Abstract
Background
The term ‘metabolic syndrome’ refers to a clustering of specific cardiovascular disease (CVD) risk factors whose underlying pathophysiology is thought to be related to insulin resistance.
Methods
Since the term is widely used in research and clinical practice, we undertook an extensive review of the literature in relation to the syndrome’s definition, underlying pathogenesis, association with cardiovascular disease and to the goals and impact of treatment.
Discussion
While there is no question that certain CVD risk factors are prone to cluster, we found that the metabolic syndrome has been imprecisely defined, there is a lack of certainty regarding its pathogenesis, and there is considerable doubt regarding its value as a CVD risk marker. Our analysis indicates that too much critically important information is missing to warrant its designation as a ‘syndrome’.
Conclusion
Until much-needed research is completed, clinicians should evaluate and treat all CVD risk factors without regard to whether a patient meets the criteria for diagnosis of the ‘metabolic syndrome’.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
For most of the 20th century, cardiovascular disease (CVD) was identified as the major cause of morbidity and mortality in the developed world. During this period there was considerable effort to understand the underlying biology of the disease and to identify the contributing risk factors. As risk factors were identified, it became apparent that more than one were often present in the same individual. Towards the end of the century, the clustering of CVD risk factors was first described, most notably the simultaneous presence of obesity, type 2 diabetes, hyperlipidaemia, and hypertension [1–3]. Although insulin resistance (i.e. resistance to insulin-stimulated glucose uptake) as a feature of type 2 diabetes was first described many years earlier [4], hyperinsulinaemia was also found to be a key feature of type 2 diabetes [5, 6], as well as hyperlipidaemia [7–9], obesity [10–13] and hypertension [12–14]. In addition, a cluster of heart disease risk factors seemed clearly related to type 2 diabetes [15].
This risk factor clustering, and its association with insulin resistance, led investigators to propose the existence of a unique pathophysiological condition, called the ‘metabolic’ [1–3] or ‘insulin resistance’ [11] syndrome. This concept was unified and extended with the landmark publication of Reaven’s 1988 Banting Medal award lecture [16]. Reaven postulated that insulin resistance and its compensatory hyperinsulinaemia predisposed patients to hypertension, hyperlipidaemia and diabetes, and thus was the underlying cause of much CVD. Although obesity was not included in Reaven’s primary list of disorders caused by insulin resistance, he acknowledged that it, too, was correlated with insulin resistance or hyperinsulinaemia, and that the obvious ‘treatment’ for what he termed ‘Syndrome X’ was weight maintenance (or weight loss) and physical activity.
Reaven’s seminal paper was followed by many studies documenting the clustering of CVD risk factors and their relationship to insulin resistance [17–25]. Indeed, since Reaven’s publication in 1988, a recent Medline search for articles using the key words ‘syndrome X’ or ‘insulin resistance syndrome’ or ‘metabolic syndrome’ (conducted 28.01.2005) identified 4,646 citations, with 3,948 studies performed on human subjects.
The term ‘metabolic syndrome’ has now taken hold in the medical literature. It has been defined and institutionalised, principally by the World Health Organization (WHO) [26] and the Third Report of the National Cholesterol Education Program’s Adult Treatment Panel (ATP) III [27, 28], albeit with different definitions. In addition, other organisations have developed similar, but again not identical, definitions [29, 30]. The fact that a version of the metabolic syndrome has its own ICD-9 code (277.7) also suggests that it is well thought out [31, 32].
In this review we examine the evidence for its definition and underlying pathogenesis, as well as analysing the evidence for its association with CVD. We also discuss the evidence for the goals and impact of treatment. We mainly focused our review on papers addressing the metabolic syndrome as defined by ATPIII, the definition that appears to be used most often in the literature. Because the ATPIII and WHO definitions are sometimes used almost interchangeably or compared with one another, we also examined the literature that used the WHO criteria.
For two reasons, we did not consider papers whose focus was on the ability of the metabolic syndrome to predict diabetes. First, ample data show that the presence of the metabolic syndrome is effective in predicting the future risk of diabetes. That association, however, is probably due to the fact that the definition of the syndrome includes glucose intolerance, i.e. IFG or IGT, themselves powerful predictors of future diabetes. Second, the practical use of diagnosing metabolic syndrome has not centred on its power to predict diabetes, but rather on its being a multivariate risk factor for CVD.
This review argues that the metabolic syndrome is not nearly as well defined and characterised as often assumed, and that the notion that it is a useful marker of CVD risk above and beyond the risk associated with its individual components is uncertain. In addition, although certain CVD risk factors undoubtedly occur together more often than expected by chance, the underlying pathophysiology of the syndrome is unclear. Moreover, the list of risk factors comprising the cluster is not grounded by well-defined criteria. Therefore, this manuscript is intended as a cautionary reminder to practitioners, and as an urgent call for further research.
Our analysis addresses three key questions related to the metabolic syndrome:
-
1.
How clear is the existing definition of the metabolic syndrome for diagnostic purposes?
-
A.
How useful is the syndrome definition in predicting CVD risk? Do the individual components of the syndrome convey ‘risk’ differently from the syndrome as a whole?
-
B.
Is the cluster of symptoms associated with the syndrome the result of a common underlying pathological process?
-
A.
-
2.
Does the treatment of the metabolic syndrome differ from the treatment of its individual components?
-
3.
What additional work should be done to improve our current knowledge of the metabolic syndrome?
Clarity of the existing definition
The textbox (Definitions of the metabolic syndrome) shows the ATPIII and WHO definitions of the metabolic syndrome. These definitions raise three important issues. First, some of the criteria used for defining the syndrome are ambiguous or incomplete [26–28]. For example, it is unclear whether the blood pressure definition is systolic pressure >130 mmHg and diastolic >85 mmHg or whether it is either >130 mmHg or >85 mmHg. It is also not defined whether a patient with hypertension who is now normotensive meets the blood pressure criterion, nor is it specified how blood pressure should be measured, e.g. supine, sitting, mean of two measurements. Given that there is no widespread agreement on the method for measuring waist circumference, it is also unclear how that factor should be determined. Does a patient previously diagnosed with IFG, but who now has normal glucose levels because of modest weight loss, meet the ATPIII glucose criteria?
Definitions of the metabolic syndrome Any three or more of the following criteria: 1. Waist circumference: >102 cm in men and >88 cm in women 2. Serum triglycerides: >1.7 mmol/l 3. Blood pressure: >130/85 mmHg 4. HDL-cholesterol: <1.0 mmol/l in men and <1.3 mmol/l in women 5. Serum glucose: >6.1 mmol/l (>5.6 mmol/l may be applicable) WHO definition [26] Diabetes or IFG or IGT or insulin resistance (assessed by clamp studies), plus at least two of the following criteria: 1. Waist-to-hip ratio >0.90 in men or >0.85 in women 2. Serum triglycerides >1.7 mmol/l or HDL-cholesterol <0.9 mmol/l in men and <1.0 mmol/l in women 3. Blood pressure >140/90 mmHg 4. Urinary albumin excretion rate >20 μg/min or albumin : creatinine ratio >30 mg/g |
Such ambiguities affect the sensitivity and specificity of the diagnosis and have undoubtedly led some physicians to diagnose the metabolic syndrome in patients who would not be labelled as such by other providers. The impact of a false positive or a false negative diagnosis has not been reported.
Second, it is apparent is that the definitions of the syndrome differ in the criteria listed. For example, microalbuminuria is listed in the WHO criteria, but not in the ATPIII; insulin resistance (as measured under hyperinsulinaemic–euglycaemic conditions) is relevant for WHO, but not for ATPIII. And while only an elevated fasting plasma glucose value is considered important in the ATPIII definition, the WHO criteria recognise any measure whatsoever of insulin resistance. Although it would promote better understanding of the justification for the criteria selected, no review of the clinical evidence for inclusion or exclusion criteria for either of the two definitions of the syndrome has been published to date.
Third and finally, the originally stated rationale for the criteria is that the syndrome components are associated with insulin resistance [26, 27]. But, as discussed below, there is considerable doubt whether all patients with the metabolic syndrome are indeed insulin-resistant. More recently, a review of the ATPIII definition [28] broadened the aetiological basis for the syndrome from insulin resistance alone, to include ‘obesity and disorders of adipose tissue’, as well as a ‘constellation of independent factors that indicate specific components of the metabolic syndrome’. However, it remains unclear why some factors associated with the latter two categories of abnormalities have not been included in the definition.
The fact that there are cutpoints for the various risk factors implies that values above the specified thresholds are associated with excess risk, yet the rationale for the specific cutpoints, i.e. as opposed to higher or lower values, has never been delineated. Laaka et al. [33], in a study conducted in men with the metabolic syndrome, showed that CVD and overall mortality was more consistently increased using a waist circumference criterion of 102 cm rather than 94 cm. Other investigators [34] found that reducing the threshold for impaired fasting glucose from 6.1 to 5.6 mmol/l did not materially change the hazard ratio (HR) for risk of CHD, though it did increase the number of individuals identified. Other components of the syndrome show a continuous relationship with CVD risk [35]. Although the thresholds defining the syndrome are generally derived from other well-established guidelines, we found no study that systematically examined the impact of all the metabolic syndrome thresholds on the risk of CVD; nor did we find a study that sought to optimise the positive predictive value of the definition by changing the cutpoints of the risk factors.
Some of the criteria (e.g. waist circumference, HDL) have sex-specific cutpoints, implying that the relationship between the risk factor level and outcomes differs between the sexes. However, we found no evidence that warrants establishing the sex-specific cutpoints used in the criteria as they relate to CVD risk. It is, for example, not known whether the same intra-abdominal fat mass carries a different risk in men than in women. An analogous argument can be made regarding whether cutpoints should vary according to race and ethnic groups.
There is ample evidence to show that CVD risk is a function of the criteria cited in the definitions of the metabolic syndrome, but it is unjustified to assume that the optimal predictive power would be obtained by arbitrary dichotomies. Risk is a progressive function of, for example, hyperglycaemia and hypertension, and cannot simply be regarded as present or absent, depending on whether thresholds are exceeded or not.
Although the WHO and ATPIII definitions generally identify the same individuals, important differences have been found [36, 37]. Ford and Giles [36] showed that in the National Health and Nutrition Examination Survey (NHANES), a representative sample of the adult US population, about the same proportions were identified as having the syndrome by the WHO or ATPIII criteria (25.1 vs 23.9%, respectively). However, about 15 to 20% of individuals were classified as having the syndrome by one definition but not the other, with equal discordance. Meigs et al. [37] determined the prevalence of the syndrome, defined by ATPIII or WHO criteria, in a population of non-Hispanic Whites and Mexican–American subjects in San Antonio and in subjects participating in the Framingham Offspring Study. Although the syndrome was common in these populations (affecting 20–30%), more Mexican–American men were classified as having the syndrome using the WHO definition, whereas the ATPIII criteria classified more Mexican–American women. Depending on the sex and ethnicity of the populations, the prevalence of metabolic syndrome varied up to 24% between the two definitions.
The question of how to define a syndrome (i.e. what factors comprise the syndrome) rests in large part on the purpose of the construct. A syndrome can be defined on the basis of its ability to predict (a) future adverse event(s). Such a definition implies that the risk associated with having the syndrome is greater than the sum of its parts, and that the factors included have greater predictive power than do other combinations. Alternatively, if the syndrome purports to identify factors related to a unifying pathological process (e.g. insulin resistance/hyperinsulinaemia), then the definition should include all the factors clearly associated with that underlying pathophysiology, such that there is little ambiguity regarding the aetiology of the clustering. If the aetiology is unclear, it becomes much more difficult to decide what factors to include in the definition, since the word ‘cluster’ itself can be ambiguous.
In the case of the metabolic syndrome, the existing definition attempts to bridge both constructs, and as will be pointed out in the following two Sections, does not succeed very well with either.
Relationship between CVD risk and the metabolic syndrome
Many studies have shown that patients diagnosed with the metabolic syndrome, by either the ATPIII or WHO definition (or by their modifications), have more prevalent cardiovascular disease or are at greater risk of developing it [33, 38–50]. In these studies, the increased CVD risk in patients with the syndrome ranged from 30 to 400%; this wide variation is probably due to the population studied, the precise definition of the syndrome adopted, and to the length of follow-up.
There are three notable exceptions to the large body of evidence documenting the adverse impact of the metabolic syndrome. One is a study by Bruno et al. [51] conducted in 1,565 elderly diabetic subjects from the Italian town of Casale Monferrato, who were followed for a median of 8 years. At baseline, the prevalence of the metabolic syndrome was 76%, and those with the syndrome had HRs for all-cause and CVD mortality that were no different from those of subjects without the syndrome. With frank diabetes of long duration, the incremental risk attributable, for example, to raised triglycerides or low HDL, is likely to be ‘swamped’ by the presence of diabetes itself [48]. The fact that subjects were, on average, much older (mean age at baseline: 69 years) than in virtually all other studies, and that hypertension was highly prevalent in the cohort may have masked the detrimental effects of the syndrome. Another study was conducted in non-diabetic American Indians [52] and showed a non-significant HR for risk of CVD in those with the syndrome. The small number of events that occurred during the follow-up period, as well as several other factors reviewed by the authors, could have contributed to their borderline results. Finally, the presence of the metabolic syndrome in a cohort of women with suspected CVD who had no angiographically significant coronary artery disease did not result in an increased 4-year risk of CVD, whereas the presence of the syndrome resulted in significantly higher risk in those who were angiographically positive [53].
Three studies have examined whether the difference in prevalence between the two definitions affects the predictive power for subsequent development of CVD [33, 37, 49]. Two of these found the ATPIII definition to be a slightly better predictor of all-cause and cardiovascular mortality [49] or CHD [37], whereas one [33] showed that the WHO definition more consistently predicted CVD and all-cause mortality. The fact that all three studies made modifications to one or both of the definitions, and that they included populations with dissimilar baseline characteristics, precludes drawing any conclusion as to which definition is superior.
Nonetheless, individuals with metabolic syndrome, however defined, have a much higher CVD risk than subjects without the syndrome. This conclusion is not surprising, since the individual components of the syndrome have long been known to be major cardiovascular risk factors [54–59]. Thus when they occur simultaneously, it is logical that adverse outcomes should be more likely [60–62].
ATPIII uses the term metabolic syndrome to imply that certain risk factors are associated with each other, and that insulin resistance is the primary cause [27, 28]. They identify six components of the metabolic syndrome as ‘underlying’, ‘major’ and ‘emerging’ CVD risk factors [28]. However, some risk factors associated with insulin resistance in each of those categories are not included in the definition of the syndrome. For example, physical inactivity is omitted as an underlying risk factor, while obesity is included. Family history, sex and age are major CVD risk factors that do not enter into the definition, but hypertension is included. Some emerging risk factors associated with insulin resistance, e.g. certain proinflammatory and prothrombotic markers, are not included, but elevated triglycerides and glucose intolerance are. Interestingly, although the latter four were designated ‘metabolic risk factors’ and a ‘component of the metabolic syndrome’, only elevated triglycerides and glucose intolerance are included in the official list of components [28]. The lack of any standardised methodology or rationale for how the definition was constructed, or can be modified, hampers its optimisation and utility.
It is not known whether the substitution or addition of any other well-known, conventional CVD risk factor(s) would improve the predictive value of the syndrome. In studies demonstrating that metabolic syndrome was associated with higher CVD risk [33, 38–50], this excess risk remained after adjustment for other conventional risk factors. This would suggest that if other risk factors are included in the definition, the predictive value of the syndrome may improve. However, we found no study that examined the impact of substituting another CVD risk factor for one already included in the definition. The issue of whether the risk factors act synergistically has also not been analysed.
Conversely, there are many studies suggesting that relatively new indices related to both insulin resistance and CVD may also be useful predictive tools (or useful additions to the syndrome definition). Since it is now well accepted that inflammation plays a major role in atherogenesis [63], it is not surprising that markers of inflammation might be used to predict CVD events. One such marker, C-reactive protein (CRP), has been studied in great detail, and has been found to be an independent CVD risk factor [64–68] and an independent marker of insulin resistance [69–72]. Three large population studies examined the relationship between CRP, the metabolic syndrome, and incident cardiovascular events [68, 73, 74]. In all three, CRP was a strong independent predictor of events, and its predictive value was equal to that of the metabolic syndrome. In the two studies [68, 73] that dichotomised CRP levels (above and below 3.0 mg/dl), the age-adjusted relative risk of future events was no different in subjects with high CRP but without the metabolic syndrome than it was in subjects with low CRP and with the metabolic syndrome. However, in subjects with high CRP levels plus the metabolic syndrome, the relative risk of events virtually doubled from that found with either parameter alone, indicating that CRP might be a valuable addition to the definition of the syndrome.
Rutter et al. [74] also found that CRP and the metabolic syndrome were independent risk factors, but in contrast to the two other reports, combining CRP and metabolic syndrome did not improve the predictive value of either used alone. Reilly et al. [75] also found that CRP did not add significantly to the metabolic syndrome, but their study did not include CVD outcomes. It is unclear why some studies show great value when CRP is added, while others do not. The discrepant results have not, however, deterred some investigators from advocating that CRP be included in the definition of the metabolic syndrome [76].
There is also an association between other markers of inflammation and insulin resistance/hyperinsulinaemia [70, 72], as well as inflammation and obesity [77–79], leading some investigators to conclude that inflammation is integrally related to the components of the metabolic syndrome [76, 77, 80]. CRP is also strongly associated with adipose-derived cytokines including interleukin-6 and tumor necrosis factor α [81], and is more likely to be elevated in obese insulin-resistant, but not obese insulin-sensitive, subjects [71]. Because obesity (particularly in the visceral compartment) is associated with insulin resistance, and these adipose-derived inflammatory markers have been linked to dyslipidaemia, hypertension and insulin action [70, 72], there is a heightened interest in markers from adipose tissue that are predictive of CVD [81].
One such marker is adiponectin. It is now well established that there is a strong and consistent inverse association between adiponectin and both insulin resistance and inflammation [70, 82, 83]. In addition, adiponectin is also inversely associated with other CVD risk factors such as blood pressure, LDL-cholesterol and triglycerides [84, 85]. Moreover, several studies have shown adiponectin to be a strong (inverse) independent risk factor for CVD [86–89].
Several other molecules have also been found to be closely associated with insulin resistance, metabolic syndrome risk factors and the risk of CVD. These include plasminogen activator inhibitor [90–92] and fibrinogen [91–94].
All told, therefore, many candidate markers could be included in the metabolic syndrome. In combination with other markers related to CVD and insulin resistance, more research may lead to a clearer understanding of the aetiology of the syndrome and hence to a definition that has strong (or stronger) CVD predictive value.
Some investigators have compared the predictive value of the metabolic syndrome with that of the Framingham risk prediction model. For example, a recent post hoc analysis of the placebo-treated groups in the 4S and AFCAPS/TexCAPS trials [47] showed that the increased event rate in subjects with the metabolic syndrome remained significant after adjustment for the Framingham 10-year risk score (which uses both dichotomised and continuous variables), suggesting that the syndrome carries risk not captured by Framingham risk scoring. It should be noted, however, that this analysis omitted diabetes (or any other measure of glucose intolerance) from the metabolic syndrome definition, thereby requiring patients to meet three of the remaining four factors to qualify and removing what may be a critical criterion from the definition. This modification may have biased their findings. Also, they dichotomised the Framingham score (i.e. >20% risk vs ≤20%), so a precise determination of the predictive ability of Framingham vs metabolic syndrome could not be determined.
In contrast, Wilson and colleagues [95], using data from Framingham found no advantage in risk assessment above the Framingham algorithm (i.e. age, sex, smoking, blood pressure, total cholesterol, diabetes, HDL-cholesterol) when some of the unique metabolic syndrome factors (obesity, triglycerides) were added or substituted. Further, when taking an elevated blood glucose level out of the metabolic syndrome definition, the 10-year risk for CHD did not achieve the threshold for ATPIII’s CHD risk equivalent [27], suggesting that glucose intolerance is a critical component contributing to the predictive power of the syndrome. Additional evidence for the greater predictive value of the Framingham scoring system has been provided by Stern and co-workers [96] in a study of the Hispanic and non-Hispanic whites who participated in the San Antonio Heart Study (n=2,570 ), were free of diabetes and CVD at baseline, and were followed for 7 to 8 years. Their analysis showed that the Framingham score had significantly higher sensitivity for predicting events than the presence of the metabolic syndrome, and when used in combination, the predictive value did not improve. In a multivariate analysis for predicting CVD, using a model that incorporated the Framingham equation and the metabolic syndrome, the former had an HR of 7.9 (95% CI, 5.3–11.7) compared with 1.5 (95% CI, 1.0–2.2) for the latter, confirming the superiority of the Framingham equation. Expressed differently, they found that the metabolic syndrome predicts CVD with a sensitivity of 55% and a false positive rate of 22%, whereas the Framingham risk score had a significantly higher sensitivity (69%) when the false positive rate was held to 22%. In another study that compared receiver operating characteristic curves (which denote the effectiveness of an assay or test), the metabolic syndrome provided identical risk prediction to that achieved by the Framingham score [34].
In the studies reviewed so far, a person was diagnosed with the metabolic syndrome if he or she had any three or more of the five criteria (see textbox Definitions of the metabolic syndrome). (To meet the WHO definition, three of five possible criteria must be present, one of which is mandatory.) Thus, there are 16 possible combinations that will meet the ATPIII definition of the syndrome and 11 for the WHO definition. Do all these combinations portend the same CVD risk?
This question has not yet been answered, and may never be, since an extremely large population that includes sufficient numbers of people with each combination of criteria would have to be followed for many years. There are studies, however, that partially address this issue. Malik et al. [48] reported that, compared with individuals with no risk factors, those with one or two syndrome factors had an HR=2.1 for CHD mortality and an HR=3.5 if they had the full syndrome (i.e. 3–5 risk factors). Other investigators [34, 73, 97] also found that the risk for CVD increased with the number of factors present.
Other studies, using multivariate analysis, have shown that the individual risk factors comprising the syndrome each carried a different odds ratio for predicting either prevalent CHD, incident CHD, or CVD mortality. In addition to hyperglycaemia, low HDL-cholesterol and hypertension usually conferred a significantly greater risk compared with the presence of obesity or high triglycerides [44, 49], although McNeill et al. [34] found that only an elevated blood pressure and low HDL-cholesterol were significantly associated with CHD. Golden et al. [98] assessed carotid intimal medial thickness (IMT) related to 57 combinations of six factors related to insulin resistance. In their analysis, 29 of the 57 groupings were associated with excess carotid IMT. The difference in excess IMT between individuals with two, three or four factors was minimal, but those with five or six factors showed an appreciable increase in excess IMT. Hypertension and hypertriglyceridaemia were the two factors that most contributed to the excess IMT. Taken together, these studies suggest that not all combinations that lead to the diagnosis of the syndrome convey equal risk, although the actual hierarchy of risk predictability for each of the syndrome combinations remains unknown.
The studies also illustrate another likely shortcoming of the current approach to diagnosing the syndrome. Both the ATPIII and WHO definitions weigh each risk component equally, yet it is clear that some risk factors included in the definition have greater CVD predictive importance than others. This fact is highlighted in other algorithms used to predict CVD risk using regression coefficients to assign different weights to risk factors [95, 99], and it is apparent from studies that examined the risk of CVD in persons with one or two components of the syndrome vs three or more [97]. For example, the disproportionate impact of glucose intolerance (IFG/IGT/diabetes) in the syndrome definition was demonstrated by Malik et al. [48] in their study on NHANES II participants. They observed that diabetes alone conveyed a much greater risk of CHD (HR=5), CVD (HR=3.6) or overall mortality (HR=2.1) than the presence of the metabolic syndrome (HR=3.5, 2.7, 1.5, respectively) according to definitions that included subjects with and without IFG/IGT/diabetes. Adding pre-existing CVD to diabetes was an even more powerful predictor of mortality (HR=11.3, 7.9, 2.9, respectively) over the 13-year follow-up period. Similarly, Stern et al. [100] showed that, among patients with prevalent CVD, the excess risk for all-cause and CVD mortality associated with the metabolic syndrome was entirely driven by the inclusion of diabetes in the definition, and once diabetes was controlled for, the presence of the metabolic syndrome no longer conferred excess risk. Finally, Hunt et al. [49] also showed that the presence of impaired fasting glucose (>6.1 mmol/l) alone was a stronger predictor of CVD or all-cause mortality in a general population than either the syndrome as a whole or any of its individual components. These reports raise the question of why glucose intolerance (particularly diabetes) is included in the definition of the metabolic syndrome, since it appears to account for most, if not all, of the CVD predictive value.
Since the metabolic syndrome does not include all known CVD risk factors, it should convey risk independently of other conventional risk factors (e.g. LDL, age, smoking, family history); however the proportion of the global CVD risk captured by the syndrome is unknown. It would be invaluable to know, from a list of all known CVD risk factors, the hierarchy of combinations with the highest predictive value. Then, a true comparison between the metabolic syndrome, other models using different risk factors [72, 73], or perhaps some new combination would tell us what is the best CVD predictive model.
Another, important question is the degree to which the presence of the syndrome in itself adds to CVD prediction beyond the contribution of the component risk factors. In other words, is the whole greater than the sum of its parts? If the syndrome conveyed no additional risk beyond its components then clinicians would have little reason to treat cases of the syndrome rather than addressing each risk factor as it was identified. At least five studies address this issue. One is the study by Golden et al. [98] reviewed above, which examined all possible combinations of six factors related to insulin resistance. Individuals with any four, five or six component groupings had no greater excess IMT than the sum of the same factors taken separately. The cross-sectional studies by Alexander et al. [44] and Yarnell et al. [101] showed that the impact of the syndrome on CVD was greatly attenuated in a multivariate analysis by controlling for certain of its components, thereby suggesting that the whole is not greater than its parts. Also, in a prospective study of diabetic and non-diabetic subjects free of CVD and followed for an average of 11 years, the risk of incident CHD associated with the syndrome was no greater than that explained by the presence of its components [34]. Finally, in the secondary analysis of the prospective West of Scotland Coronary Prevention Study, Satter et al. [73] showed that the metabolic syndrome was not a significant predictor of coronary heart disease when adjusted for its component factors in a multivariate model. Thus, these studies suggest that the syndrome itself conveys no greater information than the sum of its component risk factors.
In summary, if the metabolic syndrome is a multi-component risk factor for CVD, the components of which may be more or less strongly linked with insulin resistance, neither the ATPIII nor the WHO definitions consider the many other similarly related CVD risk factors, such as age, physical activity or history of CVD events. Some of these and other risk factors are included in the Framingham algorithm [95], which has been shown to be in general a more powerful tool for predicting future CVD events. However, even the Framingham risk equation does not include important CVD risk factors (e.g. previous CVD events, family history), and has been shown to be much less useful than other risk equations in predicting future CVD events in people with diabetes [102–104]. Other newly identified CVD risk factors have been shown to be strongly associated with insulin resistance and CVD, but it is unclear if they should be added to the syndrome and given equal or greater weight than the current components.
Because the criteria for the syndrome will capture individuals with frank disease (e.g. diabetes, hypertension, proteinuria, clinical cardiovascular disease), as well as with far milder forms of the same conditions, it is likely that there is a risk gradient for CVD events among people with the syndrome. Thus, the definition will capture a spectrum of severities, and it is highly likely that a person who satisfies the diagnostic criteria with risk factor levels just over the cutpoint will have a much lower CVD risk than another individual with the same combination but higher risk factor levels. This problem stands in contrast to the Framingham [95] and UKPDS [99] risk models, in which the spectrum of severities is weighted, so that it is clear who may be at greater or lesser risk.
Finally, people with diabetes and clinical cardiovascular disease should be excluded from the definition of metabolic syndrome since their inclusion provides no additional clinically useful information to guide treatment beyond current guidelines.
Does the syndrome reflect a single underlying pathological process?
When the concept of the syndrome was first proposed, insulin resistance and/or hyperinsulinaemia were initially thought to be the primary aetiological process, since most subjects with the syndrome had one or the other abnormality. Consistent with the primary adverse outcome of the metabolic syndrome (CVD), many studies [11, 105–116], but not all [117–119], have shown that insulin resistance or hyperinsulinaemia is a CVD risk factor. Although the reasons for the disparity in results have been debated elsewhere [120–122], it is important to note that the measurement of insulin itself, whether to determine the presence of insulin resistance or hyperinsulinaemia, is fraught with errors and inconsistency [123, 124] and the methods used vary considerably from laboratory to laboratory. Thus, many subjects who are insulin-resistant or hyperinsulinaemic in one institution may not be classified as such when tested in another setting, because the measurement of insulin is not standardised. Moreover, insulin-mediated glucose disposal varies six to eight-fold in apparently healthy, non-diabetic men [125], absolute insulin concentrations vary widely [16, 126], and there is no absolute criterion with which to classify individuals as being insulin-resistant or insulin-sensitive.
Very recently, however, Stern et al. [35] analysed the results of insulin clamp measurements in a large number of non-diabetic and diabetic subjects. An analysis of the distribution of insulin-mediated glucose disposal as measured by the clamp, showed evidence of bimodality and the optimal cutpoint classified 33% of non-diabetic subjects and 93% of diabetic subjects as insulin-resistant. Some investigators have chosen a cutpoint based on the relationship between insulin resistance and the frequency of adverse outcomes; however their sample sizes were very small [114, 127].
Although many non-diabetic adult subjects with a wide range of age and body mass are hyperinsulinaemic and insulin-resistant (∼50%), about 25% are insulin-resistant but without hyperinsulinaemia, and the same proportion are hyperinsulinaemic but without insulin resistance [126]. The relationship between insulin resistance and hyperinsulinaemia, reviewed in detail by Ferrannini and Balkau [126], is complex, and although both parameters will capture individuals with the metabolic syndrome, each makes an independent contribution to the clinical findings associated with the syndrome [128, 129]. Thus, hyperinsulinaemia and insulin resistance each identify partially different groups of individuals, they each cluster with various CVD risk factors, and individuals with the metabolic syndrome may have either, both, or none of these ‘insulin-related’ abnormalities.
Even though most people who have the metabolic syndrome are insulin-resistant as discussed earlier, that is probably due to the fact that almost all people with an elevated blood glucose value (the most prevalent characteristic among those with the syndrome) are insulin-resistant. Conversely, many studies have shown that only a minority of non-diabetic individuals with insulin resistance (but who may have IFG or IGT) will have the metabolic syndrome. In a study of 260 non-diabetic, overweight/obese individuals, McLaughlin et al. [130] found that 78% of those with metabolic syndrome were insulin-resistant, but only 48% with insulin resistance had metabolic syndrome. Liao et al. [131] reported that 39% of 74 overweight/obese non-diabetic adults were insulin-resistant, and 31% with insulin resistance met ATPIII criteria. Moreover, the ATPIII-negative/insulin-resistant individuals had CVD risk factor profiles that were significantly worse than the ATPIII-negative/insulin-sensitive group, implying that many presumably high-risk individuals will be not be identified by screening for metabolic syndrome. Also, they found that the sensitivity, specificity and positive predictive value for predicting insulin resistance in non-diabetic individuals with three or more metabolic syndrome traits were 20, 92 and 50%, respectively, denoting poor clinical utility. Cheal et al. [132] determined that 16% of 443 healthy, non-diabetic subjects were insulin-resistant and/or positive for metabolic syndrome, with a sensitivity, specificity and positive predictive value for metabolic syndrome as predictor of insulin resistance of 46, 93 and 76%, respectively. This study also showed that very few of the possible three, four or five factor combinations occurred in the non-diabetic patients classified with the syndrome.
As noted above, most investigators use the phrase ‘insulin resistance’ to describe the hallmark of the metabolic syndrome, even though insulin resistance or hyperinsulinaemia may not be present in subjects with the syndrome. Furthermore, the extent to which an elevated risk of CVD is due to insulin resistance itself, vs isolated hyperinsulinaemia, vs some other related factor is still unclear. Some investigators turn to studies on the relationship of insulin resistance to the aetiology of atherosclerosis [133–136], and to the underlying aetiology of type 2 diabetes [137], as evidence that insulin resistance is the more important abnormality [138]. Unfortunately, we could find no study that has compared insulin resistance, as measured by sensitive and specific methods (e.g. euglycaemic insulin clamp), with fasting insulin levels to determine which variable is a better predictor of cardiovascular events in non-diabetic individuals.
Perhaps most important is the fact that the multitude of reports relating insulin resistance to any risk factor or CVD are all association studies. It may well be that there is a more basic defect that can result in insulin resistance and/or other CVD risk factors. The uncertainty surrounding the causative factor(s) that give rise to the syndrome has prompted many investigators to perform a ‘factor analysis’ as an approach towards understanding the fundamental cause of the clustering. Factor analysis is a multivariate correlation method that seeks to explain the relationship between a set of observed variables (in this case the clinical features of the metabolic syndrome) and a smaller set of unknown underlying variables (e.g. the aetiology) termed ‘factors’. The factors ideally represent unique, independent domains that have not been directly measured but give rise to the observed variables. Thus an array of CVD risk variables, occurring more than would be expected by chance [25], may be related to one underlying factor, thereby supporting a single unifying aetiology; or they may be related to two or more factors suggesting a relationship between the underlying domains. Alternatively, more than one underlying factor suggests a more complex aetiology, and often the analysis cannot account for all the variability, thereby providing additional evidence that more than one physiological process underlies the expression of the observed correlations.
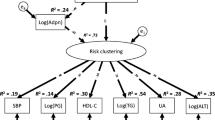
Although factor analysis was developed some time ago [139–141], there is no standardised methodology, and its use and interpretation is often problematic [142]. Nonetheless, factor analysis is an intriguing exploratory, somewhat subjective and qualitative approach towards understanding the root cause(s) of the metabolic syndrome. The results of many of these studies are shown in Table 1.
As shown, all of the studies [24, 33, 38, 143–149] found that at least two and usually three or four factors underlie the overall correlation between risk variables, even though different factor analysis methods were used. Shen and colleagues [150] using ‘confirmatory factor analysis’, which provides some advantages and is complementary to exploratory factor analysis, proposed a correlated four factor model that nicely depicts the major factors related to the syndrome (Fig. 1). Thus, it is clear that more than one distinct pathophysiological process underlies the clinical expression of the syndrome, but insulin resistance/hyperinsulinaemia appears related in some fashion to most. Also, and equally important, all the factors can account for no more than about two-thirds of the total variance observed in the clustering, suggesting that the syndrome may be even more complex than that inferred by factor analyses. These studies, therefore, again call into question the appropriateness of implying that a handful of CVD risk factors have a common underlying pathophysiology.
Factor structure of the metabolic syndrome. Adapted with permission from Shen et al. [150]
To the extent that insulin resistance/hyperinsulinaemia is itself a cardiovascular risk factor, many investigators have sought to identify ways to better and more simply identify persons with insulin resistance apart from the diagnosis of the metabolic syndrome. Indeed, easier, simpler and at least equally effective ways are now available to identify insulin-resistant subjects. Laws and Reaven [151] showed that a high triglyceride and low HDL-cholesterol concentration is a strong indicator of insulin resistance, and when expressed as a ratio [130], the optimal cutpoints in overweight/obese individuals resulted in a sensitivity, specificity and positive predictive value for insulin resistance of 64, 68, 67%, respectively. The addition of extra measurements (i.e. blood glucose, blood pressure, BMI) was less sensitive (52%) but more specific (85%) in predicting insulin resistance. Thus, both the metabolic syndrome and this abbreviated index have only a moderate likelihood of identifying the person with insulin resistance. More recently , Stern and colleagues [35] collected euglycaemic clamp (the gold standard for measuring insulin resistance) data from over 2,000 lean and overweight/obese individuals and used a decision tree classification scheme to develop decision rules for identifying insulin-resistant individuals based on common clinical measurements. In their study, decision rules based on either HOMA-IR and BMI, or BMI and family history of diabetes had sensitivities and specificities in the range of 80%. Thus, if the aim is to identify insulin resistance in either lean or overweight/obese subjects, there are simpler ways to do so than by identifying those with the metabolic syndrome.
In summary, the attempt to define the metabolic syndrome as the result of a single (or even major) unifying pathophysiological process, e.g. insulin resistance, is problematic. Although insulin resistance or hyperinsulinaemia is clearly an important feature of the syndrome, many other as yet unidentified factors are also important. Insulin resistance may simply be one of many abnormalities linked to a more fundamental, truly unifying pathophysiology. Moreover, the definition of the syndrome includes risk factors that are only weakly related to insulin resistance or hyperinsulinaemia (e.g. blood pressure) and excludes others that are closely related (e.g. CRP, adiponectin). Finally, although many clinical values are significantly associated with insulin resistance/hyperinsulinaemia, the strength of their association (which has not exceeded a correlation coefficient of 0.7 and is usually 0.3–0.6) is not particularly impressive.
Although the studies reviewed above question the hypothesis that insulin resistance/hyperinsulinaemia is the major underlying pathological process, it must be remembered that the clustering of CVD risk factors has been well documented and thus it is likely (but not assured) that there is some underlying aetiology. It may be that insulin resistance is simply a risk factor not unlike other metabolic syndrome components, and that the underlying aetiology for some of the syndrome factors is related to abnormalities in visceral adipose tissue [152] or an altered inflammatory state; other factors may be associated with the cluster because they relate indirectly to one of its components.
Treatment of patients who have the metabolic syndrome
When a person is identified as having the metabolic syndrome, it is not always clear what should be the treatment of choice or the goals of therapy. In a post-hoc analysis of the Scandinavian Simvastatin Survival Study, Pyorala et al. [153] found that simvastatin reduced CVD events to the same degree in non-diabetic patients with or without the metabolic syndrome. We found no other controlled trial results examining the value of a specific pharmacological therapy on patients with the metabolic syndrome. It should again be remembered that the current definitions of the syndrome capture many people with frank disease (e.g. diabetes, hypertension, clinical CVD), as well as those who have milder conditions or ‘normal’ values that, while qualifying them for the diagnosis of the syndrome, are not high enough to warrant specific therapy. Thus, it is important to distinguish the approach and value of treating various metabolic syndrome combinations. However, no studies have examined the value of tailoring the treatment algorithm to the particular combination of criteria that resulted in the diagnosis of the syndrome.
Many of the syndrome characteristics are acknowledged to be closely related to insulin resistance or hyperinsulinaemia and their correlates. Yet at the same time it is unknown whether treating ‘insulin resistance’ itself would be of value in preventing CVD in all, or a subset, of metabolic syndrome patients. Although some studies suggest that the newer insulin-sensitising agents (i.e. thiazolidinediones) improve glycaemic control, reduce CVD risk factors and generally result in a beneficial CVD profile [154–161], at the time of writing no controlled studies have shown that thiazolidinediones reduce CVD events even in the setting of diabetes, although one major trial that will help address this issue will be reported shortly (i.e. the PROactive study); studies using metformin are equivocal. Since thiazolidinediones affect a wide variety of parameters, even favourable trial results will not prove that reducing insulin resistance itself is the critical factor. Moreover, even if positive trial evidence were to emerge relatively soon, other important issues have been identified [162], such as how will insulin resistance be measured, what is the cutpoint to begin treatment, and is the target population only patients similar to those included in the trials? Thus, our knowledge base is such that we cannot yet contemplate drug treatment for insulin resistance, let alone the metabolic syndrome.
Other modifiers of insulin resistance include weight reduction and exercise, and they have been identified as key elements in the treatment of the metabolic syndrome [27, 31, 32]. But they are also key elements in the treatment of all components of the syndrome when they occur in isolation [27, 163, 164]. Clinicians, therefore, should neither rely on, nor require a diagnosis of metabolic syndrome to prescribe and encourage what is now a fundamental tenet of medicine—weight maintenance (or reduction), exercise and a healthy meal plan.
The conundrum of treating the metabolic syndrome is illustrated in the case studies shown in Table 2. Patient A is obese and has severe hypertension; the likely treatment is lifestyle modification (exercise and weight loss) to include smoking cessation counselling, an antihypertensive drug, and aspirin. Patient B is obese, has pre-hypertension and pre-diabetes; his likely treatment is aggressive lifestyle modification and aspirin. Patient C is also obese, has diabetes and a history of acute myocardial infarction; his treatment would also be aggressive lifestyle modification with perhaps a glucose-lowering agent and aspirin.
Does it matter that only Patient B has the metabolic syndrome (by ATPIII criteria)? If that were immediately apparent, would the treatment change? Who will have the next CVD event, patient A, B or C? We submit that at this time, the diagnosis of the metabolic syndrome itself, or the lack of it adds virtually nothing to the treatment of one or more CVD risk factors in a given patient. If, however, it were known that insulin resistance causes cardiovascular disease and there was a sensitive way to measure and treat it, or if we knew the relative risk of CVD among various configurations of risk factors, or if we knew that a combination of risk variables found at borderline disease levels elevates one’s CVD risk considerably—then knowing that a patient has the metabolic syndrome could also be useful. But at present, none of those criteria has been fulfilled.
These case studies also raise additional concerns. For patients with type 2 diabetes, a comprehensive set of evidence-based prevention services should be provided (e.g. regular eye and foot exams). In patients who are diagnosed with the metabolic syndrome and who also have diabetes, the importance and treatment of the diabetes may, in contrast to what might be expected, take a backseat relative to the syndrome, with patients or their doctors possibly neglecting or overlooking essential factors of diabetes management. Also, for patients who fail to meet the necessary number of criteria to diagnose the syndrome (e.g. only two of the factors are present), the absence of the syndrome may divert attention away from addressing risk factors that are present.
Further research needed
In the preceding Sections, we identified many unanswered questions related to the metabolic syndrome. Many are unresolved fundamental issues that raise considerable doubt about the construct itself.
All this should prompt an aggressive research agenda, and based on what is highlighted by this review as missing, should give pause to those in medical practice or in industry. Importantly, there have yet to be any controlled randomised trials, or systematic, prospective, longitudinal studies that carefully document the clinical value of treating a disease label now being given out to a huge number of our population.
At the very least, we suggest that the following are urgently needed:
-
1.
A critical analysis of how the syndrome is defined. Are all risk factors equally important? Do some combinations (of two, three or four factors) portend greater CVD risk than others?
-
2.
A definition of the syndrome, in which variables have defined lower and upper cutpoints or that uses continuous variables in a multivariate score system (e.g. Framingham/UKPDS risk engine).
-
3.
An evidence-based analysis assessing the rationale and value of adding (or replacing) other CVD risk factors (e.g. age, CRP, family history, a direct measure of insulin resistance) to the definition.
-
4.
An assessment of CVD risk in subjects with combinations of intermediate phenotypes only (e.g. IFG/IGT, mildly elevated triglycerides, blood pressure 120–140 mmHg) and who have, or don’t have, insulin resistance or hyperinsulinaemia.
-
5.
An aggressive research agenda to identify the underlying cause(s) of the CVD risk factor clustering.
Conclusion
As a construct that denotes risk factor clustering, the metabolic syndrome has been a useful paradigm. That is, it draws attention to the fact that some CVD risk factors tend to cluster in patients so pre-disposed. The teaching point implied by the term, and explicitly stated by ATPIII, is that the identification of one of the risk variables in a patient should prompt a search for others.
At the time these relationships were first documented, the advent of a phrase to capture the prevalence of the clustering was probably a helpful reminder to clinicians, and certainly served to open a wide avenue of research into its aetiology and impact. Now, however, it has taken on meaning and import greater than is justified by our current knowledge. Indeed, only recently the International Diabetes Federation developed yet a new definition which suggests that the key element is central obesity [165]. In their paper, however, no data were reviewed indicating the impact or benefit derived from their new definition.
As shown in this review (see textbox Summary of concerns regarding the metabolic syndrome), there is much fundamental, clinically important and critically missing information about the metabolic syndrome to warrant a more serious examination of whether medical science is doing any good by drawing attention to [166] and labelling millions of people [36] with a presumed disease that does not stand on firm ground. In particular, patients with diabetes or clinical CVD should be excluded from the case definition of metabolic syndrome as they provide no additional understanding of risk or treatment recommendations that are otherwise not currently recommended.
Summary of concerns regarding the metabolic syndrome 1. Criteria are ambiguous or incomplete. Rationale for thresholds are ill-defined. 2. Value of including diabetes in the definition is questionable. 3. Insulin resistance as the unifying aetiology is uncertain. 4. No clear basis for including/excluding other CVD risk factors. 5. CVD risk value is variable and dependent on the specific risk factors present. 6. The CVD risk associated with the ‘syndrome’ appears to be no greater than the sum of its parts. 7. Treatment of the syndrome is no different than the treatment for each of its components. 8. The medical value of diagnosing the syndrome is unclear. |
Medical science usually defines a syndrome as an ‘aggregate of symptoms and signs associated with any morbid process, and constituting together the picture of the disease’ [167]. The specific signs and symptoms are usually caused by a unifying underlying pathology, and their combination confers a risk that is different from the sum of the parts. In almost every way—from the term itself, to the underlying pathophysiology, to the variables included or excluded, to the value of making the diagnosis, and finally to its treatment—the metabolic syndrome requires much more study before its designation as a ‘syndrome’ is truly warranted, and before its clinical utility is adequately defined. We hope this reappraisal gives pause to the growing use of the term, and also stimulates urgently needed research.
Consequently, in addition to the research suggested above, our recommendations to clinicians are:
-
1.
Adults with any major CVD risk factor should be evaluated for the presence of other CVD risk factors.
-
2.
Patients with CVD risk variables above the cutpoint for normal should receive counselling for life-style modification, and at cutpoints indicative of frank disease (e.g. blood pressure >140/90, fasting plasma glucose >7.0 mmol/l), treatment should correspond to established guidelines [27, 163, 168].
-
3.
Providers should avoid labelling patients with the term ‘metabolic syndrome’, as this might create the impression that the metabolic syndrome denotes a greater risk than its components, or that it is more serious than other CVD risk factors, or that the underlying pathophysiology is clear.
-
4.
All CVD risk factors should be individually and aggressively treated.
-
5.
Until randomised controlled trials have been completed, there is no appropriate pharmacological treatment for the metabolic syndrome, nor should it be assumed that pharmacological therapy to reduce insulin resistance will be beneficial to patients with the metabolic syndrome.
Duality of Interest
None of the authors has any conflict of interest with regard to the information contained in this manuscript.
Abbreviations
- ATPIII:
-
Adult Treatment Panel III
- CVD:
-
Cardiovascular disease
- CRP:
-
C-reactive protein
- HOMA-IR:
-
homeostasis model assessment of insulin resistance
- HR:
-
Hazard ratio
- IMT:
-
Intimal medial thickness
- NHANES:
-
National Health and Nutrition Examination Survey
- UKPDS:
-
UK Prospective Diabetes Study
- WHO:
-
World Health Organization
References
Avogaro P, Crepaldi G, Enzi G, Tiengo A (1967) Associazione di iperlidemia, diabete mellito e obesita di medio grado. Acto Diabetol Lat 4:36–41
Haller H (1977) Epidemiology and associated risk factors of hyperlipoproteinemia. Z Gesamte Inn Med 32:124–128
Singer P (1977) Diagnosis of primary hyperlipoproteinemias. Z Gesamte Inn Med 32:129–133
Himsworth H (1936) Diabetes mellitus: a differentiation into insulin-sensitive and insulin-insensitive types. Lancet 1:127–130
Shen S-W, Reaven GM, Farquhar JW (1970) Comparison of impedance to insulin mediated glucose uptake in normal and diabetic subjects. J Clin Invest 49:2151–2160
Ginsburg H, Kimmerling G, Olefsky JM, Reaven GM (1975) Demonstration of insulin resistance in untreated adult onset diabetic subjects with fasting hyperglycemia. J Clin Invest 55:454–461
Reaven GM, Lerner R, Stern M, Farquhar JW (1967) Role of insulin in endogenous hypertriglyceridemia. J Clin Invest 46:1756–1767
Olefsky JM, Farquhar JW, Reaven GM (1974) Reappraisal of the role of insulin in hypertriglyceridemia. Am J Med 57:551–560
Orchard TJ, Becker DJ, Bates M, Kuller LH, Drash AL (1983) Plasma insulin and lipoprotein concentrations: an atherogenic association? Am J Epidemiol 118:326–337
Olefsky JM, Kolterman OG, Scarlett JA (1982) Insulin action and resistance in obesity and non-insulin-dependent type II diabetes mellitus. Am J Physiol 243:E15–E30
Stern MP, Haffner SM (1986) Body fat distribution and hyperinsulinemia as risk factors for diabetes and cardiovascular disease. Arteriosclerosis 6:123–130
Modan M, Halkin H, Almog S et al (1985) Hyperinsulinemia: a link between hypertension, obesity, and glucose intolerance. J Clin Invest 75:809–817
Haffner SM, Fong D, Hazuda HP, Pugh JA, Patterson JK (1988) Hyperinsulinemia, upper body adiposity, and cardiovascular risk factors in non-diabetics. Metabolism 37:338–345
Ferrannini E, Buzzigoli G, Bonadonna R et al (1987) Insulin resistance in essential hypertension. N Engl J Med 317:350–357
Wingard DL, Barrett-Connor E, Criqui MH, Suarez L (1983) Clustering of heart disease risk factors in diabetic compared to non-diabetic adults. Am J Epidemiol 117:19–26
Reaven GM (1988) Role of insulin resistance in human disease. Diabetes 37:1595–1607
Zavaroni I, Bonora E, Pagliara M et al (1989) Risk factors for coronary artery disease in healthy persons with hyperinsulinemia and normal glucose tolerance. N Engl J Med 320:502–506
Ferrannini E, Haffner SM, Mitchell BD, Stern MP (1991) Hyperinsulinaemia: the key feature of a cardiovascular and metabolic syndrome. Diabetologia 34:416–422
Haffner SM, Valdez RA, Hazuda HP, Mitchell BD, Morales PA, Stern MP (1992) Prospective analysis of the insulin-resistance syndrome (syndrome X). Diabetes 41:715–722
Mykkanen L, Kuusisto J, Pyorala K, Laakso M (1993) Cardiovascular disease risk factors as predictors of type 2 (non-insulin-dependent) diabetes mellitus in elderly subjects. Diabetologia 36:553–559
Schmidt MI, Duncan BB, Watson RL, Sharrett AR, Brancati FL, Heiss G A (1996) metabolic syndrome in whites and African-Americans. The Atherosclerosis Risk in Communities baseline study. Diabetes Care 19:414–418
Laakso M, Sarlund H, Mykkanen L (1990) Insulin resistance is associated with lipid and lipoprotein abnormalities in subjects with varying degrees of glucose tolerance. Arteriosclerosis 10:223–231
Schmidt MI, Watson RL, Duncan BB et al (1996) Clustering of dyslipidemia, hyperuricemia, diabetes, and hypertension and its association with fasting insulin and central and overall obesity in a general population. Atherosclerosis Risk in Communities Study Investigators. Metabolism 45:699–706
Meigs JB, D’Agostino RB, Wilson PWF, Cupples LA, Nathan DM, Singer DE (1997) Risk variable clustering in the insulin resistance syndrome: the Framingham Offspring Study. Diabetes 46:1594–1600
Liese AD , Mayer-Davis EJ, Haffner SM (1998) Development of the multiple metabolic syndrome: an epidemiologic perspective. Epidemiol Rev 20:157–172
World Health Organization (1999) Definition, diagnosis, and classification of diabetes mellitus and its complications: report of a WHO consultation. World Health Organization, Geneva
Expert panel on the detection, evaluation, and treatment of high blood cholesterol in adults (2001) Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA 285:2486–2497
Grundy SM, Brewer HB, Cleeman JI, Smith SC, Lenfant C (2004) Definition of metabolic syndrome: report of the National Heart, Lung, and Blood Institute/American Heart Association conference on scientific issues related to definition. Circulation 109:433–438
Balkau B, Charles MA, Drivsholm T et al (2002) Frequency of the WHO metabolic syndrome in European cohorts, and an alternative definition of an insulin resistance syndrome. European Group for the Study of Insulin Resistance (EGIR). Diabetes Metab 28:364–376
Einhorn D, Reaven GM, Cobin RH et al (2003) American College of Endocrinology position statement on the insulin resistance syndrome. Endocr Pract 9:237–252
Wilson PWF, Grundy SM (2003) The metabolic syndrome. Practical guide to origins and treatment: Part I. Circulation 108:1422–1425
Grundy SM, Hansen B, Smith SC Jr, Cleeman JI, Kahn RA (2004) Clinical management of metabolic syndrome. Circulation 109:551–556
Lakka HM, Laaksonen DE, Lakka TA et al (2002) The metabolic syndrome and total and cardiovascular disease mortality in middle-aged men. JAMA 288:2709–2716
McNeill AM, Rosamond WD, Girman CJ et al (2005) The metabolic syndrome and 11-year risk of incident cardiovascular disease in the atherosclerosis risk in communities study. Diabetes Care 28:385–390
Stern SE, Williams K, Ferrannini E, Defronzo RA, Bogardus C, Stern MP (2005) Identification of individuals with insulin resistance using routine clinical measurements. Diabetes 54:333–339
Ford ES, Giles WH (2003) A comparison of the prevalence of the metabolic syndrome using two proposed definitions. Diabetes Care 26:575–581
Meigs JB, Wilson PW, Nathan DM, D’Agostino RB Sr, Williams K, Haffner SM (2003) Prevalence and characteristics of the metabolic syndrome in the San Antonio Heart and Framingham Offspring Studies. Diabetes 52:2160–2167
Lempiainen P, Mykkanen L, Pyorala K, Laakso M, Kuusisto J (1999) Insulin resistance syndrome predicts coronary heart disease events in elderly non-diabetic men. Circulation 100:123–128
Kekalainen P, Sarlund H, Pyorala K, Laakso M (1999) Hyperinsulinemia cluster predicts the development of type 2 diabetes independent of a family history of diabetes. Diabetes Care 22:86–92
Pyorala M, Miettinen H, Halonen P, Laakso M, Pyorala K (2000) Insulin resistance syndrome predicts the risk of coronary heart disease and stroke in healthy middle-aged men: the 22-year follow-up results of the Helsinki Policemen Study. Arterioscler Thromb Vasc Biol 20:538–544
Kuusisto J, Lempiainen P, Mykkanen L, Laakso M (2001) Insulin resistance syndrome predicts coronary heart disease events in elderly type 2 diabetic men. Diabetes Care 24:1629–1633
Isomaa B, Almgren P, Tuomi T et al (2001) Cardiovascular morbidity and mortality associated with the metabolic syndrome. Diabetes Care 24:683–689
Onat A, Ceyhan K, Basar O, Erer B, Tobrak S, Sansoy V (2002) Metabolic syndrome: major impact on coronary risk in a population with low cholesterol levels—a prospective and cross-sectional evaluation. Atherosclerosis 165:285–292
Alexander CM, Landsman PB, Teutsch SM, Haffner SM (2003) NCEP metabolic syndrome, diabetes mellitus and prevalence of coronary heart disease. Diabetes 52:1210–1214
Katzmarzyk PT, Church TS, Blair SN (2004) Cardiorespiratory fitness attenuates the effects of the metabolic syndrome on all-cause and cardiovascular disease mortality in men. Arch Intern Med 164:1092–1097
Ford ES (2004) The metabolic syndrome and mortality from cardiovascular disease and all-causes: findings from the National Health and Nutrition Examination Survey II Mortality Study. Atherosclerosis 173:309–314
Girman CJ, Rhodes T, Mercuri M et al (2004) The metabolic syndrome and risk of major coronary events in the Scandinavian Simvastatin Survival Study (4S) and the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS). Am J Cardiol 93:136–141
Malik S, Wong ND, Franklin SS et al (2004) Impact of the metabolic syndrome on mortality from coronary heart disease, cardiovascular disease and on all causes in United States adults. Circulation 110:1245–1250
Hunt KJ, Resendez RG, Williams K, Haffner SM, Stern MP (2004) National Cholesterol Education Program versus World Health Organization metabolic syndrome in relation to all-cause and cardiovascular mortality in the San Antonio Heart Study. Circulation 110:1251–1257
Scuteri A, Najjar S, Morrell C, Lakatta E (2005) The metabolic syndrome in older individuals: prevalence and prediction of cardiovascular events. Diabetes Care 28:882–887
Bruno G, Merletti F, Biggeri A et al (2004) Metabolic syndrome as a predictor of all-cause and cardiovascular mortality in type 2 diabetes: the Casale Monferrato Study. Diabetes Care 27:2689–2694
Resnick HE, Jones K, Ruotolo G et al (2003) Insulin resistance, the metabolic syndrome, and risk of incident cardiovascular disease in non-diabetic American Indians: the Strong Heart Study. Diabetes Care 26:861–867
Marroquin OC, Kip KE, Kelley DE et al (2004) Metabolic syndrome modifies the cardiovascular risk associated with angiographic coronary artery disease in women: a report from the Women’s Ischemia Syndrome Evaluation. Circulation 109:714–721
Fuller JH , Shipley MJ, Rose G, Jarrett RJ, Keen H (1980) Coronary heart disease risk and impaired glucose tolerance: the Whitehall Study. Lancet 1:1373–1376
Lippel K, Tyroler H, Eder H, Gotto A, Vahouny G (1981) Relationship of hypertriglyceridemia to atherosclerosis. Atherosclerosis 1:406–417
Miller GJ, Miller NE (1975) Plasma high density lipoprotein concentration and development of ischemic heart disease. Lancet 1:16–19
Keys A, Aravanis C, Blackburn H et al (1972) Coronary heart disease: overweight and obesity as risk factors. Ann Intern Med 77:15–27
Deckert T, Feldt-Rasmussen B, Jensen T, Kofoed-Enevoldsen A (1989) Albuminuria reflects widespread vascular damage: the Steno hypothesis. Diabetologia 32:219–226
Lewington S, Clarke R, Qizilbash N, Peto R, Collins R (2002) Age-specific relevance of usual blood pressure to vascular mortality: a meta-analysis of individual data for one million adults in 61 prospective studies. Prospective Studies Collaboration. Lancet 360:1903–1913
Kannel WB, McGee DL (1979) Diabetes and cardiovascular risk factors in the Framingham study. Circulation 59:8–13
Rosengren A, Welin L, Tsipigianni A, Wilhelmsen L (1989) Impact of cardiovascular risk factors on coronary heart disease and mortality among middle-aged diabetic men: a general population study. Br Med J 299:1127–1131
Stamler J, Vaccaro O , Neaton JD, Wentworth D (1993) Diabetes, other risk factors, and 12-year cardiovascular mortality for men screened in the Multiple Risk Factor Intervention Trial. Diabetes Care 16:434–444
Ross R(1999) Atherosclerosis: an inflammatory disease. N Engl J Med 340:115–126
Danesh J, Collins R, Appleby P, Peto R (1998) Association of fibrinogen, C-reactive protein, albumin, or leukocyte count with coronary heart disease: meta-analyses of prospective studies. JAMA 279:1477–1482
Ridker PM, Hennekens CH, Buring JE, Rifai N (2000) C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med 342:836–843
Ridker PM (2003) Clinical applications of C-reactive protein for cardiovascular disease detection and prevention. Circulation 107:363–369
Ridker PM, Cannon CP, Morrow D et al (2005) C-reactive protein levels and outcomes after statin therapy. Pravastatin or Atorvastatin Evaluation and Infection Therapy—Thrombolysis in Myocardial Infarction 22 (PROVE IT-TIMI 22). N Engl J Med 352:20–28
Ridker PM, Buring JE, Cook NR, Rifai N (2003) C-reactive protein, the metabolic syndrome, and risk of incident cardiovascular events: an 8-year follow-up of 14,719 initially healthy American women. Circulation 107:391–397
Pradhan AD, Cook NR, Buring JE, Manson JE, Ridker PM (2003) C-reactive protein is independently associated with fasting insulin in nondiabetic women. Arterioscler Thromb Vasc Biol 23:650–655
Yudkin JS, Stehouwer CD, Emeis JJ, Coppack SW (1999) C-reactive protein in healthy subjects: associations with obesity, insulin resistance, and endothelial dysfunction: a potential role for cytokines originating from adipose tissue? Arterioscler Thromb Vasc Biol 19:972–978
McLaughlin T, Abbasi F, Lamendola C et al (2002) Differentiation between obesity and insulin resistance in the association with C-reactive protein. Circulation 106:2908–2912
Festa A, D’Agostino R Jr, Howard G, Mykkanen L, Tracy RP, Haffner SM (2000) Chronic subclinical inflammation as part of the insulin resistance syndrome: the Insulin Resistance Atherosclerosis Study (IRAS). Circulation 102:42–47
Sattar N, Gaw A, Scherbakova O et al (2003) Metabolic syndrome with and without C-reactive protein as a predictor of coronary heart disease and diabetes in the West of Scotland Coronary Prevention Study. Circulation 108:414–419
Rutter MK, Meigs JB, Sullivan LM, D’Agostino RB Sr, Wilson PW (2004) C-reactive protein, the metabolic syndrome, and prediction of cardiovascular events in the Framingham Offspring Study. Circulation 110:380–385
Reilly MP, Wolfe ML, Rhodes T, Girman C, Mehta N, Rader DJ (2004) Measures of insulin resistance add incremental value to the clinical diagnosis of metabolic syndrome in association with coronary atherosclerosis. Circulation 110:803–809
Ridker PM, Wilson PW, Grundy SM (2004) Should C-reactive protein be added to metabolic syndrome and to assessment of global cardiovascular risk? Circulation 109:2818–2825
Yudkin JS, Kumari M, Humphries SE, Mohamed-Ali V (2000) Inflammation, obesity, stress and coronary heart disease: is interleukin-6 the link? Atherosclerosis 148:209–214
Xu H, Barnes GT, Yang Q et al (2003) Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest 112:1821–1830
Wellen KE, Hotamisligil GS (2003) Obesity-induced inflammatory changes in adipose tissue. J Clin Invest 112:1785–1788
Pearson TA, Mensah GA, Alexander RW et al (2003) Markers of inflammation and cardiovascular disease: application to clinical and public health practice: a statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association. Circulation 107:499–511
Kershaw EE, Flier JS (2004) Adipose tissue as an endocrine organ. J Clin Endocrinol Metab 89:2548–2556
Weyer C, Funahashi T, Tanaka S et al (2001) Hypoadiponectinemia in obesity and type 2 diabetes: close association with insulin resistance and hyperinsulinemia. J Clin Endocrinol Metab 86:1930–1935
Chandran M, Phillips SA, Ciaraldi T, Henry RR (2003) Adiponectin: more than just another fat cell hormone? Diabetes Care 26:2442–2450
Matsubara M, Maruoka S, Katayose S (2002) Decreased plasma adiponectin concentrations in women with dyslipidemia. J Clin Endocrinol Metab 87:2764–2769
Kazumi T, Kawaguchi A, Sakai K, Hirano T, Yoshino G (2002) Young men with high-normal blood pressure have lower serum adiponectin, smaller LDL size, and higher elevated heart rate than those with optimal blood pressure. Diabetes Care 25:971–976
Kumada M, Kihara S, Sumitsuji S et al (2003) Association of hypoadiponectinemia with coronary artery disease in men. Osaka CAD Study Group. Coronary artery disease. Arterioscler Thromb Vasc Biol 23:85–89
Kojima S, Funahashi T, Sakamoto T et al (2003) The variation of plasma concentrations of a novel, adipocyte derived protein, adiponectin, in patients with acute myocardial infarction. Heart 89:667–672
Zoccali C, Mallamaci F, Tripepi G et al (2002) Adiponectin, metabolic risk factors, and cardiovascular events among patients with end-stage renal disease. J Am Soc Nephrol 13:134–141
Pischon T, Girman CJ, Hotamisligil GS, Rifai N, Hu FB, Rimm EB (2004) Plasma adiponectin levels and risk of myocardial infarction in men. JAMA 291:1730–1737
Potter van Loon BJ, Kluft C, Radder JK, Blankenstein MA, Meinders AE (1993) The cardiovascular risk factor plasminogen activator inhibitor type 1 is related to insulin resistance. Metabolism 42:945–949
Haffner SM, D’Agostino R Jr, Mykkanen L et al (1999) Insulin sensitivity in subjects with type 2 diabetes. Relationship to cardiovascular risk factors: the Insulin Resistance Atherosclerosis Study. Diabetes Care 22:562–568
Festa A, D’Agostino R Jr, Mykkanen L et al (1999) Relative contribution of insulin and its precursors to fibrinogen and PAI-1 in a large population with different states of glucose tolerance. The Insulin Resistance Atherosclerosis Study (IRAS). Arterioscler Thromb Vasc Biol 19:562–568
Ernst E, Resch KL (1993) Fibrinogen as a cardiovascular risk factor: a meta-analysis and review of the literature. Ann Intern Med 18:956–963
Imperatore G, Riccardi G, Iovine C, Rivellese AA, Vaccaro O (1998) Plasma fibrinogen: a new factor of the metabolic syndrome. A population-based study. Diabetes Care 21:649–654
Wilson PW, D’Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB (1998) Prediction of coronary heart disease using risk factor categories. Circulation 97:1837–1847
Stern MP, Williams K, Gonzalez-Villalpando C, Hunt KJ, Haffner SM (2004) Does the metabolic syndrome improve identification of individuals at risk of type 2 diabetes and/or cardiovascular disease? Diabetes Care 27:2676–2681 (‘Erratum’ in Diabetes Care 28:238, 2005)
Klein BE, Klein R, Lee KE (2002) Components of the metabolic syndrome and risk of cardiovascular disease and diabetes in beaver dam. Diabetes Care 25:1790–1794
Golden SH, Folsom AR, Coresh J, Sharrett AR, Szklo M, Brancati F (2002) Risk factor groupings related to insulin resistance and their synergistic effects on subclinical atherosclerosis: the atherosclerosis risk in communities study. Diabetes 51:3069–3076
Stevens RJ, Kothari V, Adler AI, Stratton IM (2001) The UKPDS risk engine: a model for the risk of coronary heart disease in Type II diabetes (UKPDS 56). United Kingdom Prospective Diabetes Study (UKPDS) Group. Clin Sci 101:671–679
Stern MP, Williams K, Hunt KJ (2005) Impact on diabetes/metabolic syndrome in patients with established cardiovascular disease. Atheroscler Suppl 6:3–6
Yarnell JWG, Patterson CC, Bainton D, Sweetnam PM (1998) Is metabolic syndrome a discrete entity in the general population? Evidence from the Caerphilly and Speedwell population studies. Heart 79:248–252
Yeo WW, Yeo KR (2001) Predicting CHD risk in patients with diabetes mellitus. Diabet Med 18:341–344
Song SH, Brown PM (2004) Coronary heart disease risk assessment in diabetes mellitus: comparison of UKPDS risk engine with Framingham risk assessment function and its clinical implications. Diabet Med 21:238–245
McEwan P, Williams JE, Griffiths JD et al (2004) Evaluating the performance of the Framingham risk equations in a population with diabetes. Diabet Med 21:318–323
Pyorala K (1979) Relationship of glucose tolerance and plasma insulin to the incidence of coronary heart disease: results from two population studies in Finland. Diabetes Care 2:131–141
Welborn TA, Wearne K (1979) Coronary heart disease incidence and cardiovascular mortality in Busselton with reference to glucose and insulin concentrations. Diabetes Care 2:154–160
Fontbonne A, Charles MA, Thibult N et al (1991) Hyperinsulinaemia as a predictor of coronary heart disease mortality in a healthy population: the Paris Prospective Study, 15-year follow-up. Diabetologia 34:356–361
Moller LF, Jespersen J (1995) Fasting serum insulin levels and coronary heart disease in a Danish cohort: 17-year follow-up. J Cardiovasc Risk 2:235–240
Perry IJ, Wannamethee SG, Whincup PH, Shaper AG, Walker MK, Alberti KG (1996) Serum insulin and incident coronary heart disease in middle-aged British men. Am J Epidemiol 144:224–234
Despres JP, Lamarche B, Mauriege P et al (1996) Hyperinsulinemia as an independent risk factor for ischemic heart disease. N Engl J Med 334:952–957
Pyorala M, Miettinen H, Laakso M, Pyorala K (1998) Hyperinsulinemia predicts coronary heart disease risk in healthy middle-aged men: the 22-year follow-up results of the Helsinki Policemen Study. Circulation 98:398–404
Ducimetiere P, Eschwege E, Papoz L, Richard JL, Claude JR, Rosselin G (1980) Relationship of plasma insulin levels to the incidence of myocardial infarction and coronary heart disease mortality in a middle-aged population. Diabetologia 19:205–210
Folsom AR, Szklo M, Stevens J, Liao F, Smith R, Eckfeldt JH (1997) A prospective study of coronary heart disease in relation to fasting insulin, glucose, and diabetes. The Atherosclerosis Risk in Communities (ARIC) Study. Diabetes Care 20:935–942
Yip J, Facchini FS, Reaven GM (1998) Resistance to insulin-mediated glucose disposal as a predictor of cardiovascular disease. J Clin Endocrinol Metab 83: 2773–2776
Uusitupa MI, Niskanen LK, Siitonen O, Voutilainen E, Pyorala K (1990) 5-year incidence of atherosclerotic vascular disease in relation to general risk factors, insulin level, and abnormalities in lipoprotein composition in non-insulin dependent diabetic and non-diabetic subjects. Circulation 82:27–36
Hu G, Qiao Q, Tuomilehto J, Eliasson M, Feskens EJ, Pyorala K (2004) Plasma insulin and cardiovascular mortality in non-diabetic European men and women: a meta-analysis of data from eleven prospective studies. DECODE Insulin Study. Diabetologia 47:1245–1256
Welin L, Eriksson H, Larsson B, Ohlson LO, Svardsudd K, Tibblin K (1992) Hyperinsulinaemia is not a major coronary risk factor in elderly men. The study of men born in 1913. Diabetologia 35:766–770
Orchard TJ, Eichner J, Kuller LH, Becker DJ, McCallum LM, Grandits GA (1994) Insulin as a predictor of coronary heart disease: interaction with apolipoprotein E phenotype. A report from the Multiple Risk Factor Intervention Trial. Ann Epidemiol 4:40–45
Ferrara A, Barrett-Connor EL, Edelstein SL (1994) Hyperinsulinemia does not increase the risk of fatal cardiovascular disease in elderly men or women without diabetes: the Rancho Bernardo Study, 1984–1991. Am J Epidemiol 140:857–869
Stern MP (1994) The insulin resistance syndrome: the controversy is dead, long live the controversy! Diabetologia 37:956–958
Wingard DL, Barrett-Connor EL, Ferrara A (1995) Is insulin really a heart disease risk factor? Diabetes Care 18:1299–1304
McKeigue P, Davey G (1995) Associations between insulin levels and cardiovascular disease are confounded by comorbidity. Diabetes Care 18:1294–1298
Robbins DC, Andersen L, Bowsher R et al (1996) Report of the American Diabetes Association’s Task Force on standardization of the insulin assay. Diabetes 45:242–256
Wallace TM, Levy JC, Matthews DR (2004) Use and abuse of HOMA modeling. Diabetes Care 27:1487–1495
Yeni-Komshian H, Carantoni M, Abbasi F, Reaven GM (2000) Relationship between several surrogate estimates of insulin resistance and quantification of insulin-mediated glucose disposal in 490 healthy non-diabetic volunteers. Diabetes Care 23:171–175
Ferrannini E, Balkau B (2002) Insulin: in search of a syndrome. Diabet Med 19:724–729
Facchini FS, Hua N, Abbasi F, Reaven GM (2001) Insulin resistance as a predictor of age-related diseases. J Clin Endocrinol Metab 86:3574–3578
Mykkanen L, Haffner SM, Ronnemaa T, Bergman RN, Laakso M (1997) Low insulin sensitivity is associated with clustering of cardiovascular disease risk factors. Am J Epidemiol 146:315–321
Weyer C, Hanson RL, Tataranni PA, Bogardus C, Pratley RE (2000) A high fasting plasma insulin concentration predicts type 2 diabetes independent of insulin resistance: evidence for a pathogenic role of relative hyperinsulinemia. Diabetes 49:2094–2101
McLaughlin T, Abbasi F, Cheal K, Chu J, Lamendola C, Reaven G (2003) Use of metabolic markers to identify overweight individuals who are insulin-resistant. Ann Intern Med 139:802–809
Liao Y, Kwon S, Shaughnessy S et al (2004) Critical evaluation of adult treatment panel III criteria in identifying insulin resistance with dyslipidemia. Diabetes Care 27:978–983
Cheal KL, Abbasi F, Lamendola C, McLaughlin T, Reaven GM, Ford ES (2004) Relationship to insulin resistance of the adult treatment panel III diagnostic criteria for identification of the metabolic syndrome. Diabetes 53:1195–1200
Laakso M, Sarlund H, Salonen R et al (1991) Asymptomatic atherosclerosis and insulin resistance. Arterioscler Thromb 11:1068–1076
Agewall S, Fagerberg B, Attvall S, Wendelhag I, Urbanavicius V, Wikstrand J (1995) Carotid artery wall intima–media thickness is associated with insulin-mediated glucose disposal in men at high and low coronary risk. Stroke 26:956–960
Howard G, O’Leary DH, Zaccaro D et al (1996) Insulin sensitivity and atherosclerosis. The Insulin Resistance Atherosclerosis Study (IRAS) Investigators. Circulation 93:1809–1817
Haffner SM, Mykkanen L, Festa A, Burke JP, Stern MP (2000) Insulin-resistant prediabetic subjects have more atherogenic risk factors than insulin-sensitive prediabetic subjects: implications for preventing coronary heart disease during the prediabetic state. Circulation 101:975–980
DeFronzo RA (1988) Lilly lecture 1987. The triumvirate: beta-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 37:667–687
Reaven GM (1995) Pathophysiology of insulin resistance in human disease. Physiol Rev 75:473–486
Mardia KV, Kent JT, Bibby JM (1979) Factor analysis. In: Mardia KV, Kent JT, Bibby JM (eds) Multivariate analysis. Academic, London pp 255–280
Cureton EE, D’Agostino RB (1986) Factor analysis: an applied approach. Lawrence Erlbaum Associates, Hillsdale NJ
Krzanowski WJ (1994) Explaining observed associations: latent-variable models. In: Krzanowski WJ (ed) Principles of multivariate statistical methods. A primer. Chapman and Hall, London, pp 93–106
Lawlor DA, Ebrahim S, May M, Davey Smith G (2004) (Mis)use of factor analysis in the study of insulin resistance syndrome. Am J Epidemiol 159:1013–1018
Gray RS, Fabsitz RR, Cowan LD, Lee ET, Howard BV, Savage PJ (1998) Risk factor clustering in the insulin resistance syndrome. The Strong Heart Study. Am J Epidemiol 148:869–878
Lehto S, Ronnemaa T, Pyorala K, Laakso M (2000) Cardiovascular risk factors clustering with endogenous hyperinsulinaemia predict death from coronary heart disease in patients with Type II diabetes. Diabetologia 43:148–155
Sakkinen PA, Wahl P, Cushman M, Lewis MR, Tracy RP (2000) Clustering of procoagulation, inflammation, and fibrinolysis variables with metabolic factors in insulin resistance syndrome. Am J Epidemiol 152:897–907
Maison P, Byrne CD, Hales CN, Day NE, Wareham NJ (2001) Do different dimensions of the metabolic syndrome change together over time? Evidence supporting obesity as the central feature. Diabetes Care 24:1758–1763
Hanley AJ, Karter AJ, Festa A et al (2002) Factor analysis of metabolic syndrome using directly measured insulin sensitivity: the insulin resistance atherosclerosis study. Diabetes 51:2642–2647
Wang JJ, Qiao Q, Miettinen ME, Lappalainen J, Hu G, Tuomilehto J (2004) The metabolic syndrome defined by factor analysis and incident type 2 diabetes in a Chinese population with high postprandial glucose. Diabetes Care 27:2429–2437
Ford ES (2003) Factor analysis and defining the metabolic syndrome. Ethn Dis 13:429–437
Shen BJ, Todaro JF, Niaura R et al (2003) Are metabolic risk factors one unified syndrome? Modeling the structure of the metabolic syndrome X. Am J Epidemiol 157:701–711
Laws A, Reaven GM (1992) Evidence for an independent relationship between insulin resistance and fasting plasma HDL-cholesterol, triglyceride and insulin concentrations. J Intern Med 231:25–30
Carr DB, Utzschneider KM, Hull RL et al (2004) Intra-abdominal fat is a major determinant of the National Cholesterol Education Program Adult Treatment Panel III criteria for the metabolic syndrome. Diabetes 53:2087–2094
Pyorala K, Ballantyne CM, Gumbiner B et al (2004) Reduction of cardiovascular events by simvastatin in non-diabetic coronary heart disease patients with and without the metabolic syndrome: subgroup analyses of the Scandinavian Simvastatin Survival Study (4S). Diabetes Care 27:1735–1740
UKPDS Group (1998) Effect of intensive blood-glucose control with metformin on complications in overweight patients with type 2 diabetes (UKPDS 34). UK Prospective Diabetes Study (UKPDS) Group. Lancet 352:854–865
Knowler WC, Barrett-Connor E, Fowler SE et al (2002) Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. Diabetes Prevention Program Research Group. N Engl J Med 346:393–403
Shadid S, Jensen MD (2003) Effects of pioglitazone versus diet and exercise on metabolic health and fat distribution in upper body obesity. Diabetes Care 26:3148–3152
Lebovitz HE, Dole JF, Patwardhan R, Rappaport EB, Freed MI (2001) Rosiglitazone monotherapy is effective in patients with type 2 diabetes. Rosiglitazone Clinical Trials Study Group. J Clin Endocrinol Metab 86:280–288
Aronoff S, Rosenblatt S, Braithwaite S, Egan JW, Mathisen AL, Schneider RL (2000) Pioglitazone hydrochloride monotherapy improves glycemic control in the treatment of patients with type 2 diabetes: a 6-month randomized placebo-controlled dose-response study. The pioglitazone 001 study group. Diabetes Care 23:1605–1611
Freed MI, Ratner R, Marcovina SM et al (2002) Effects of rosiglitazone alone and in combination with atorvastatin on the metabolic abnormalities in type 2 diabetes mellitus. Rosiglitazone study 108. Am J Cardiol 90:947–952
Haffner SM, Greenberg AS, Weston WM, Chen H, Williams K, Freed MI (2002) Effect of rosiglitazone treatment on non-traditional markers of cardiovascular disease in patients with type 2 diabetes mellitus. Circulation 106:679–684
Satoh N, Ogawa Y, Usui T et al (2003) Anti-atherogenic effect of pioglitazone in type 2 diabetic patients irrespective of the responsiveness to its antidiabetic effect. Diabetes Care 26:2493–2499
Davidson MB (2003) Is treatment of insulin resistance beneficial independent of glycemia? Diabetes Care 26:3184–3186
Chobanian AV, Bakris GL, Black HR et al (2003) Seventh report of the joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Hypertension 42:1206–1252
Smith SC Jr, Jackson R, Pearson TA et al (2004) Principles for national and regional guidelines on cardiovascular disease prevention: a scientific statement from the world heart and stroke forum. Circulation 109:3112–3121
International Diabetes Federation. The IDF consensus worldwide definition of the metabolic syndrome. http://www.idf.org (accessed 2 June 2005)
Washington Post (2005) New diagnosis for overweight. Major risk factors add up to ‘metabolic syndrome’. Washington Post 8 February 2005: Sect. A, p1
No authors listed (2000) Stedman’s medical dictionary, 27th edition. Lippincott, Williams and Wilkins, Baltimore, pp 1746
American Diabetes Association (2005) Standards of medical care in diabetes. Diabetes Care 28:S4–S36
Author information
Authors and Affiliations
Corresponding author
Additional information
This statement was reviewed and approved by the Professional Practice Committee of the American Diabetes Association and by an ad hoc committee of the European Association for the Study of Diabetes (members: H. Beck-Nielsen [Odense, Denmark], K. Borch-Johnsen [Gentofte, Denmark], E. A. M. Gale [Bristol, UK], L. Groop [Malmö, Sweden], H.-U. Häring [Tübingen, Germany], R. J. Heine [Amsterdam, The Netherlands], and D. R. Matthews [Oxford, UK]).
Simultaneous publication: This article is being simultaneously published in 2005 in Diabetes Care and Diabetologia by the American Diabetes Association and the European Association for the Study of Diabetes.
Copyright 2005 by the American Diabetes Association, Inc. and Springer-Verlag. Copying with attribution allowed for any noncommercial use of the work.
Rights and permissions
About this article
Cite this article
Kahn, R., Buse, J., Ferrannini, E. et al. The metabolic syndrome: time for a critical appraisal. Diabetologia 48, 1684–1699 (2005). https://doi.org/10.1007/s00125-005-1876-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-005-1876-2