Summary
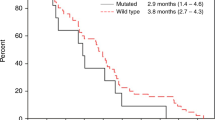
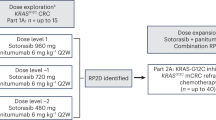
Background Resistance to Epidermal Growth Factor inhibition (EGFRi) in patients with KRAS wild-type (wt) Colorectal Cancer (CRC) may occur as a result of PI3K/AKT/mTOR signaling. We conducted a study to establish the recommended phase II dose (RP2D) and response rate of panitumumab, an EGFRi, plus BKM120, a PI3K inhibitor, in advanced CRC. Methods Patients with chemotherapy refractory KRAS wt CRC, who were EGFRi naive were enrolled. A 3 + 3 dose escalation design was utilized. The starting dose of panitumumab was 6 mg/kg iv every 2 weeks with BKM120 at 60 mg oral daily. Results Nineteen patients were treated and 17 were evaluable for response. The starting dose was not tolerable (mucositis, fatigue). At dose level (DL) 1, three of six patients discontinued treatment due to toxicity, DL − 1 had no significant toxicity. Panitumumab 6 mg/kg iv q 2 weeks with BKM120 60 mg given 5 out of 7 days per week was declared the RP2D. One patient (5.9%) who was PTEN and PIK3CA negative by IHC had a partial response, seven had stable disease, and nine had disease progression. Conclusion Panitumumab (6 mg/kg iv q 2 weeks) with BKM120 60 mg given 5 out of 7 days per week was declared the RP2D. Toxicities including fatigue, rash and mucositis. There was little evidence of activity in this biomarker unselected cohort.


Similar content being viewed by others
References
Canadian Cancer Statistics, Canadian Cancer Society's Advisory Committee on Cancer Statistics (2016)
Malvezzi M et al (2013) European cancer mortality predictions for the year 2013. Ann Oncol 24(3):792–800
Meyerhardt JA, Mayer RJ (2005) Systemic therapy for colorectal cancer. N Engl J Med 352(5):476–487
Ciardiello F, Tortora G (2001) A novel approach in the treatment of cancer: targeting the epidermal growth factor receptor. Clin Cancer Res 7(10):2958–2970
Salomon DS et al (1995) Epidermal growth factor-related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol 19(3):183–232
Mayer A, Takimoto M, Fritz E, Schellander G, Kofler K, Ludwig H (1993) The prognostic significance of proliferating cell nuclear antigen, epidermal growth factor receptor, and mdr gene expression in colorectal cancer. Cancer 71(8):2454–2460
Van Cutsem E (2007) Optimizing administration of epidermal growth factor receptor-targeted agents in the treatment of colorectal cancer. Clin Colorectal Cancer 6(Suppl 2):S60–S65
Amado RG, Wolf M, Peeters M, van Cutsem E, Siena S, Freeman DJ, Juan T, Sikorski R, Suggs S, Radinsky R, Patterson SD, Chang DD (2008) Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol 26(10):1626–1634
Frattini M et al (2007) PTEN loss of expression predicts cetuximab efficacy in metastatic colorectal cancer patients. Br J Cancer 97(8):1139–1145
Loupakis F et al (2009) PTEN expression and KRAS mutations on primary tumors and metastases in the prediction of benefit from cetuximab plus irinotecan for patients with metastatic colorectal cancer. J Clin Oncol 27(16):2622–2629
Sartore-Bianchi A, Martini M, Molinari F, Veronese S, Nichelatti M, Artale S, di Nicolantonio F, Saletti P, de Dosso S, Mazzucchelli L, Frattini M, Siena S, Bardelli A (2009) PIK3CA mutations in colorectal cancer are associated with clinical resistance to EGFR-targeted monoclonal antibodies. Cancer Res 69(5):1851–1857
Mao C, Liao RY, Chen Q (2010) Loss of PTEN expression predicts resistance to EGFR-targeted monoclonal antibodies in patients with metastatic colorectal cancer. Br J Cancer 102(5):940
Mao C, Yang ZY, Hu XF, Chen Q, Tang JL (2012) PIK3CA exon 20 mutations as a potential biomarker for resistance to anti-EGFR monoclonal antibodies in KRAS wild-type metastatic colorectal cancer: a systematic review and meta-analysis. Ann Oncol 23(6):1518–1525
Bendell JC, Rodon J, Burris HA, de Jonge M, Verweij J, Birle D, Demanse D, de Buck SS, Ru QC, Peters M, Goldbrunner M, Baselga J (2012) Phase I, dose-escalation study of BKM120, an oral pan-class I PI3K inhibitor, in patients with advanced solid tumors. J Clin Oncol 30(3):282–290
Eisenhauer EA et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Lux MP et al (2016) The PI3K pathway: background and treatment approaches. Breast Care (Basel) (11, 6):398–404
Liu J, Hu J, Cheng L, Ren W, Yang M, Liu B, Xie L, Qian X (2016) Biomarkers predicting resistance to epidermal growth factor receptor-targeted therapy in metastatic colorectal cancer with wild-type KRAS. Onco Targets Ther 9:557–565
Perrone F et al (2009) PI3KCA/PTEN deregulation contributes to impaired responses to cetuximab in metastatic colorectal cancer patients. Ann Oncol 20(1):84–90
De Roock W et al (2010) Effects of KRAS, BRAF, NRAS, and PIK3CA mutations on the efficacy of cetuximab plus chemotherapy in chemotherapy-refractory metastatic colorectal cancer: a retrospective consortium analysis. Lancet Oncol 11(8):753–762
Huang L et al (2014) Anti-epidermal growth factor receptor monoclonal antibody-based therapy for metastatic colorectal cancer: a meta-analysis of the effect of PIK3CA mutations in KRAS wild-type patients. Arch Med Sci 10(1):1–9
Mackay HJ, Eisenhauer EA, Kamel-Reid S, Tsao M, Clarke B, Karakasis K, Werner HMJ, Trovik J, Akslen LA, Salvesen HB, Tu D, Oza AM (2014) Molecular determinants of outcome with mammalian target of rapamycin inhibition in endometrial cancer. Cancer 120(4):603–610
Laurent-Puig P et al (2009) Analysis of PTEN, BRAF, and EGFR status in determining benefit from cetuximab therapy in wild-type KRAS metastatic colon cancer. J Clin Oncol 27(35):5924–5930
Sood A, McClain D, Maitra R, Basu-Mallick A, Seetharam R, Kaubisch A, Rajdev L, Mariadason JM, Tanaka K, Goel S (2012) PTEN gene expression and mutations in the PIK3CA gene as predictors of clinical benefit to anti-epidermal growth factor receptor antibody therapy in patients with KRAS wild-type metastatic colorectal cancer. Clin Colorectal Cancer 11(2):143–150
Razis E et al (2014) EGFR gene gain and PTEN protein expression are favorable prognostic factors in patients with KRAS wild-type metastatic colorectal cancer treated with cetuximab. J Cancer Res Clin Oncol 140(5):737–748
Van Emburgh BO et al (2014) Acquired resistance to EGFR-targeted therapies in colorectal cancer. Mol Oncol 8(6):1084–1094
Leto SM, Trusolino L (2014) Primary and acquired resistance to EGFR-targeted therapies in colorectal cancer: impact on future treatment strategies. J Mol Med (Berl) 92(7):709–722
Funding
CCTG is supported by the Canadian Cancer Society Research Institute to the Canadian Cancer Trials Group (grant #021039).
This study was carried out by the Canadian Cancer Trials Group. Novartis provided BKM120 and partial financial support for the trial.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
R Goodwin: Honorarium Novartis, Honorarium Amgen.
D Jonker: No conflicts of interest.
E. Chen: No conflicts of Interest.
H Kennecke: No Conflicts of Interest.
M Cabanero: No conflicts of Interest.
M Tsao: No conflicts of interest.
M Vickers: No Conflicts of Interest.
C Bohemier: No Conflicts of Interest.
H Lim: No conflicts of Interest.
H Ritter: No Conflicts of Interest.
D Tu: No Conflicts of Interest.
L Seymour: Funding to support conduct of the trial.
Ethics approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Goodwin, R., Jonker, D., Chen, E. et al. A phase Ib study of a PI3Kinase inhibitor BKM120 in combination with panitumumab in patients with KRAS wild-type advanced colorectal cancer. Invest New Drugs 38, 1077–1084 (2020). https://doi.org/10.1007/s10637-019-00814-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10637-019-00814-3