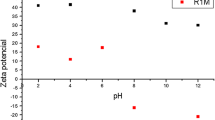
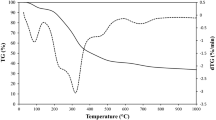
Abstract
An overview of the use of ash wastes from a combined heat and power (CHP) plant as secondary resources in various areas of industry and agriculture is given. The physicochemical and sorption properties of ash and slag from the Apatity CHP plant with respect to the \({\text{NH}}_{4}^{ + }\) ion were studied. It was established that the process of sorption is adequately described by the Langmuir equation of monomolecular adsorption. The capacity of an adsorption monolayer of the ash and slag of 20.43 mg \({\text{NH}}_{4}^{ + }\)/g, which is consistent with the values noted in international practice, and the adsorption equilibrium constant (0.0632 L/g) were calculated. It was shown that the sorption process is most accurately described by a pseudo-second-order kinetic model, and it proceeds in a mixed-diffusion mode involving not only the outer surface of the material but also the inner surface of its particles due to their porosity.








Similar content being viewed by others
REFERENCES
Perlov, A.G., Tekhnologii ochistki prirodnykh vod (Natural Water Treatment Technologies), Moscow: Izd. ASV, 2016.
O’Shea, K.E. and Dionysiou, D.D., Phys. Chem. Lett., 2012, vol. 3, no. 15, p. 2112.
Chee Yang Teh, Pretty Mori Budiman, Katrina Pui, Yee Shak, and Ta Yeong Wu, Ind. Eng. Chem. Res., 2016, vol. 55, no. 16, p. 4363.
Miller, D.J., Dreyer, D.R., Bielawski, Ch.W., Paul, D.R., and Freeman, B.D., Angew. Chem. Int. Ed., 2017, vol. 56, no. 17, p. 4662.
Mohammed Boshir Ahmed, Zhou, J.L., Huu Hao Ngo, Wenshan Guo, Thomaidis, N.S., and Jiang Xu, J. Haz. Mat., 2017, vol. 323, p. 274.
Arami, M., Limaee, N.Y., and Mahmoodia, N.M., Chem. Eng. J., 2008, no. 139, p. 2.
Lee, S.M. and Tiwar, D., Appl. Clay Sci., 2012, nos. 59–60, p. 84.
Considine, R., Denoyel, R., Pendleton, P., Schumann, R., and Wong, S.H., Colloids Surf., A, 2001, no. 179, p. 271.
Su, F., Lu, C., and Hu, S., Colloids Surf., A, 2010, no. 353, p. 83.
Hao, R., Xiao, X., Zuo, X.X., Nan, J.M., and Zhang, W.D., J. Hazard. Mater., 2012, nos. 209–210, p. 137.
Hou, M.F., Ma, C.X., Zhang, W.D., Tang, X.Y., Fan, Y.N., and Wan, H.F., J. Hazard. Mater., 2011, no. 186, p. 1118.
Jafari, S., Aziziana, S., and Jalehb, B., Colloids Surf., A, 2011, no. 384, p. P. 618.
Yudovich, Ya.E. and Ketris, M.P., Toksichnye elementy-primesi v iskopaemykh uglyakh (Toxic Impurity Elements in Fossil Coals), Yekaterinburg: Izd. UrORAN, 2005.
Javadian, H., Khavarpour, M., Belviso, C., Taghavi, M., and Maghsudi, M., J. Cleaner Prod., 2019, no. 208, p. 1131. https://doi.org/10.1016/j.jclepro.2018.10.186
Xu, G. and Shi, X., Resour. Conserv. Recycling, 2018, no. 136, p. 95. https://doi.org/10.1016/j.resconrec.2018.04.010
Iyer, R.S. and Scott, J.A., Resour. Conserv. Recycling, 2001, no. 31(3), p. 217. https://doi.org/10.1016/S0921-3449(00)00084-7
Ding, J., Ma, S., Shen, S., Xie, Z., Zheng, S., and Zhang, Y., Waste Manage., 2017, no. 60, p. 375. https://doi.org/10.1016/j.wasman.2016.06.009
Sahoo, P.K., Kim, K., Powell, M.A., and Equeenuddin, S.M., Int. J. Coal Sci. Technol., 2016, no. 3(10–11), p. 1958. https://doi.org/10.1007/s40789-016-0141-2
Li, C. and Qiao, X., Chem. Eng. J., 2016, no. 302, p. 388. https://doi.org/10.1016/j.cej.2016.05.029
Ram, L.C. and Masto, R.E., Earth-Science Rev., 2014, no. 128, p. 52. https://doi.org/10.1016/j.earscirev.2013.10.003
Sabry, M.S., Peter, S.H., and Tsadilas, C.D., J. Environ. Manage., 2014, no. 145, p. 249. https://doi.org/10.1016/j.jenvman.2014.07.005
Mushtaq, F., Zahid, M., Bhatti, I.A., Nasir, S., and Hussain, T., J. Environ. Manage., 2019, no. 240, p. 27. https://doi.org/10.1016/j.jenvman.2019.03.054
Hower, J.C., Groppo, J.G., Graham, U.M., Ward, C.R., Kostova, I.J., Maroto-Valer, M.M., and Dai, S., Int. J. Coal Geol, 2017, no. 179, p. 11. https://doi.org/10.1016/j.coal.2017.05.007
Vassilev, S.V. and Vassileva, C.G., Energy Fuel, 2005, no. 19, p. 1084.
Visa, M., Isac, L., and Duta, A., Appl. Surface Sci., 2012, no. 258 (17), p. 6345.
Hermassi, M., Valderrama, C., Moreno, N., Font, O., Querol, X., Batis, N.H., and Cortina, J.L., J. Environ. Chem. Eng., 2017, no. 5 (1), p. 160. https://doi.org/10.1016/j.jece.2016.11.027
Wang, X., Lu, S., Gao, C., Xu, X., Zhang, X., Bai, X., Liu, M., and Wu, L., Chem. Eng. J., 2014, no. 252, p. 404. https://doi.org/10.1016/j.cej.2014.04.097
GOST (State Standard) 27753.3-88: Greenhouse Grounds. Method for Determination of Water Extract pH, 1988.
Ikonnikova, K.V., Ikonnikova, L.F., Minakova, T.S., and Sarkisov, Yu.S., Teoriya i praktika opredeleniya kislotno-osnovnykh svoistv poverkhnosti tverdykh tel metodom pH-metrii: Uchebnoe posobie (Theory and Practice of the Determination of the Surface Acid–Base Properties of Solids by pH-Metry: A Textbook), Tomsk: Izd. Tomsk. Politekhn. Univ., 2014.
Lagergren, S., Kung. Sven. Veten. Hand., 1898, vol. 24, p. 1.
Ho, Y.S., Ng, J.C.Y., and McKay, G., Separat. Purificat. Methods, 2000, no. 29(2), p. 189.
Wendling, L.A., Douglas, G.B., Coleman, S., and Yuan, Z., Sci. Total Environ., 2013, no. 442, p. 63. https://doi.org/10.1016/j.scitotenv.2012.10.008
ASTM Standard C618-08: Standard Specification for Coal Fly Ash and Raw or Calcined Natural Pozzolan for Use in Concrete, 2008.
Taleb, K., Markovski, J., Velickovic, Z., Rusmirovic, J., Rancic, M., Pavlovic, V., and Marinkovic, A., Arab. J. Chem., 2016, no. 12, p. 4675.
Thommes, M., Kaneko, K., Neimark, A.V., Olivier, J.P., Rodriguez-Reinoso, F., Rouquerol, J., and Sing, K.S.W., Pure Appl. Chem., 2015, vol. 87, nos. 9–10, p. 1051. https://doi.org/10.1515/pac-2014-1117
Yakovleva, N.V., Vopr. Materialoved., 2013, no. 1(73), p. 95.
Moreno, N., Querol, X., Andres, J.M., Stanton, K., Towler, M., Nugteren, H., Janssen-Jurkovicova, M., and Jones, R., Fuel, 2005, no. 84 (11), p. 1351. https://doi.org/10.1016/j.fuel.2004.06.038
Pereverzev, V.N., Korobeinikova, N.M., and Baskova, L.A., Agrokhimiya, 2009, no. 2, p. 35.
Boyd, G.E., Adamson, A.W., and Mayers, L.S., J. Am. Chem. Soc., 1949, vol. 67, no. 9, p. 2836.
Weber, W.J., Jr., Morris, J.C., and Sanit, J., J. Sanitary Eng. Div., 1963, no. 89, p. 31.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Makhlyarchuk
About this article
Cite this article
Gorbacheva, T.T., Maiorov, D.V. Sorption of Ammonium Ions from Aqueous Solutions on the Ash and Slag of a CHP Plant. Solid Fuel Chem. 56, 276–284 (2022). https://doi.org/10.3103/S0361521922040048
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0361521922040048