Abstract
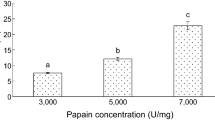
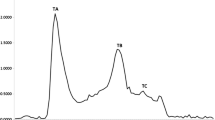
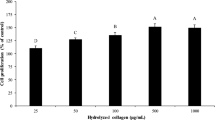
In this study, rainbow trout skin (Oncorhynchus mykiss) was hydrolyzed using Alcalase (HA) and Flavourzyme (HF) enzymes and bioactive peptides separated by membrane ultrafiltration method (< 3, 3–30 and > 30 kDa). The antioxidant properties of the protein hydrolyzed (non-fractionated) and anti-cancer (peptide fractions) were evaluated. The results showed that DPPH radical inhibitory power and ferric reducing antioxidant power in hydrolyzed skin protein by Flavourzyme were significantly higher than Alcalase hydrolyzed protein (p < 0.05). By increasing protein concentrations, the antioxidant property of the hydrolyzed protein increased. To evaluate anti-cancer activity, MTT assay and HCT-116 cell line were used. Hydrolyzed skin protein with molecular weight less than 3 kDa had the highest inhibitory concentration (IC50). The results proved rainbow trout skin protein hydrolysate showed antioxidant properties and could be used as an antioxidant in food. The isolated fractions on HCT-116 cancer cells have cytotoxic properties and inhibit the growth of these cells in vitro. In order to achieve more accurate results, evaluation of properties is recommended in in vivo conditions.





Similar content being viewed by others
References
AOAC (2002) Official methods of analysis of the association of official analytical chemists, 14th edn. AOAC, Washington, DC
Chalamaiah M, Yu W, Wu J (2018) Immunomodulatory and anticancer protein hydrolysates (peptides) from food proteins: a review. Food Chem 245:205–222. https://doi.org/10.1016/j.foodchem.2017.10.087
Choksawangkarn W, Phiphattananukoon S, Jaresitthikunchai J, Roytrakul S (2018) Antioxidative peptides from fish sauce by-product: isolation and characterization. Agric Nat Resour 52(5):460–466. https://doi.org/10.1016/j.anres.2018.11.001
Ehrenstein G, Lecar H (1977) Electrically gated ionic channels in lipid bilayers. Q Rev Biophys 10(1):1–34
Guerard F, Guimas L, Binet A (2002) Production of tuna waste hydrolysates by a commercial neutral protease preparation. J Mol Catal B 19:489–498. https://doi.org/10.1016/S1381-1177(02)00203-5
Halim NRA, Yusof HM, Sarbon NM (2016) Functional and bioactive properties of fish protein hydolysates and peptides: a comprehensive review. Trends Food Sci Technol 51:24–33. https://doi.org/10.1016/j.tifs.2016.02.007
Halim NRA, Azlan A, Yusof HM, Sarbon NM (2018) Antioxidant and anticancer activities of enzymatic eel (Monopterus sp.) protein hydrolysate as influenced by different molecular weight. Biocatal Agric Biotechnol 16:10–16. https://doi.org/10.1016/j.bcab.2018.06.006
Hamzeh A, Rezaei M, Khodabandeh S, Motamedzadegan A, Noruzinia M (2017) Antiproliferative an d antioxidative activities of cuttlefish (Sepia pharaonis) protein hydrolysates as affected by degree of hydrolysis. J Food Meas Charact 12(2):721–727. https://doi.org/10.1007/s11694-017-9685-0
Hubenak JR, Zhang Q, Branch CD, Kronowitz SJ (2014) Mechanisms of injury to normal tissue after radiotherapy: a review. Plast Reconstr Surg 133(1):49e. https://doi.org/10.1097/01.prs.0000440818.23647.0b
Ishak NH, Sarbon NM (2018) A review of protein hydrolysates and bioactive peptides deriving from wastes generated by fish processing. Food Bioprocess Technol 11(1):2–16
Jemil I et al (2014) Functional, antioxidant and antibacterial properties of protein hydrolysates prepared from fish meat fermented by Bacillus subtilis A26. Process Biochem 49(6):963–972. https://doi.org/10.1016/j.procbio.2014.03.004
Karadag A, Ozcelik B, Saner S (2009) Review of methods to determine antioxidant capacities. Food Anal Methods 2(1):41–60. https://doi.org/10.1007/s12161-008-9067-7
Ketnawa S, Liceaga AM (2017) Effect of microwave treatments on antioxidant activity and antigenicity of fish frame protein hydrolysates. Food Bioprocess Technol 10(3):582–591. https://doi.org/10.1007/s11947-016-1841-8
Kim SM (2011) Antioxidant and anticancer activities of enzymatic hydrolysates of solitary tunicate (Styela clava). Food Sci Biotechnol 20(4):1075–1085. https://doi.org/10.1007/s10068-011-0146-y
Klompong V, Benjakul S, Kantachote D, Hayes KD, Shahidi F (2008) Comparative study on antioxidative activity of yellow stripe trevally protein hydrolysate produced from alcalase and flavourzyme. Int J Food Sci Technol 43(6):1019–1026. https://doi.org/10.1111/j.1365-2621.2007.01555.x
Kristinsson HG, Rasco BA (2000) Biochemical and functional properties of Atlantic salmon (Salmo salar) muscle proteins hydrolyzed with various alkaline proteases. J Agric Food Chem 48(3):657–666. https://doi.org/10.1021/jf990447v
Le Gouic AV, Harnedy PA, FitzGerald RJ (2019) Bioactive peptides from fish protein by-products. Bioact Mol Food. https://doi.org/10.1007/978-3-319-78030-6_29
Leng B, Liu XD, Chen QX (2005) Inhibitory effects of anticancer peptide from Mercenaria on the BGC-823 cells and several enzymes. FEBS J 579(5):1187–1190. https://doi.org/10.1016/j.febslet.2004.12.089
Leong AFPK, Seow-Choen F, Tang CL (1998) Diminutive cancers of the colon and rectum: comparison between flat and polypoid cancers. Int J Colorectal Dis 13(4):151–153. https://doi.org/10.1007/s003840050155
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–275
Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7(9):405–410. https://doi.org/10.1016/S1360-1385(02)02312-9
Mohebbi M et al (2008) Geographical spread of gastrointestinal tract cancer incidence in the Caspian Sea region of Iran: spatial analysis of cancer registry data. BMC Cancer 8(1):137–165. https://doi.org/10.1186/1471-2407-8-137
Moheghi N, Afshari JT, Brook A (2011) The cytotoxic effect of zingiber afficinale in breast cancer (MCF7) cell line. Horiz Med Sci 17(3):28–34
Mokhtari MJ, Motamed N, Shokrgozar MA (2008) Evaluation of silibinin on the viability, migration and adhesion of the human prostate adenocarcinoma (PC-3) cell line. Cell Biol Int 32(8):888–892. https://doi.org/10.1016/j.cellbi.2008.03.019
Oyaizu M (1986) Studies on product of browning reaction. Jpn J Nutr Diet 44(6):307–315
Pezeshk S, Ojagh SM, Rezaei M, Shabanpour B (2018) Fractionation of protein hydrolysates of fish waste using membrane ultrafiltration: investigation of antibacterial and antioxidant activities. Probiotics Antimicrob Proteins. https://doi.org/10.1007/s12602-018-9483-y
Razali AN, Amin AM, Sarbon NM (2015) Antioxidant activity and functional properties of fractionated cobia skin gelatin hydrolysate at different molecular weight. Int Food Res J 22(2):651–660
Rustad T, Storrø I, Slizyte R (2011) Possibilities for the utilisation of marine by-products. Int J Food Sci Technol 46(10):2001–2014. https://doi.org/10.1111/j.1365-2621.2011.02736.x
Sheu MJ et al (2008) Ethanol extract of Dunaliella salina induces cell cycle arrest and apoptosis in A549 human nonsmall cell lung cancer. In vivo 22(3):369–378
Sripokar P, Benjakul S, Klomklao S (2019) Antioxidant and functional properties of protein hydrolysates obtained from starry triggerfish muscle using trypsin from albacore tuna liver. Biocatal Agric Biotechnol 17:447–454. https://doi.org/10.1016/j.bcab.2018.12.013
Ug Y, Bhat I, Karunasagar I, Bs M (2018) Antihypertensive activity of fish protein hydrolysates and its peptides. Crit Rev Food Sci Nutr 13:1–12. https://doi.org/10.1080/10408398.2018.1452182
Umayaparvathi S, Meenakshi S, Vimalraj V, Arumugam M, Sivagami G, Balasubramanian T (2014) Antioxidant activity and anticancer effect of bioactive peptide from enzymatic hydrolysate of oyster (Saccostrea cucullata). Biomed Prev Nutr 4(3):343–353. https://doi.org/10.1016/j.bionut.2014.04.006
Yen GC, Wu JY (1999) Antioxidant and radical scavenging properties of extracts from Ganoderma tsugae. Food Chem 65(3):375–379. https://doi.org/10.1016/S0308-8146(98)00239-8
Zhang M, Mu TH (2018) Contribution of different molecular weight fractions to anticancer effect of sweet potato protein hydrolysates by six proteases on HT-29 colon cancer cells. Int J Food Sci Technol 53(2):525–532. https://doi.org/10.1111/ijfs.13625
Zhang M, Mu TH, Sun MJ (2014) Purification and identification of antioxidant peptides from sweet potato protein hydrolysates by Alcalase. J Funct Foods 7:191–200. https://doi.org/10.1016/j.jff.2014.02.012
Acknowledgements
We appreciate the sincere collaboration of the Microbiology Department, Ayatollah Amoli branch of Islamic Azad University for assistance in conduct of the study.
Funding
This study was supported by the Ayatollah Amoli Branch, Islamic Azad University, Amol, Mazandaran province, Iran. The funding body had no role in the design of the study and collection, analysis, and interpretation of data and in writing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest relevant to this study.
Ethical Approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed and this article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yaghoubzadeh, Z., Peyravii Ghadikolaii, F., Kaboosi, H. et al. Antioxidant Activity and Anticancer Effect of Bioactive Peptides from Rainbow Trout (Oncorhynchus mykiss) Skin Hydrolysate. Int J Pept Res Ther 26, 625–632 (2020). https://doi.org/10.1007/s10989-019-09869-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-019-09869-5