Abstract
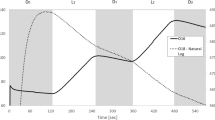
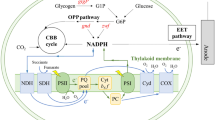
Certain anaerobic bacteria, termed electrogens, produce an electric current when electrons from oxidized organic molecules are deposited to extracellular metal oxide acceptors. In these heterotrophic “metal breathers”, the respiratory electron transport chain (R-ETC) works in concert with membrane-bound cytochrome oxidases to transfer electrons to the extracellular acceptors. The diversity of bacteria able to generate an electric current appears more widespread than previously thought, and aerobic phototrophs, including cyanobacteria, possess electrogenic activity. However, unlike heterotrophs, cyanobacteria electrogenic activity is light dependent, which suggests that a novel pathway could exist. To elucidate the electrogenic mechanism of cyanobacteria, the current studies used site-specific inhibitors to target components of the photosynthetic electron transport chain (P-ETC) and cytochrome oxidases. Here, we show that (1) P-ETC and, particularly, water photolysed by photosystem II (PSII) is the source of electrons discharged to the environment by illuminated cyanobacteria, and (2) water-derived electrons are transmitted from PSII to extracellular electron acceptors via plastoquinone and cytochrome bd quinol oxidase. Two cyanobacterial genera (Lyngbya and Nostoc) displayed very similar electrogenic responses when treated with P-ETC site-specific inhibitors, suggesting a conserved electrogenic pathway. We propose that in cyanobacteria, electrogenic activity may represent a form of overflow metabolism to protect cells under high-intensity light. This study offers insight into electron transfer between phototrophic microorganisms and the environment and expands our knowledge into biologically based mechanisms for harnessing solar energy.






Similar content being viewed by others
References
Adams WW, Zarter CR, Mueh KE, Amiard V, Demmig-Adams B (2008) Energy dissipation and photoinhibition: a continuum of photoprotection. In: Demmig-Adams B, Adams WW, Mattoo AK (eds) Photoprotection, photoinhibition, gene regulation, and environment. Springer, Dordrecht, pp 49–64
Berry S, Schneider D, Vermaas WFJ, Rogner M (2002) Electron transport routs in whole cells of Synechocystis sp. strain PCC 6803: the role of the cytochrome bd-type oxidase. Biochemistry 41:3422–3429
Chaudhuri SK, Lovley DR (2003) Electricity generation by direct oxidation of glucose in mediatorless microbial fuel cells. Nat Biotechnol 21:1229–1232
Cooley JW, Vermaas WF (2001) Succinate dehydrogenase and other respiratory pathways in thylakoid membranes of Synechocystis sp. strain PCC 6803: capacity comparisons and physiological function. J Bacteriol 183:4251–4258
Dismukes GC, Carrieri D, Bennette N, Ananyev GM, Posewitz MC (2008) Aquatic phototrophs: efficient alternatives to land-based crops for biofuels. Curr Opin Biotechnol 19:235–240
Esteve-Nunez A, Sosnik J, Visconti P, Lovley DR (2008) Fluorescent properties of c-type cytochromes reveal their potential role as an extracytoplasmic electron sink in Geobacter sulfurreducens. Environ Microbiol 10:497–505
Gonen-Zurgil Y, Carmeli-Schwartz Y, Sukenik A (1996) Selective effect of the herbicide DCMU on unicellular algae—a potential tool to maintain monoalgal mass culture of Nannochloropsis. J Appl Phycol 8:415–419
Greenwell HC, Laurens LM, Shields RJ, Lovitt RW, Flynn KJ (2010) Placing microalgae on the biofuels priority list: a review of the technological challenges. J Royal Soc Interface (in press)
Gutthann F, Egert M, Marques A, Appel J (2007) Inhibition of respiration and nitrate assimilation enhances photohydrogen evolution under low oxygen concentrations in Synechocystis sp. PCC 6803. Biochim Biophys Acta 1767:161–169
Howitt CA, Vermaas WF (1998) Quinol and cytochrome oxidases in the cyanobacterium Synechocystis sp. PCC 6803. Biochemistry 37:17944–17951
Karapetyan NV (2007) Non-photochemical quenching of fluorescence in cyanobacteria. Biochemistry 72:1127–1135
Kilian O, Steunou AS, Fazeli F, Bailey S, Bhaya D, Grossman AR (2007) Responses of a thermophilic Synechococcus isolate from the microbial mat of Octopus Spring to light. Appl Environ Microbiol 73:4268–4278
Kirilovsky DL, Ajlani G, Picaud M, Etienne AL (1989) Mutations responsible for high light sensitivity in an atrazine-resistant mutant of Synechocystis 6714. Plant Mol Biol 13:355–363
Kufryk GI, Vermaas WF (2006) Sll1717 affects the redox state of the plastoquinone pool by modulating quinol oxidase activity in thylakoids. J Bacteriol 188:1286–1294
Lovley DR (2008) The microbe electric: conversion of organic matter to electricity. Curr Opin Biotechnol 19:1–8
Matoo AK, Pick U, Hoffman-Falk H, Edelman M (1981) The rapidly metabolized 32,000-dalton polypeptide of the chloroplast is the “proteinaceous shield” regulating photosystem II electron transport and mediating diuron herbicide sensitivity. Proc Acad Natl Sci U S A 78:1572–1576
Meyer J (1986) Photosynthetic and respiratory electron transport in a cyanobacterium. Photosynth Res 9:1573–1579
Mogi T, Miyoshi H (2009) Properties of cytochrome bd plastoquinol oxidase from the cyanobacterium Synechocystis sp. PCC 6803. J Biochem 145:395–401
Nomura CT, Persson S, Shen G, Inoue-Sakamoto K, Bryant DA (2006) Characterization of two cytochrome oxidase operons in the marine cyanobacterium Synechococcus sp. PCC 7002: inactivation of ctaDI affects the PS I:PS II ratio. Photosynth Res 87:215–228
Paumann M, Regelsberger G, Obinger C, Peschek GA (2005) The bioenergetic role of dioxygen and the terminal oxidase(s) in cyanobacteria. Biochim Biophys Acta 1707:231–253
Pils D, Schmetterer G (2001) Characterization of three bioenergetically active respiratory terminal oxidases in the cyanobacterium Synechocystis sp. strain PCC 6803. FEMS Microbiol Lett 203:217–222
Pils D, Gregor W, Schmetterer G (1997) Evidence for in vivo activity of three distinct respiratory terminal oxidases in the cyanobacterium Synechocystis sp. strain PCC6803. FEMS Microbiol Lett 152:83–88
Pisciotta J, Zou Y, Baskakov IV (2010) Light-dependent electrogenic activity of cyanobacteria. PLoS ONE 5:e10821
Reguera G, McCarthy KD, Mehta T, Nicoll JS, Tuominen MT, Lovley DR (2005) Extracellular electron transfer via microbial nanowires. Nature 435:1098–1101
Roberts AG, Bowman MK, Kramer DM (2004) The inhibitor DBMIB provides insight into the functional architecture of the Qo site in the cytochrome b6f complex. Biochemistry 43:7707–7716
Roeselers G, Loosdrecht MC, Muyzer G (2008) Phototrophic biofilms and their potential applications. J Appl Phycol 20:227–235
Rosenbaum M, He Z, Angenent LT (2010) Light energy to bioelectricity: photosynthetic microbial fuel cells. Curr Opin Biotechnol 21:1–6
Schneider D, Berry S, Rich P, Seidler A, Rogner M (2001) A regulatory role of the PetM subunit in a cyanobacterial cytochrome b6f complex. J Biol Chem 276:16780–16785
Shi L, Squier TC, Zachara JM, Fredrickson JK (2007) Respiration of metal (hydr)oxides by Shewanella and Geobacter: a key role for multihaem c-type cytochromes. Mol Microbiol 65:12–20
Trubitsin BV, Ptushenko VV, Koksharova OA, Mamedov MD, Vitukhnovskaya LA, Grigor'ev IA, Semenov AY, Tikhonov AN (2005) EPR study of electron transport in the cyanobacterium Synechocystis sp. PCC 6803: oxygen-dependent interrelations between photosynthetic and respiratory electron transport chains. Biochim Biophys Acta 1708:238–249
Tsunoyama Y, Bernát G, Dyczmons NG, Schneider D, Rögner M (2009) Multiple Rieske proteins enable short- and long-term light adaptation of Synechocystis sp. PCC 6803. J Biol Chem 284:27875–27883
Vermaas WF, Shen G, Styring S (1994) Electrons generated by photosystem II are utilized by an oxidase in the absence of photosystem I in the cyanobacterium Synechocystis sp. PCC 6803. FEBS Lett 337:103–108
Yan J, Kurisu G, Cramer WA (2006) Intraprotein transfer of the quinone analogue inhibitor 2,5-dibromo-3-methyl-6-isopropyl-p-benzoquinone in the cytochrome b6f complex. Proc Acad Natl Sci U S A 103:69–74
Zang LY, Sommerburg O, van Kuijk FJGM (1997) Absorbance changes of carotenoids in different solvents. Free Radic Biol Med 23:1086–1089
Zou Y, Pisciotta J, Billmyre RB, Baskakov IV (2009) Photosynthetic microbial fuel cells with positive light response. Biotechnol Bioeng 104:939–946
Zou Y, Pisciotta J, Baskakov IV (2010) Nanostructured polypyrrole-coated anode for sun-powered microbial fuel cells. Bioelectrochem 79:50–56
Acknowledgments
This research was supported by Elkins Professorship Award to IVB.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Suppl. Fig. 1
Effect of atrazine on electrogenic activity of Lyngbya (a) or Nostoc (b). Both cultures were dosed first with 25 μM of atrazine (left arrows) followed by an additional 75 μM (right arrows). Periods of illumination or darkness are indicated by white or black bars along x-axis, respectively (JPEG 42 kb)
Rights and permissions
About this article
Cite this article
Pisciotta, J.M., Zou, Y. & Baskakov, I.V. Role of the photosynthetic electron transfer chain in electrogenic activity of cyanobacteria. Appl Microbiol Biotechnol 91, 377–385 (2011). https://doi.org/10.1007/s00253-011-3239-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3239-x