor H, Nikolajsen L, Staehelin Jensen T (2006) Phantom limb pain: a case of maladaptive CNS plasticity? Nat Rev Neurosci 7:873–881
Jarvis MF, Boyce-Rustay JM (2009) Neuropathic pain: models and mechanisms. Curr Pharm Des 15:1711–1716
Sumitani M, Miyauchi S, Uematsu H et al (2010) Phantom limb pain originates from dysfunction of the primary motor cortex. Masui Jap J Anesthesiol 59:1364–1369
Chen A, Kumar SM, Sahley CL et al (2000) Nitric oxide influences injury-induced microglial migration and accumulation in the leech CNS. J Neurosci 20:1036–1043
Kim KH, Kim JI, Han JA et al (2011) Upregulation of neuronal nitric oxide synthase in the periphery promotes pain hypersensitivity after peripheral nerve injury. Neuroscience 190:367–378
Katano T, Furue H, Okuda-Ashitaka E et al (2008) N-ethylmaleimide-sensitive fusion protein (NSF) is involved in central sensitization in the spinal cord through GluR2 subunit composition switch after inflammation. Eur J Neurosci 27:3161–3170
Zeilhofer HU (2008) Loss of glycinergic and GABAergic inhibition in chronic pain--contributions of inflammation and microglia. Int Immunopharmacol 8:182–187
Wu Z, Yang Q, Crook RJ et al (2013) TRPV1 channels make major contributions to behavioral hypersensitivity and spontaneous activity in nociceptors after spinal cord injury. Pain 154:2130–2141
Kern KU, Baust H, Hofmann W et al (2014) Capsaicin 8% cutaneous patches for phantom limb pain. Results from everyday practice (non-interventional study). Schmerz 28:374–383
Stemkowski PL, Noh MC, Chen Y et al (2015) Increased excitability of medium-sized dorsal root ganglion neurons by prolonged interleukin-1beta exposure is K(+) channel dependent and reversible. J Physiol 593:3739–3755
Bourinet E, Francois A, Laffray S (2016) T-type calcium channels in neuropathic pain. Pain 157(Suppl 1):S15–S22
Yin R, Liu D, Chhoa M et al (2016) Voltage-gated sodium channel function and expression in injured and uninjured rat dorsal root ganglia neurons. Int J Neurosci 126:182–192
Xie W, Strong JA, Zhang JM (2015) Local knockdown of the NaV1.6 sodium channel reduces pain behaviors, sensory neuron excitability, and sympathetic sprouting in rat models of neuropathic pain. Neuroscience 291:317–330
Zheng JH, Walters ET, Song XJ (2007) Dissociation of dorsal root ganglion neurons induces hyperexcitability that is maintained by increased responsiveness to cAMP and cGMP. J Neurophysiol 97:15–25
Luo J, Walters ET, Carlton SM et al (2013) Targeting pain-evoking transient receptor potential channels for the treatment of pain. Curr Neuropharmacol 11:652–663
Zhou YQ, Liu Z, Liu ZH et al (2016) Interleukin-6: an emerging regulator of pathological pain. J Neuroinflammation 13:141
Song XJ, Wang ZB, Gan Q et al (2006) cAMP and cGMP contribute to sensory neuron hyperexcitability and hyperalgesia in rats with dorsal root ganglia compression. J Neurophysiol 95:479–492
Walters ET (2014) Neuroinflammatory contributions to pain after SCI: roles for central glial mechanisms and nociceptor-mediated host defense. Exp Neurol 258:48–61
Bavencoffe A, Li Y, Wu Z et al (2016) Persistent electrical activity in primary nociceptors after spinal cord injury is maintained by Scaffolded adenylyl cyclase and protein kinase A and is associated with altered adenylyl cyclase regulation. J Neurosci 36:1660–1668
Yamanaka H, Kobayashi K, Okubo M et al (2016) Annexin A2 in primary afferents contributes to neuropathic pain associated with tissue type plasminogen activator. Neuroscience 314:189–199
Ogon I, Takebayashi T, Miyakawa T et al (2016) Attenuation of pain behaviour by local administration of alpha-2 adrenoceptor antagonists to dorsal root ganglia in a rat radiculopathy model. Eur J Pain 20:790–799
Wu S, Marie Lutz B, Miao X et al (2016) Dorsal root ganglion transcriptome analysis following peripheral nerve injury in mice. Mol Pain 12:1–14
Zhang Y, Laumet G, Chen SR et al (2015) Pannexin-1 up-regulation in the dorsal root ganglion contributes to neuropathic pain development. J Biol Chem 290:14647–14655
Zhang M, Han W, Zheng J et al (2015) Inhibition of hyperpolarization-activated cation current in medium-sized DRG neurons contributed to the antiallodynic effect of methylcobalamin in the rat of a chronic compression of the DRG. Neural Plast 2015:197392
Wang D, Xue Y, Yan Y et al (2016) Reversal of neurochemical alterations in the spinal dorsal horn and dorsal root ganglia by Mas-related gene (Mrg) receptors in a rat model of spinal nerve injury. Neurobiol Dis 91:274–283
Wang D, Xue Y, Chen Y et al (2016) Mas-related gene (Mrg) C receptors inhibit mechanical allodynia and spinal microglia activation in the early phase of neuropathic pain in rats. Neurosci Lett 618:115–121
Acknowledgements
No support was involved in the production of this paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The author declares that he has no conflicts of interest.
Rights and permissions
About this article
Cite this article
Kuffler, D.P. Origins of Phantom Limb Pain. Mol Neurobiol 55, 60–69 (2018). https://doi.org/10.1007/s12035-017-0717-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-017-0717-x