Summary
Goal-oriented management of traumatic brain injury (TBI) can save the lives and/or improve the long-term outcome of millions of affected patients worldwide. Additionally, enhancing quality of life will save enormous socio-economic costs; however, promising TBI treatment strategies with neuroprotective agents, such as citicoline (CDP-choline), lacked evidence or produced contradictory results in clinical trials. During a prehospital TBI project to optimize early TBI care within 14 Austrian trauma centers, data on 778 TBI patients were prospectively collected. As preceding evaluations suggested a beneficial outcome in TBI patients treated at the Wiener Neustadt Hospital (WNH), we aimed to investigate the potential role of citicoline administration, solely applied in WNH, in those patients. In a retrospective subgroup analysis we compared 67 patients from WNH with citicoline administration and 67 matched patients from other Austrian centers without citicoline use. Patients with Glasgow Coma Scale score <13 on site and/or Abbreviated Injury Scale of the region “head” >2 were included. Our analysis revealed significantly reduced rates of intensive care unit (ICU) mortality (5% vs. 24%, p < 0.01), in-hospital mortality (9% vs. 24%, p = 0.035) and 6‑month mortality (13% vs. 28%, p = 0.031), as well as of unfavorable outcome (34% vs. 57%, p = 0.015) and observed vs. expected ratio for mortality (0.42 vs. 0.84) in the WNH (citicoline receivers) group. Despite the limitations of a retrospective subgroup analysis our findings suggest a possible correlation between early and consequent citicoline administration and beneficial outcomes. Therefore, we aim to set up an initiative for a prospective, multicenter randomized controlled trial with citicoline in sTBI (severe TBI) patients.
Similar content being viewed by others
References
Hyder AA, Wunderlich CA, Puvanachandra P, Gururaj G, Kobusingye OC. The impact of traumatic brain injuries: a global perspective. NeuroRehabilitation. 2007;22(5):341–53.
Mauritz W, Brazinova A, Majdan M, Leitgeb J. Epidemiology of traumatic brain injury in Austria. Wien Klin Wochenschr. 2014;126(1–2):42–52.
Andriessen TM, Jacobs B, Vos PE. Clinical characteristics and pathophysiological mechanisms of focal and diffuse traumatic brain injury. J Cell Mol Med. 2010;14(10):2381–92.
Dhillon HS, Dose JM, Scheff SW, Prasad MR. Time course of changes in lactate and free fatty acids after experimental brain injury and relationship to morphologic damage. Exp Neurol. 1997;146(1):240–9.
Baskaya MK, Dogan A, Rao AM, Dempsey RJ. Neuroprotective effects of citicoline on brain edema and blood-brain barrier breakdown after traumatic brain injury. J Neurosurg. 2000;92(3):448–52.
Dixon CE, Bao J, Long DA, Hayes RL. Reduced evoked release of acetylcholine in the rodent hippocampus following traumatic brain injury. Pharmacol Biochem Behav. 1996;53(3):679–86.
Dixon CE, Ma X, Marion DW. Reduced evoked release of acetylcholine in the rodent neocortex following traumatic brain injury. Brain Res. 1997;749(1):127–30.
Grieb P. Neuroprotective properties of citicoline: facts, doubts and unresolved issues. CNS Drugs. 2014;28(3):185–93.
Secades JJ. Citicoline: pharmacological and clinical review, 2010 update. Rev Neurol. 2011;52(Suppl 2):S1–S62.
Blount PJ, Nguyen CD, McDeavitt JT. Clinical use of cholinomimetic agents: a review. J Head Trauma Rehabil. 2002;17(4):314–21.
Lopez GCI, Agut J, Von Borstel R, Wurtman RJ. Metabolism of cytidine (5′)-diphosphocholine (cdp-choline) following oral and intravenous administration to the human and the rat. Neurochem Int. 1987;11(3):293–7.
Dixon CE, Ma X, Marion DW. Effects of CDP-choline treatment on neurobehavioral deficits after TBI and on hippocampal and neocortical acetylcholine release. J Neurotrauma. 1997;14(3):161–9.
Lopez GCI, Agut J, Ortiz JA, Wurtman RJ. Effects of orally administered cytidine 5′-diphosphate choline on brain phospholipid content. J Nutr Biochem. 1992;3(6):313–5.
G-Coviella I, Wurtman RJ. Enhancement by cytidine of membrane phospholipid synthesis. J Neurochem. 1992;59(1):338–43.
Dorman RV, Dabrowiecki Z, Horrocks LA. Effects of CDPcholine and CDPethanolamine on the alterations in rat brain lipid metabolism induced by global ischemia. J Neurochem. 1983;40(1):276–9.
Zweifler RM. Membrane stabilizer: citicoline. Curr Med Res Opin. 2002;18(Suppl 2):s14–s7.
Savci V, Wurtman RJ. Effect of cytidine on membrane phospholipid synthesis in rat striatal slices. J Neurochem. 1995;64(1):378–84.
Le Poncin-Lafitte M, Duterte D, Lageron A, Rapin JR. CDP choline and experimental cerebrovascular disorder. Agressologie. 1986;27(5):413–6.
Ma X, Zhang H, Pan Q, Zhao Y, Chen J, Zhao B, et al. Hypoxia/Aglycemia-induced endothelial barrier dysfunction and tight junction protein downregulation can be ameliorated by citicoline. PLOS ONE. 2013;8(12):e82604.
Spiers PA, Hochanadel G. Citicoline for traumatic brain injury: report of two cases, including my own. J Int Neuropsychol Soc. 1999;5(3):260–4.
Lazowski T, Kierul K, Bartnicki M, Mayzner-Zawadzka E, Toczylowska B, Ryba M, et al. Effects of citicoline treatment in patients with isolated head trauma: a randomized trial. Crit Care. 2003;7(2):1.
Zafonte RD, Bagiella E, Ansel BM, Novack TA, Friedewald WT, Hesdorffer DC, et al. Effect of citicoline on functional and cognitive status among patients with traumatic brain injury: Citicoline Brain Injury Treatment Trial (COBRIT). JAMA. 2012;308(19):1993–2000.
Conant R, Schauss AG. Therapeutic applications of citicoline for stroke and cognitive dysfunction in the elderly: a review of the literature. Altern Med Rev. 2004;9(1):17–31.
Clark WM, Warach SJ, Pettigrew LC, Gammans RE, Sabounjian LA, Citicoline Stroke Study Group. A randomized dose-response trial of citicoline in acute ischemic stroke patients. Neurology. 1997;49(3):671–8. doi:10.1212/wnl.49.3.671.
Brazinova A, Majdan M, Leitgeb J, Trimmel H, Mauritz W. Factors that may improve outcomes of early traumatic brain injury care: prospective multicenter study in Austria. Scand J Trauma Resusc Emerg Med. 2015;23(1):53.
Committee on Trauma, American College of Surgeons.. Resources for optimal care of the injured patient 2014 (6th ed) 2014. https://www.facs.org/quality-programs/trauma/vrc/resources. Accessed 30 May 2017.
Garthe E, States JD, Mango NK. Abbreviated injury scale unification: the case for a unified injury system for global use. J Trauma. 1999;47(2):309–23.
Marmarou A, Lu J, Butcher I, McHugh GS, Murray GD, Steyerberg EW, et al. Prognostic value of the Glasgow Coma Scale and pupil reactivity in traumatic brain injury assessed pre-hospital and on enrollment: an IMPACT analysis. J Neurotrauma. 2007;24(2):270–80.
Wilson JT, Pettigrew LE, Teasdale GM. Structured interviews for the Glasgow Outcome Scale and the extended Glasgow Outcome Scale: guidelines for their use. J Neurotrauma. 1998;15(8):573–85.
Teasdale GM, Pettigrew LE, Wilson JT, Murray G, Jennett B. Analyzing outcome of treatment of severe head injury: a review and update on advancing the use of the Glasgow Outcome Scale. J Neurotrauma. 1998;15(8):587–97.
Calatayud Maldonado VC, Calatayud Perez JB, Aso Escario J. Effects of CDP-choline on the recovery of patients with head injury. J Neurol Sci. 1991;103(Suppl):S15–S8.
Hukkelhoven CW, Steyerberg EW, Rampen AJ, Farace E, Habbema JD, Marshall LF, et al. Patient age and outcome following severe traumatic brain injury: an analysis of 5600 patients. J Neurosurg. 2003;99(4):666–73.
Maas AI, Hukkelhoven CW, Marshall LF, Steyerberg EW. Prediction of outcome in traumatic brain injury with computed tomographic characteristics: a comparison between the computed tomographic classification and combinations of computed tomographic predictors. Neurosurgery. 2005;57(6):1173–82, discussion –1182.
Boyd CR, Tolson MA, Copes WS. Evaluating trauma care: the TRISS method. Trauma Score and the Injury Severity Score. J Trauma. 1987;27(4):370–8.
Majdan M, Lingsma HF, Nieboer D, Mauritz W, Rusnak M, Steyerberg EW. Performance of IMPACT, CRASH and Nijmegen models in predicting six month outcome of patients with severe or moderate TBI: an external validation study. Scand J Trauma Resusc Emerg Med. 2014;22:68.
Maas AI, Marmarou A, Murray GD, Teasdale SG, Steyerberg EW. Prognosis and clinical trial design in traumatic brain injury: the IMPACT study. J Neurotrauma. 2007;24(2):232–8.
Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons. Guidelines for the management of severe traumatic brain injury. J Neurotrauma. 2007;24(Suppl 1):S1–S106.
Herzer G, Illievich U, Voelckel WG, Trimmel H. Current practice in neurocritical care of patients with subarachnoid haemorrhage and severe traumatic brain injury: results of the Austrian Neurosurvey study. Wien Klin Wochenschr. 2016;128(17–18):649–57.
Rusnak M, Janciak I, Majdan M, Wilbacher I, Mauritz W. Severe traumatic brain injury in Austria I: introduction to the study. Wien Klin Wochenschr. 2007;119(1–2):23–8.
Rosso A, Brazinova A, Janciak I, Wilbacher I, Rusnak M, Mauritz W. Severe traumatic brain injury in Austria II: epidemiology of hospital admissions. Wien Klin Wochenschr. 2007;119(1–2):29–34.
Lenartova L, Janciak I, Wilbacher I, Rusnak M, Mauritz W. Severe traumatic brain injury in Austria III: prehospital status and treatment. Wien Klin Wochenschr. 2007;119(1–2):35–45.
Mauritz W, Janciak I, Wilbacher I, Rusnak M. Severe traumatic brain injury in Austria IV: intensive care management. Wien Klin Wochenschr. 2007;119(1–2):46–55.
Leitgeb J, Erb K, Mauritz W, Janciak I, Wilbacher I, Rusnak M. Severe traumatic brain injury in Austria V: CT findings and surgical management. Wien Klin Wochenschr. 2007;119(1–2):56–63.
Rusnak M, Janciak I, Majdan M, Wilbacher I, Mauritz W. Severe traumatic brain injury in Austria VI: effects of guideline-based management. Wien Klin Wochenschr. 2007;119(1–2):64–71.
Adibhatla RM, Hatcher JF. Citicoline mechanisms and clinical efficacy in cerebral ischemia. J Neurosci Res. 2002;70(2):133–9.
Bustamante A, Giralt D, Garcia-Bonilla L, Campos M, Rosell A, Montaner J. Citicoline in pre-clinical animal models of stroke: a meta-analysis shows the optimal neuroprotective profile and the missing steps for jumping into a stroke clinical trial. J Neurochem. 2012;123(2):217–25.
Tayebati SK, Amenta F. Choline-containing phospholipids: relevance to brain functional pathways. Clin Chem Lab Med. 2013;51(3):513–21.
Lozano R. CDP-choline in the treatment of cranio-encephalic traumata. J Neurol Sci. 1991;103(Suppl):S43–S7.
Leon-Carrion J, Dominguez-Roldan JM, Murillo-Cabezas F, del Rosario Dominguez-Morales M, Munoz-Sanchez MA. The role of citicholine in neuropsychological training after traumatic brain injury. NeuroRehabilitation. 2000;14(1):33–40.
Levin HS. Treatment of postconcussional symptoms with CDP-choline. J Neurol Sci. 1991;103(Suppl):S39–S42.
Secades JJ. Probably role of citicoline in stroke rehabilitation: review of the literature. Rev Neurol. 2012;54(3):173–9.
Secades JJ. Citicoline for the treatment of head injury: a systematic review and meta-analysis of controlled clinical trials. J Trauma Treat. 2014;4(1):4.
Adibhatla RM. Citicoline in stroke and TBI clinical trials. Nat Rev Neurol. 2013;9(3):173.
Ruff RL, Riechers RG. Effective treatment of traumatic brain injury: learning from experience. JAMA. 2012;308(19):2032–3.
Fresta M, Wehrli E, Puglisi G. Enhanced therapeutic effect of cytidine-5′-diphosphate choline when associated with GM1 containing small liposomes as demonstrated in a rat ischemia model. Pharm Res. 1995;12(11):1769–74.
Horn SD, Corrigan JD, Bogner J, Hammond FM, Seel RT, Smout RJ, et al. Traumatic brain injury-practice based evidence study: design and patients, centers, treatments, and outcomes. Arch Phys Med Rehabil. 2015;96(8 Suppl):S178–S196.e15.
Phillips JP, Devier DJ, Feeney DM. Rehabilitation pharmacology: bridging laboratory work to clinical application. J Head Trauma Rehabil. 2003;18(4):342–56.
Acknowledgements
The authors acknowledge the members of the International Neurotrauma Research Organization (INRO) and collaborative partners for their work and the collection of data used for analyses in this article. In particular, we want to express our special thanks and dedicate this publication to Walter Mauritz who provided the impetus for this investigation. Walter passed away in May 2015. He was Vice President of the INRO and Professor of Anesthesiology and Intensive Care Medicine at Trauma Hospital “Lorenz Boehler” 2 in Vienna. We thank Chiesi Pharmaceuticals for providing a research grant to facilitate data analysis and writing assistance. We thank Ulrike Posch for providing medical writing services on behalf of Chiesi Pharmaceuticals.
Funding
The INRO prehospital project was funded jointly by the Austrian Ministry of Health (Contract Oct. 15, 2008) and by the Austrian Worker’s Compensation Board (AUVA; FK 11/2008 and FK 11/2010). Data analysis was supported by a grant from AUVA (FK 09/13). INRO is supported by an annual grant from Mrs. Ala Auersperg-Isham and Mr. Ralph Isham through the Neurotrauma Foundation, and by small donations from various sources. Chiesi Pharmaceuticals provided a non-educational grant to facilitate initial data analysis at INRO and writing assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
H. Trimmel reports non-financial support from Chiesi Pharmaceuticals during the conduct of the study. M. Majdan, A. Wodak, G. Herzer, D. Csomor and A. Brazinova declare that they have no competing interests.
Additional information
Author contributions H. Trimmel concepted, initiated and designed the study, collected, analyzed and interpreted data, drafted and critically revised the manuscript. M. Majdan performed the statistical analysis, contributed to data interpretation and helped to draft and revise the manuscript. A. Wodak, G. Herzer and D. Csomor contributed to data acquisition and data analysis, and helped to revise the manuscript. A. Brazinova conceived the INRO prehospital TBI project, participated in its design, coordination and data analysis, and critically revised the manuscript. All authors read and approved the final manuscript.
Caption Electronic Supplementary Material
508_2017_1240_MOESM1_ESM.tif
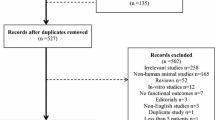
S1 Fig. Flow diagram of participating Austrian centers and patients from the INRO prehospital database. (*lost to follow up due to incomplete dataset. AIS abbreviated injury scale, GCS Glasgow coma scale, INRO International Neurotrauma Research Organization, WNH Wiener Neustadt Hospital)
508_2017_1240_MOESM2_ESM.tif
S2 Fig. Pressure parameters and heart rate of survivors and non-survivors under citicoline therapy. (CPP cerebral perfusion pressure, HR heart rate, ICP intracranial pressure, MAP mean arterial pressure)
Rights and permissions
About this article
Cite this article
Trimmel, H., Majdan, M., Wodak, A. et al. Citicoline in severe traumatic brain injury: indications for improved outcome. Wien Klin Wochenschr 130, 37–44 (2018). https://doi.org/10.1007/s00508-017-1240-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00508-017-1240-9