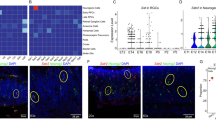
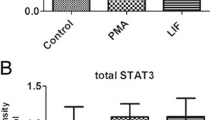
Abstract
We showed previously that the neuropeptide pituitary adenylyl cyclase-activating polypeptide (PACAP) negatively regulates proliferation of postnatal rat retinal progenitor cells through the downregulation of cyclin D1 in a cAMP/protein kinase A dependent manner. In the present study, we describe by microarray analysis several putative PACAP targets regulated by different transcription factor families. One of these families is the Sp/Klf family of transcriptional factors capable of regulating cyclin D1, and among members, we demonstrate by immunocytochemistry that KLF4 is expressed throughout rat retinal development by retinal progenitor cells and in most differentiated cell types. Using retinal explants preparations, PACAP treatment can transiently increase Klf4 mRNA levels; from electrophoretic mobility shift assays, PACAP is also able to increase the nuclear KLF4 content. From these results, we suggest that KLF4 may be involved in the anti-proliferative effects of PACAP as one mechanism regulating progenitor cell transition from proliferation to differentiation throughout retinal development.







Similar content being viewed by others
Abbreviations
- KLF:
-
Kruppel-like factor
- BrdU:
-
5-Bromo-2-deoxyuridine
- CREB:
-
cAMP response element-binding protein
- RPCs:
-
Retinal progenitor cells
- DPM:
-
Disintegrations per minute
- GCL:
-
Ganglion cell layer
- HRP:
-
Horseradish peroxidase
- INL:
-
Inner cell layer
- PAC1:
-
Pituitary adenylyl cyclase activating polypeptide-specific receptor
- PACAP:
-
Pituitary adenylyl cyclase-activating polypeptide
- PCNA:
-
Proliferating cell nuclear antigen
- pCREB:
-
Phosphorylated cAMP response element-binding protein
- PKA:
-
cAMP-dependent protein kinase (protein kinase A)
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- Shh:
-
Sonic hedgehog
- VIP:
-
Vasoactive intestinal peptide
- VPAC:
-
Receptor activated by both pituitary adenylyl cyclase-activating polypeptide and vasoactive intestinal peptide
References
Anchan RM, Reh TA (1995) Transforming growth factor-beta-3 is mitogenic for rat retinal progenitor cells in vitro. J Neurobiol 28(2):133–145
Anchan RM, Reh TA, Angello J, Balliet A, Walker M (1991) EGF and TGF-alpha stimulate retinal neuroepithelial cell proliferation in vitro. Neuron 6(6):923–936
Atlasz T, Szabadfi K, Kiss P, Marton Z, Griecs M, Hamza L, Gaal V, Biro Z, Tamas A, Hild G, Nyitrai M, Toth G, Reglodi D, Gabriel R (2011) Effects of PACAP in UV-A radiation-induced retinal degeneration models in rats. J Mol Neurosci 43(1):51–57
Atlasz T, Szabadfi K, Reglodi D, Kiss P, Tamas A, Toth G, Molnar A, Szabo K, Gabriel R (2009) Effects of pituitary adenylate cyclase activating polypeptide and its fragments on retinal degeneration induced by neonatal monosodium glutamate treatment. Ann N Y Acad Sci 1163:348–352
Baxter PS, Martel MA, McMahon A, Kind PC, Hardingham GE (2011) Pituitary adenylate cyclase-activating peptide induces long-lasting neuroprotection through the induction of activity-dependent signaling via the cyclic AMP response element-binding protein-regulated transcription co-activator 1. J Neurochem 118(3):365–378
Borba JC, Henze IP, Silveira MS, Kubrusly RC, Gardino PF, de Mello MC, Hokoc JN, de Mello FG (2005) Pituitary adenylate cyclase-activating polypeptide (PACAP) can act as determinant of the tyrosine hydroxylase phenotype of dopaminergic cells during retina development. Brain Res Dev Brain Res 156(2):193–201
Braas KM, Schutz KC, Bond JP, Vizzard MA, Girard BM, May V (2007) Microarray analyses of pituitary adenylate cyclase activating polypeptide (PACAP)-regulated gene targets in sympathetic neurons. Peptides 28(9):1856–1870
Carey RG, Li B, DiCicco-Bloom E (2002) Pituitary adenylate cyclase activating polypeptide anti-mitogenic signaling in cerebral cortical progenitors is regulated by p57Kip2-dependent CDK2 activity. J Neurosci 22(5):1583–1591
Cepko CL (1999) The roles of intrinsic and extrinsic cues and bHLH genes in the determination of retinal cell fates. Curr Opin Neurobiol 9(1):37–46
Chen X, Johns DC, Geiman DE, Marban E, Dang DT, Hamlin G, Sun R, Yang VW (2001) Kruppel-like factor 4 (gut-enriched Kruppel-like factor) inhibits cell proliferation by blocking G1/S progression of the cell cycle. J Biol Chem 276(32):30423–30428
Chen X, Whitney EM, Gao SY, Yang VW (2003) Transcriptional profiling of Kruppel-like factor 4 reveals a function in cell cycle regulation and epithelial differentiation. J Mol Biol 326(3):665–677
Close JL, Gumuscu B, Reh TA (2005) Retinal neurons regulate proliferation of postnatal progenitors and Muller glia in the rat retina via TGF beta signaling. Development 132(13):3015–3026
Dai X, Segre JA (2004) Transcriptional control of epidermal specification and differentiation. Curr Opin Genet Dev 14(5):485–491
Ehlermann J, Pfisterer P, Schorle H (2003) Dynamic expression of Kruppel-like factor 4 (Klf4), a target of transcription factor AP-2alpha during murine mid-embryogenesis. Anat Rec A: Discov Mol Cell Evol Biol 273(2):677–680
Feng XH, Zhang Y, Wu RY, Derynck R (1998) The tumor suppressor Smad4/DPC4 and transcriptional adaptor CBP/p300 are coactivators for smad3 in TGF-beta-induced transcriptional activation. Genes Dev 12(14):2153–2163
Ghaleb AM, Nandan MO, Chanchevalap S, Dalton WB, Hisamuddin IM, Yang VW (2005) Kruppel-like factors 4 and 5: the yin and yang regulators of cellular proliferation. Cell Res 15(2):92–96
Grueneberg DA, Henry RW, Brauer A, Novina CD, Cheriyath V, Roy AL, Gilman M (1997) A multifunctional DNA-binding protein that promotes the formation of serum response factor/homeodomain complexes: identity to TFII-I. Genes Dev 11(19):2482–2493
Grumolato L, Louiset E, Alexandre D, Ait-Ali D, Turquier V, Fournier A, Fasolo A, Vaudry H, Anouar Y (2003) PACAP and NGF regulate common and distinct traits of the sympathoadrenal lineage: effects on electrical properties, gene markers and transcription factors in differentiating PC12 cells. Eur J Neurosci 17(1):71–82
Hatakeyama J, Kageyama R (2004) Retinal cell fate determination and bHLH factors. Semin Cell Dev Biol 15(1):83–89
Hellemans J, Mortier G, De Paepe A, Speleman F, Vandesompele J (2007) qBase relative quantification framework and software for management and automated analysis of real-time quantitative PCR data. Genome Biol 8(2):R19
Hu W, Hofstetter WL, Li H, Zhou Y, He Y, Pataer A, Wang L, Xie K, Swisher SG, Fang B (2009) Putative tumor-suppressive function of Kruppel-like factor 4 in primary lung carcinoma. Clin Cancer Res 15(18):5688–5695
Hurgin V, Novick D, Rubinstein M (2002) The promoter of IL-18 binding protein: activation by an IFN-gamma-induced complex of IFN regulatory factor 1 and CCAAT/enhancer binding protein beta. Proc Natl Acad Sci U S A 99(26):16957–16962
Husmann M, Dragneva Y, Romahn E, Jehnichen P (2000) Nuclear receptors modulate the interaction of Sp1 and GC-rich DNA via ternary complex formation. Biochem J 352(Pt 3):763–772
Jensen AM, Wallace VA (1997) Expression of Sonic hedgehog and its putative role as a precursor cell mitogen in the developing mouse retina. Development 124(2):363–371
Kanekar S, Gandham M, Lucero MT (2010) PACAP protects against TNFalpha-induced cell death in olfactory epithelium and olfactory placodal cell lines. Mol Cell Neurosci 45(4):345–354
Katz JP, Perreault N, Goldstein BG, Actman L, McNally SR, Silberg DG, Furth EE, Kaestner KH (2005) Loss of Klf4 in mice causes altered proliferation and differentiation and precancerous changes in the adult stomach. Gastroenterology 128(4):935–945
Lang B, Song B, Davidson W, MacKenzie A, Smith N, McCaig CD, Harmar AJ, Shen S (2006) Expression of the human PAC1 receptor leads to dose-dependent hydrocephalus-related abnormalities in mice. J Clin Invest 116(7):1924–1934
Lelievre V, Seksenyan A, Nobuta H, Yong WH, Chhith S, Niewiadomski P, Cohen JR, Dong H, Flores A, Liau LM, Kornblum HI, Scott MP, Waschek JA (2008) Disruption of the PACAP gene promotes medulloblastoma in ptc1 mutant mice. Dev Biol 313(1):359–370
Leone TC, Cresci S, Carter ME, Zhang Z, Lala DS, Strauss AW, Kelly DP (1995) The human medium chain acyl-CoA dehydrogenase gene promoter consists of a complex arrangement of nuclear receptor response elements and Sp1 binding sites. J Biol Chem 270(41):24622
Lillien L, Cepko C (1992) Control of proliferation in the retina: temporal changes in responsiveness to FGF and TGF alpha. Development 115(1):253–266
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Lu N, Zhou R, DiCicco-Bloom E (1998) Opposing mitogenic regulation by PACAP in sympathetic and cerebral cortical precursors correlates with differential expression of PACAP receptor (PAC1-R) isoforms. J Neurosci Res 53(6):651–662
Marquardt T, Gruss P (2002) Generating neuronal diversity in the retina: one for nearly all. Trends Neurosci 25(1):32–38
Martins RA, Linden R, Dyer MA (2006) Glutamate regulates retinal progenitors cells proliferation during development. Eur J Neurosci 24(4):969–980
Martins RA, Pearson RA (2008) Control of cell proliferation by neurotransmitters in the developing vertebrate retina. Brain Res 1192:37–60
Miyata A, Arimura A, Dahl RR, Minamino N, Uehara A, Jiang L, Culler MD, Coy DH (1989) Isolation of a novel 38 residue-hypothalamic polypeptide which stimulates adenylate cyclase in pituitary cells. Biochem Biophys Res Commun 164(1):567–574
Miyata A, Jiang L, Dahl RD, Kitada C, Kubo K, Fujino M, Minamino N, Arimura A (1990) Isolation of a neuropeptide corresponding to the N-terminal 27 residues of the pituitary adenylate cyclase activating polypeptide with 38 residues (PACAP38). Biochem Biophys Res Commun 170(2):643–648
Moore DL, Blackmore MG, Hu Y, Kaestner KH, Bixby JL, Lemmon VP, Goldberg JL (2009) KLF family members regulate intrinsic axon regeneration ability. Science 326(5950):298–301
Moore DL, Goldberg JL (2011) Multiple transcription factor families regulate axon growth and regeneration. Dev Neurobiol 71(12):1186–1211
Nicot A, Lelievre V, Tam J, Waschek JA, DiCicco-Bloom E (2002) Pituitary adenylate cyclase-activating polypeptide and sonic hedgehog interact to control cerebellar granule precursor cell proliferation. J Neurosci 22(21):9244–9254
Njaine B, Martins RA, Santiago MF, Linden R, Silveira MS (2010) Pituitary adenylyl cyclase-activating polypeptide controls the proliferation of retinal progenitor cells through downregulation of cyclin D1. Eur J Neurosci 32(3):311–321
Qin S, Liu M, Niu W, Zhang CL (2011) Dysregulation of Kruppel-like factor 4 during brain development leads to hydrocephalus in mice. Proc Natl Acad Sci U S A 108(52):21117–21121
Qin S, Zhang CL (2012) Role of Kruppel-like factor 4 in neurogenesis and radial neuronal migration in the developing cerebral cortex. Mol Cell Biol 32(21):4297–4305
Reglodi D, Kiss P, Szabadfi K, Atlasz T, Gabriel R, Horvath G, Szakaly P, Sandor B, Lubics A, Laszlo E, Farkas J, Matkovits A, Brubel R, Hashimoto H, Ferencz A, Vincze A, Helyes Z, Welke L, Lakatos A, Tamas A (2012) PACAP is an endogenous protective factor-insights from PACAP-deficient mice. J Mol Neurosci 48(3):482–492
Rehen SK, Varella MH, Freitas FG, Moraes MO, Linden R (1996) Contrasting effects of protein synthesis inhibition and of cyclic AMP on apoptosis in the developing retina. Development 122(5):1439–1448
Rocha-Martins M, Njaine B, Silveira MS (2012) Avoiding pitfalls of internal controls: validation of reference genes for analysis by qRT-PCR and western blot throughout rat retinal development. PLoS One 7(8):e43028
Rowland BD, Bernards R, Peeper DS (2005) The KLF4 tumour suppressor is a transcriptional repressor of p53 that acts as a context-dependent oncogene. Nat Cell Biol 7(11):1074–1082
Shimizu Y, Takeuchi T, Mita S, Notsu T, Mizuguchi K, Kyo S (2010) Kruppel-like factor 4 mediates anti-proliferative effects of progesterone with G(0)/G(1) arrest in human endometrial epithelial cells. J Endocrinol Invest 33(10):745–750
Silveira MS, Costa MR, Bozza M, Linden R (2002) Pituitary adenylyl cyclase-activating polypeptide prevents induced cell death in retinal tissue through activation of cyclic AMP-dependent protein kinase. J Biol Chem 277(18):16075–16080
Suh J, Lu N, Nicot A, Tatsuno I, DiCicco-Bloom E (2001) PACAP is an anti-mitogenic signal in developing cerebral cortex. Nat Neurosci 4(2):123–124
Szabadfi K, Szabo A, Kiss P, Reglodi D, Setalo G Jr, Kovacs K, Tamas A, Toth G, Gabriel R (2014) PACAP promotes neuron survival in early experimental diabetic retinopathy. Neurochem Int 64:84–91
Sze KL, Lee WM, Lui WY (2008) Expression of CLMP, a novel tight junction protein, is mediated via the interaction of GATA with the Kruppel family proteins, KLF4 and Sp1, in mouse TM4 Sertoli cells. J Cell Physiol 214(2):334–344
Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S (2007) Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131(5):861–872
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126(4):663–676
Varella MH, Correa DF, Campos CB, Chiarini LB, Linden R (1997) Protein kinases selectively modulate apoptosis in the developing retina in vitro. Neurochem Int 31(2):217–227
Varella MH, de Mello FG, Linden R (1999) Evidence for an antiapoptotic role of dopamine in developing retinal tissue. J Neurochem 73(2):485–492
Vaudry D, Falluel-Morel A, Bourgault S, Basille M, Burel D, Wurtz O, Fournier A, Chow BK, Hashimoto H, Galas L, Vaudry H (2009) Pituitary adenylate cyclase-activating polypeptide and its receptors: 20 years after the discovery. Pharmacol Rev 61(3):283–357
Wada Y, Nakamachi T, Endo K, Seki T, Ohtaki H, Tsuchikawa D, Hori M, Tsuchida M, Yoshikawa A, Matkovits A, Kagami N, Imai N, Fujisaka S, Usui I, Tobe K, Koide R, Takahashi H, Shioda S (2013) PACAP attenuates NMDA-induced retinal damage in association with modulation of the microglia/macrophage status into an acquired deactivation subtype. J Mol Neurosci 51(2):493–502
Wei D, Kanai M, Jia Z, Le X, Xie K (2008) Kruppel-like factor 4 induces p27Kip1 expression in and suppresses the growth and metastasis of human pancreatic cancer cells. Cancer Res 68(12):4631–4639
Yoon HS, Chen X, Yang VW (2003) Kruppel-like factor 4 mediates p53-dependent G1/S cell cycle arrest in response to DNA damage. J Biol Chem 278(4):2101–2105
Young TL, Cepko CL (2004) A role for ligand-gated ion channels in rod photoreceptor development. Neuron 41(6):867–879
Zhang X, Odom DT, Koo SH, Conkright MD, Canettieri G, Best J, Chen H, Jenner R, Herbolsheimer E, Jacobsen E, Kadam S, Ecker JR, Emerson B, Hogenesch JB, Unterman T, Young RA, Montminy M (2005) Genome-wide analysis of cAMP-response element binding protein occupancy, phosphorylation, and target gene activation in human tissues. Proc Natl Acad Sci U S A 102(12):4459–4464
Acknowledgments
We thank José Nilson dos Santos, José Francisco Tibúrcio, Damaris Sincorá, Gildo Brito de Souza, and Talita Martins for technical assistance and the Bioinformatics Core Facility at University of Vermont, USA. Support for the University of Vermont Neuroscience Core Facility from NIH NCRR P30RR032135/NIGMS P30GM103498 is also gratefully acknowledged. This investigation was supported by grants from CAEN/International Society for Neurochemistry, CNPq, and FAPERJ.
Conflict of Interest
There is no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Additional information
Brian Njaine and Maurício Rocha-Martins contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(GIF 32 kb)
Rights and permissions
About this article
Cite this article
Njaine, B., Rocha-Martins, M., Vieira-Vieira, C.H. et al. Pleiotropic Functions of Pituitary Adenylyl Cyclase-Activating Polypeptide on Retinal Ontogenesis: Involvement of KLF4 in the Control of Progenitor Cell Proliferation. J Mol Neurosci 54, 430–442 (2014). https://doi.org/10.1007/s12031-014-0299-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-014-0299-2