Abstract
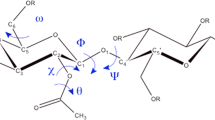
Quantum chemistry methods were used to calculate the energy parameters of an elementary unit and a cellulose macromolecule dimer (cellobiose), and structure simulation was performed and the energy of interaction between the fragments of native cellulose macromolecules was calculated. It was established that the trans conformation of cellobiose is more stable than the cis conformation by 6.7 kcal/mol. Differences in the calculated and real (according to literature data) IR spectra of cellulose were related to the presence of intramolecular and intermolecular hydrogen bonds in the native structure. It was shown that the interaction of individual fragments of cellulose macromolecules from eight monomer units is due to the manifestation of intramolecular hydrogen bonds. It was found that the energies of intermolecular interactions ∆Е essentially depend on the terminal groups X in the cellulose macromolecule fragments, and they are –26, 49, and ‒32 kcal/mol for X = –H, –COOH, and –COH, respectively. The structure of the interacting fragments of cellulose macromolecules can be regulated by replacing the hydrogen atoms of hydroxyl or terminal groups of the macromolecules with functional groups that do not form intramolecular hydrogen bonds and impede self-organization into fibrillar structures. It was shown that compounds with a high electron affinity or a negative energy of the lower vacant molecular orbital are the best reagents for complexation reactions with cellulose.





Similar content being viewed by others
REFERENCES
Bledzki, A.K. and Gassan, J., Progr. Polym. Sci., 1999, vol. 24, no. 2, p. 221.
Mohanty, A.K., Misra, M., and Hinrichsen, G., Macromolec. Mater. Eng., 2000, vol. 276, no. 1, p. 1.
Moon, R.J., Martini, A., Nairn, J., Simonsen, J., and Youngblood, J., Chem. Soc. Rev., 2011, vol. 40, no. 7, p. 3941.
Martin-Alfonso, J.E., Nunez, N., Valencia, C., Franco, J.M., and Diaz, M.J., J. Industr. Eng. Chem., 2011, no. 17, p. 818.
Sanchez, R., Franco, J.M., Delgado, M.A., Valencia, C., and Gallegos, C., Carbohydr. Polym., 2011, vol. 83, p. 151.
Nevell, T.P. and Zeronian, S.H., Cellulose Chemistry and Its Applications, New York: Wiley, 1985.
Battista, O.A., Industr. Eng. Chem., 1950, vol. 42, no. 3, p. 502.
Haensel, T., Reinmoller, M., Lorenz, P., Beenken, W.J.D., Krischok, S., and Syed Imad-Uddin, A., Cellulose, 2012, vol. 19, no. 3, p. 1005.
Akman, F., Cellulose Chem. Technol., 2017, vol. 51, nos. 3–4, p. 253.
Kocheva, L.S., Extended Abstract of Doctoral (Chem.) Dissertation, Arkhangel’sk: Arkh. State Techn. Univ., 2008.
Granovsky, A.A., GAMESS v.7.1. http://classic.chem.msu.su/gran/games/index.html
Ivanov-Omskii, V.I., Gerasyuta, S.M., and Ivanova, E.I., Izv. St.-Peterb. Lesotekhn. Akad., 2017, no. 218, p. 199.
Kačuráková, M. and Wilson, R.H., Carbohydrate Polym., 2001, vol. 44, no. 4, p. 291. https://doi.org/10.1016/S0144-8617(00)00245-9
Ali, M., et al., Polymer., 2001, vol. 42, no. 7, p. 2893.
Nugmanov, O.K., Grigor’eva, N.P., and Lebedev, N.A., Khim. Rastit. Syr’ya, 2013, no. 1, p. 40.
Langkilde, F.W. and Svantesson, A., J. Pharm. Biomed. Anal., 1995, vol. 13, nos. 4–5, p. 409.
Jiang, J.H., et al., Anal. Chem., 2002, vol. 74, no. 14, p. 3555.
Peng, B.L., Dhar, N., Liu, H.L., and Tam, K.C., Can. J. Chem. Eng., 2011, vol. 89, no. 5, p. 1191.
Eichhorn, S.J., et al., J. Mater. Sci., 2010, vol. 45, no. 1, p. 1.
Dufresne A., Curr. Opin. Colloid In. Sci., 2017, vol. 29, p. 1.
Dufresne, A., Current Opinion in Colloid & Interface Sci, 2017, vol. 29, p. 1.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Translated by V. Makhlyarchuk
About this article
Cite this article
Gyul’maliev, A.M., Safieva, R.Z., Vinokurov, V.A. et al. Structure Simulation and Calculation of the Energy of Interaction of the Fragments of Cellulose Macromolecules. Solid Fuel Chem. 53, 190–196 (2019). https://doi.org/10.3103/S0361521919030030
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3103/S0361521919030030