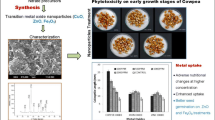
Abstract
Background
A pot experiment was conducted to evaluate the phytotoxic thresholds of zinc oxide (ZnO NPs), iron oxide (FeO NPs), copper (Cu NPs), and silver nanoparticles (Ag NPs) on cotton. All the nanoparticles were biosynthesized from Conocarpus erectus L. leaf extract. They were characterized by scanning electron microscope (SEM), X-ray diffraction (XRD), and Fourier transmission infrared spectroscopy (FTIR). Five concentrations (0, 25, 50, 75, and 100 ppm) of all the biosynthesized nanoparticles were foliar applied thrice over the duration of cotton growth. Five cotton seeds were sown in 5 kg soil pot and harvested after 6 weeks. SPAD values were evaluated before harvesting while physical growth parameters, physiological parameters biochemical, and oxidative stress parameters were measured after harvesting.
Results
The value of all parameters, except oxidative stress, increased by 13–47% in response to ZnO NPs application up to 100 ppm. However, phytotoxic threshold of iron oxide nanoparticles (FeO NPs) and copper nanoparticles (Cu NPs) was only 50 ppm as plant growth was increased by 7.9–24.3%. Above 50 ppm plant growth and biomass were decreased by 6.7–16.2%. Silver nanoparticles (Ag NPs) showed maximum growth (3.7 to 9.12% increased) at 25 ppm concentration. At higher concentrations than 25 ppm, the application of Ag NPs decreased the growth due to toxicity. Foliar application of different biosynthesized nanoparticles showed the different range of threshold value for cotton crop. Threshold value of iron oxide and copper nanoparticles for cotton was 50 ppm, whereas, it was 25 ppm for silver nanoparticles. Since the ZnO NPs application continued increasing the growth till its maximum concentration used i.e., 100 ppm, we cannot say that this is its threshold value.
Conclusion
On the basis of obtained results, it can be concluded that nanoparticles should be used within threshold to avoid adverse effects on crops. Application of nanoparticles within threshold optimum concentration, increased plant biomass and antioxidant system.
Graphical Abstract

Similar content being viewed by others
Introduction
Green nanotechnology is an emerging field nowadays, various applications of green nanoparticles are being introduced [1] especially in the field of agriculture as biofertilizers, biopesticides, and antimicrobial agents. Basic mechanism involved in the synthesis of green metallic nanoparticles is reduction of cations of precursor salt by means of capping agents produced by plants or microbes in the form of proteins, amino acids, carbohydrates, and other functional groups. Generally, there are various methods for the synthesis of nanoparticles, i.e., physical, chemical, and biological methods, whereby, in chemical methods, precursor salt is from chemical entity like sodium borohydride (NaBH4), hydrazine (N2H4), and sodium hypophosphite (NaPO2H2). Similarly, nanoparticles synthesized from physical methods are reduced by means of evaporation and condensation as reducing agent in physical method [2]. Both physical and chemical methods are either laborious or environmentally corrosive due to the use of toxic chemicals, auxiliaries, and fossil fuels.
Biological methods are environmentally friendly and safe for nanoparticle synthesis due to use of natural reducing agents [3]. The main concern while applying the nanoparticles in agriculture is their phytotoxicity [4]. In biological methods reducing agents are safe since these are mostly derived from plant, bacteria or fungus. For green synthesis, plant metabolites can be obtained from any part of plant, i.e., leaves, stem, fruit or any part of plant. These plant metabolites help in reduce precursor salt. Maximum nanoparticles yield can be obtained by plant mediated method as compared to other methods of nanoparticle synthesis [5].
Zinc is micronutrient and have narrow threshold for application. Excess amount of zinc can cause toxicity which leads to increase the competition for binding sites of ions and reduces the iron metal uptake followed by the decrease in plant yield. ZnO NPs promote antioxidant system of chickpea, tomato, java plant [6]. FeO NPs also promote antioxidants and physiological attributes of wheat [7, 8], water melon [9], and Darecocephalum plant [10]. Various studies showed toxic effects of FeO NPs on different crops when exceeded from its threshold [11]. [12] reported reduction in seed germination of cilantro (Coriandrum sativum L.) up to 50% of Cu NPs, whereas rice germination affected up to 7%. Cu NPs foliar application increase fruit size in tomato [13]. Cu NPs also alleviate heavy metal toxicity by inducing antioxidant system [14]. Cu NPs become potentially toxic when applied above threshold concentration [15]. Similarly, Ag NPs improved growth of common bean and maize. But like other nanoparticles Ag NPs also showed inhibitory effects on common bean and maize [16].
Recently, nanoparticles are being used in many fields. However, when nanoparticles designed for any application, their threshold become narrow due to their large surface area-to-volume ratio that’s why small number of nanoparticles can catalyze the whole reaction as compared to bulk materials which catalyze same reaction by huge quantity. The main reason behind this is bioavailability due to surface area-to-volume ratio [1]. But on other hand, nanoparticles possess narrow threshold and become toxic even on low concentration. The intensive literature study revealed that there exists a distinct research gap, that nanoparticles up regulate the plant metabolism [17]. The toxicity of nanoparticles hence becomes different for every crop, the threshold values for different crops are still undiscovered [18]. To address this research gap, this study proposed a novel method that solely works in such a way that it observes the nanoparticle threshold value for different types of biosynthesized nanoparticles. In this study, the main objective while using metallic nanoparticles is to determine the phytotoxicity threshold for cotton crop so that later on these threshold values of zinc oxide, iron oxide, copper, and silver can be used in cotton for different purpose of removing abiotic (salinity, drought, and temperature stress) and biotic (diseases and insect attack) stresses. Plant extract of various plants can be used as capping agent. Conocarpus erectus L. extract is widely used as reducing agent for nanoparticles synthesis because of its availability, non-toxicity, and rich in metabolites [5].
Materials and methods
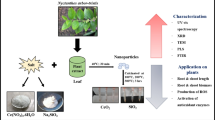
Biosynthesis of nanoparticles
Plant extract preparation
Fresh leaves of Conocarpus erectus L. were taken and washed with deionized water to remove dust and impurities. Then the leaves were shade dried and ground into fine powder. 5 g powder was suspended into 50 ml of deionized water and heated for 20 min at 75 °C and then stored at 4 °C after filtration with Whatman filter paper grade 1 having 9 cm diameter and 2.5 micron pore size [19].
Preparation of nanoparticles
Zinc oxide, iron oxide, copper, and silver nanoparticles were synthesized by zinc sulphate heptahydrate (ZnSO4.7H2O) (Sigma-Aldrich CAS number: 7446-20-0), iron sulphate heptahydrate (FeSO4.7H2O) (Sigma-Aldrich CAS number: 7782-63-0), copper sulphate pentahydrate (CuSO4.5H2O) (Sigma-Aldrich CAS number: 7758-99-8), and silver sulphate pentahydrate (AgSO4.5H2O) (Sigma-Aldrich CAS number: 10294-26-5), respectively. 20 ml plant extract was dispensed with 80 ml of 1 mM precursor salt solution shown in Fig. 1. Then solution was stirred for 15 min and then sonicated at 60 °C for 1 h and centrifuged at 10,000 rpm for 10 min.
Characterization of nanoparticles
Nanoparticles suspension was centrifuged at 12,000 rpm. Then pellet was kept dried by lyophilization and yield nanoparticles in the form of powder. Then this powder was used for characterization and for application on plants (Scheme 1).
Scanning electron microscope (SEM)
SEM was used to scan the sample surface morphology. SEM is analytical tool that determines the nanomaterial morphology through beam of electron to produce morphology by directing beam of electron to produce high magnification images of samples surface topography. Biologically synthesized nanoparticles were investigated morphologically by scanning electron microscopy (SEM LEO 1530, Germany). Highly dispersed nanoparticles solution was coated on silicon chip. After drying of chip, samples were placed for scanning.
X-ray diffraction (XRD)
XRD was used to measure the average size of nanoparticles by determining the crystallographic structure. XRD works by irradiating the X-rays on nanomaterials and measuring the intensity and scattering angle that passes through nanomaterial. Thermo-scientific diffractometer powder X-ray diffraction (PANalytical X’PERT PRO, USA) was used for X-ray diffraction. One-gram fine powder ground and homogenized powder was loaded on sample for XRD analysis. The resultant peak on 2θ scale was compared with reference peaks. CuK-alpha radiations (Lambda = 0.1542 nm, 40 kV, 20 mA) generated crystallographic structure and the phase transitions of the nanoparticles. The size of nanoparticles was calculated by using the Debye Scherrer’s equation, i.e., \({\text{t }} = {\text{ k}}\lambda \, / \, \beta {\text{ cos}}\theta ,\)where t = size of the crystallite, k = shape factor, λ = wavelength of radiation.
Fourier transform infrared spectroscopy (FTIR)
Fourier transform infrared (FTIR) spectroscopy analysis was carried out using a PerkinElmer Spectrum-100 FT-IR spectrometer (FTIR-Bruker TENSOR-27). FT-IR analyses were carried out in the spectral range of 4000–500 cm−1. One gram of fine powder of sample was placed on small crystal area against high refractive index prism. It categorizes the functional groups of samples and provide qualitative and quantitative investigation. FTIR determines the chemical bond and bendings which participate in reduction by producing an infrared absorption spectrum.
Pot experiment
Experiment site
The pot experiment was conducted in wire house of Plant Virology Section, Ayub Agricultural Research Institute (AARI), Faisalabad, Pakistan (31.4° N, 73° E) and plant analysis were done in Department of Environmental Sciences, Govt. College University Faisalabad, Pakistan (31.4° N, 73.06° E).
Growth conditions
Healthy seeds of cotton variety FH-LALAZAR were taken from AARI, Faisalabad. Seeds were treated with 80 ml of 65% dilute sulphuric acid for 15 min to remove fiber from seed, rinsed with distilled water and sown in 5 kg pots. Experiment was conducted in July when minimum and maximum temperatures were 28 °C and 39 °C with average humidity of 35%. A clayey loam soil taken from research station were meshed, homogenized and sieved by 2-mm sieve. Each pot was filled with 5 kg of soil. Physical and chemical properties of soil are given in Table 1.
There were 60 pots in this experiment. Randomly 5 seeds were sown in each pot. After 2 weeks thinning was performed to retain 3 plants in each pot.
Treatments
The fertilizer was applied in the experimental pots at (150, 60 and 60 kg ha−1) but for 5 kg at 0.375 g, 0.15 g, 0.15 g in the form of urea (46.6% N), triple super phosphate (20% P) and muriate of potash (60% K), respectively. These are recommended doses of NPK for cotton crop production. Twos-third of urea and entire dose of triple super phosphate and muriate of potash were applied to pots at sowing. The rest one-third of urea was top-dressed at 20 days after sowing just before the irrigation.
Pots were treated by foliar application of four nanoparticles, zinc oxide nanoparticles (ZnO NPs), iron oxide nanoparticles (FeO NPs), copper nanoparticles (Cu NPs) and silver nanoparticles (Ag NPs) having five levels (0, 25, 50, 75 and 100 ppm). Different levels of nanoparticles were prepared by using deionized water. After emergence every week foliar application of nanoparticles were performed thrice during whole experiment.
Measurement of plant biomass and growth
Plant samples were harvested after 6 weeks of sowing. Average data regarding shoot length (SL), root length (RL), shoot fresh weight (SFW) and root fresh weight (RFW) were calculated from each pot.
Gas exchange parameters
Gas exchange parameters, stomatal conductance (gs), net photosynthesis rate (pn), water use efficiency (Pn/E) and transpiration rate (E) were measured after 6 weeks of sowing before harvesting by using ADC-225-MK3 + WA-161-MK3A Infra-Red Gas Analyzer (Analytical Development Company, Hoddesdon, UK).
SPAD values
SPAD values (net photosynthesis rate) were determined by SPAD-Plus-502 (Soil–Plant Analysis Development). SPAD meter determines the transmittance of red (650 nm) and infrared (940 nm) light through leaf after 6 weeks of sowing by SPAD-Plus-502 m (Zhejiang Top Instrument Co., Ltd., China).
Chlorophyll contents
Chlorophyll content comprising chlorophyll a, chlorophyll b and carotenoids were determined by the method given by Patla, 1990 [20]. About 0.5 g ground leaf was taken in about 80% (v/v) methanol of 10 ml kept in almost for 10 min, then centrifuged at 10,000 rpm and the extract was carried out. Supernatant and extract were separated as after 10 min the particles of extract were settled down. Supernatant was stored in small size bottles and preserved into the refrigerator. To find out the measurement of chlorophyll, spectrophotometer was used at three different wavelengths such as 663, 645 and 480, respectively. For the calculation of chlorophyll, a and chlorophyll b, Patla, 1990 [20] given method was used:
where Car = A Car/Em 100% × 100, emission = Em 100% = 2500, OD = absorbance at respective wavelength, V = volume of the extract (mL), and W = weight of the fresh leaf tissue (g).
Electrolyte leakage estimation
Data regarding electrolyte leakage were determined by method of Dionisia and Tobita, 1998 [21]. After 6 weeks of sowing, 5 mm leaf sample was taken and placed in 8 mL deionized water and placed it in water bath at 32 °C for 2 h. Then initial electrical conductance of medium (EC1) was measured. After that samples were autoclaved for 121 °C for 20 min to extract all electrolytes from sample. Then cooled at 25 °C and second electrical conductance of medium (EC2) was measured. The total electrolyte leakage was measured by using following formula:
Antioxidants estimation
Antioxidant enzymes assay like SOD, POD, APX and CAT in Cotton plant were determined spectrometrically. Samples were taken after 6 weeks of treatment. 0.5 g fresh leaves were ground with mortar and pestle, then homogenized with 0.05 M phosphate buffer (pH 7.8) under low temperature. The homogenized sample was filtered through multilayered muslin cloth and centrifuged at 4000 rpm for 4 min at 4 °C. The supernatant was taken and protocol developed by Zhang, 1994 [22] followed for SOD and POD assay.
CAT (EC1.11.1.6) activity was determined by following the protocol dev eloped by Zhang, 1994 [22]. According to this protocol, 3 mL mixture comprised 100 μL H2O2 and 100 μL enzyme extract (3000 mM) and 3 mL of 50 mM phosphate buffer along with 2 mM EDTA (pH 7.0). The enzymatic assay of CAT was determined by decreased absorption at 240 nm due to H2O2 disappearance (ε = 39.4 mM–1 cm–1).
APX (EC 1.11.1.11) enzyme activity was determined by following the protocol developed by Dhindsa et al., 1981 [23]. The reaction mixture comprised 100µL of enzyme extract, 100µL of ascorbate (7.5 mM), 100 µL of H2O2 (300 mM), and 3 mL of 25 mM potassium phosphate buffer with 2 mM EDTA (pH7.0). The oxidation of ascorbate was determined by the change in absorbance at 290 nm (ε = 2.8 mM–1 cm–1).
Malondialdehyde (MDA) content
The level of lipid peroxidation in the leaves was measured in terms of MDA content, a product of lipid peroxidation, determined by the thiobarbituric acid (TBA) reaction using the method described by Zhang et al. 1994 [24]. 0.25 g of leaf sample was homogenized in 5 mL of 0.1% trichloroacetic acid (TCA). The homogenate was centrifuged at 10,000 rpm for 5 min. 1-mL aliquot of the supernatant, 4 mL of 20% TCA containing 0.5% TBA was added. The mixture was heated at 95 °C for 30 min and then quickly cooled in an ice bath. After centrifugation at 10,000 rpm for 10 min, the absorbance of the supernatant at 532 nm was read and the value for the nonspecific absorption at 600 nm was subtracted. The MDA content was calculated by using an extinction coefficient of 155 mM–1 cm–1.
Hydrogen peroxide (H2O2) content
H2O2 was extracted by homogenizing 50 mg of leaves with 3 mL of phosphate buffer (50 mM, pH 6.5) (Dhindsa et al. 1981 [23]). To measure H2O2 content, 3 mL of extracting solution was mixed with 1 mL of 0.1% titanium sulfate in 20% (v/v) H2SO4 and the mixture was centrifuged at 6000 rpm for 15 min. The intensity of the yellow color of the supernatant was measured at 410 nm. H2O2 content was computed by using the extinction coefficient of 0.28 µmol–1 cm–1.
Statistical analysis
Statistix software (version 8.1) was used to accomplish analysis of variance. Statistix (version 8.1) software was run to perform analysis of variance. The least significant difference test (Fisher’s LSD) at 95% confidence level was performed to determine the differences in the treatments’ means.
Results
Characterization of nanoparticles
Nanoparticles synthesis was confirmed by change of color during reduction reaction. Zinc oxide nanoparticles synthesis indicated by changing greenish to yellowish color, iron oxide, copper and silver nanoparticles were confirmed by orange brown to black color, white to yellow color and white to yellow color, respectively. SEM (scanning electron microscope) micrographs in Fig. 2 revealed flaky particles with spherical texture of zinc oxide, iron oxide, copper and silver nanoparticles. Huge degree of aggregation can be seen under high and low magnifications. The dazzling spots on the surface of nanoparticles revealed the inclusions of metabolite on the surface of nanoparticles. An X-ray diffractometer revealed in Fig. 3 the average size of zinc oxide, iron oxide, copper and silver nanoparticles. Estimation of average size was done by Scherrer formula which was found to be 37.8, 29, 31.7 and 28 nm. The reflection peaks (111), (200). (220) and (311) corresponds to 2θ = 37.8°, 43.7°, 65.1° and 77.4° indicating crystallographic form of ZnO NPs. The XRD spectra of FeO NPs reflected peaks (111), (200) and (221) corresponds to 2θ = 44.7°, 58.3° and 77.6° depicted FeO NPs. Similarly, peaks of Cu NPs (111), (200), (220) and (311) correspond to 2θ value = 38.4, 45.1, 64.9 and 79.1 indicate Cu NPs. Meanwhile, reflection of peaks (111), (200), (220) and (311) corresponds to peak value at 2θ = 43.9°, 52.3°, 75.6° and 88.1° indicating Ag NPs.
In Fig. 4a, the peak 3398 cm−1 corresponds to (OH) hydroxyl group. The peaks at 2051, 1363 cm−1 are assigned to stretching asymmetric bending vibration of (C=O) and (C–H) of carboxyl moieties like ketone and aldehyde groups. These functional groups are responsible for reduction of zinc oxide nanoparticles (ZnO NPs). The peaks at 589 cm−1 indicated the Zn–O stretch.
In Fig. 4b, c the peaks 3214 and 1619 cm−1 correspond to (OH) hydroxyl group and (C = O) carbonyl group indicating the active functional groups responsible for the reduction of iron oxide nanoparticles (FeO NPs) and copper nanoparticles (Cu NPs). Similarly, in Fig. 3d the peak 1629 cm−1 is due to (C = O) group and peak 580 cm−1 intimated (Ag–O) bending.
Pot experiment
Plant growth and biomass parameters
Physical growth parameters of shoot length (SL), root length (RL), shoot fresh weight (SFW) and root fresh weight (RFW) are presented in Fig. 5. Plant growth parameters (SL, RL, SFW and RFW) were increased as 13%, 15%, 15.9% and 31.3%, respectively, by increasing concentration of zinc oxide nanoparticles (ZnO NPs). Maximum increase in growth parameters was observed at 100 ppm concentration while minimum value of plant growth parameters was observed at 0 ppm concentration of zinc oxide nanoparticles (ZnO NPs). Iron oxide nanoparticles (FeO NPs) application showed increase in physical growth parameters (SL, RL, SFW and RFW) up to 50 ppm concentration as 7.9%, 13%, 12.7% and 23.6%, respectively. After that at 100 ppm concentration showed decrease in physical growth parameters as 6.7%, 8.2%, 16.2% and 25.8% as compared to 50 ppm concentration, respectively. Maximum increase in plant growth parameters were observed at 50 ppm while minimum at 100 ppm concentration of FeO NPs. Application of copper nanoparticles (Cu NPs) also increased the physical growth parameters (SL, RL, SFW and RFW) up to 50 ppm by 11.2%, 13.6%, 8.8% and 16.6%, respectively, and afterwards plant showed stress and decrease in values were observed as 9.6%, 11.4%, 7.5% and 14.2, respectively. Maximum increase in plant growth parameters were observed at 50 ppm while minimum at 100 ppm concentration of copper nanoparticles (Cu NPs). Physical growth parameters (SL, RL, SFW and RFW) increased as 4.8%, 1.2%, 5.9% and 3.71%, respectively, by application of silver nanoparticles (Ag NPs) up to 25 ppm and then showed decrease as 6.33%, 0.9%, 3.1% and 2.94%, respectively, up to 100 ppm due to phytotoxicity.
Photosynthetic pigments and SPAD values
Application of zinc oxide nanoparticles (ZnO NPs) induced cotton growth and biomass, which is explained by significant increase in chlorophyll a, chlorophyll b, carotenoids and SPAD values as shown in Fig. 6. Zinc oxide nanoparticles (ZnO NPs) has increased the photosynthetic pigments by 37% and SPAD values as 13.7% up to 100 ppm. Iron oxide nanoparticles (FeO NPs) application also promoted the cotton growth by increasing photosynthetic pigment as 24.3% and SPAD values 12.3% up to 50 ppm. Photosynthetic pigments and SPAD values were decreased as 11%, respectively, on 50 and 100 ppm concentration of FeO NPs. Similarly, copper nanoparticles (Cu NPs) increased photosynthetic pigment as 27% and SPAD values 13.1% on 50 ppm concentration of Cu NPs and decline as 9.7% and 7.3%, respectively, on 75 and 100 ppm concentration. While silver nanoparticles (Ag NPs) promoted photosynthetic pigment and SPAD values up to 25 ppm as 14 and 6.8%, respectively. After threshold (25 ppm) photosynthetic pigment and SPAD values decreased as compared to control and 25 ppm as 8.7% and 2.4%.
Gas exchange parameters and water use efficiency
Figure 7 indicates the gas exchange parameters, stomatal conductance (gs), net photosynthesis rate (pn), water use efficiency (Pn/E) and transpiration rate (E) were increased by foliar application of zinc oxide nanoparticles (ZnO NPs) as 14.5%, 20.9%, 41.6% and 47%, respectively, by increasing concentration up to 100 ppm concentration of ZnO NPs. Meanwhile, gas exchange parameters and water use efficiency were increased by foliar application of FeO NPs up to 50 ppm concentration as13.4%, 34.5%, 33.4% and 29.7% and then decreased up to 100 ppm as 8.4%, 11.1%, 13.6% and 9.4%, respectively. Maximum gas exchange and water use efficiency were observed at 25 ppm and minimum at 100 ppm concentration. Copper nanoparticles (Cu NPs) promote gas exchange parameters up to 50 ppm concentration as 13.2%, 28.6%, 31.6% and 29.1%. After that 75 ppm and 100 ppm concentration showed inhibition as 8.4% 11.1% 13.6 and 9.4%, respectively. Silver nanoparticles (Ag NPs) regulate gas exchange parameters up to 25 ppm as 13%, 20.4%, 14.6% and 11.9% and then showed decreased in gas exchange parameters as 6.8%, 9.8%, 14.6% and 4.7%, respectively.
Effect of different concentrations of zinc oxide, iron oxide, copper, and silver nanoparticles (0, 50, 75, and 100 ppm) on plant height: A net rate of photosynthesis; B stomatal conductance; C transpiration; and D water use efficiency in cotton plants. Values show the means of 3 replicates ± standard error (n = 3)
Antioxidant assay estimation
Enzymatic antioxidants CAT, SOD, POD and APX were increased by zinc oxide nanoparticles (ZnO NPs) as shown in Fig. 8, from 0 to 100 ppm concentration as 41%, 43.7%, 40.9% and 47.1%, respectively. But iron oxide nanoparticles (FeO NPs) application showed antioxidant increase up to 50 ppm as 37.6%, 39.3%, 41.7% and 44.1% and then decreased due to phytotoxicity as 16.4%, 13.1%, 19.4% and 21.2%. Copper nanoparticles (Cu NPs) application increased antioxidants CAT, SOD, POD and APX as 33.1%, 14.9%, 27.8% and 34.8% up to 50 ppm and showed decline as 11.2%, 9.9%, 10.8% and 13.3%, respectively. Silver nanoparticles showed increase only up to 25 ppm as 14.9%, 9.8%, 7.8% and 11.2% and decreased as 13.7%, 4.2%, 5.9% and 7.8%, respectively.
Electrolyte leakage, MDA and H2O2
Figure 9 reveals that zinc oxide, iron oxide, copper and silver nanoparticles significantly affected the electrolyte leakage, MDA and H2O2 in cotton crop. Zinc oxide nanoparticles (ZnO NPs) showed significant decrease by increasing concentration up to 100 ppm as 14.7%, 41% and 37.6%, respectively. Iron oxide nanoparticles (FeO NPs) down regulate electrolyte leakage, MDA and H2O2 in cotton crop up to 50 ppm as 9.8%, 34.3% and 32.6% and afterwards oxidate stress increased up to 100 ppm as 11.2%, 13.1% and 7.8%, respectively. Copper nanoparticles also decreased oxidative stress as 10.4%, 27.8% and 7.2% up to 50 ppm concentration and increased after threshold (50 ppm) as 15.1%, 19.6% and 11.3% up to 100 ppm concentration of Cu NPs, respectively. Silver nanoparticles (Ag NPs) decreased oxidative stress parameters up to 25 ppm by 13.6%, 8.9% and 7.8% and then increased up to 100 ppm concentration as 9.12%, 7.1% and 6.8%, respectively, for electrolyte leakage, MDA and H2O2.
Discussion
In this study zinc oxide nanoparticles (ZnO NPs) improved physical growth parameters up to 13 to 30%. ZnO NPs foliar application, improved plant growth parameters, physiological parameters and antioxidants up to concentration of 100 ppm. However, oxidative stress parameters were decreased. The health of any plant is depicted by antioxidants or physiological parameters like chlorophyll content, etc. ZnO NPs are supposed to enhance chlorophyll content that might improve the plant metabolism and physical growth parameters. The possible reason is that ZnO NPs being micronutrient might increase enzymes which play vital role as antioxidants and scavenge (ROS) reactive oxygen species. Zinc is important micronutrient and regulates many enzymatic reactions like alcohol dehydrogenase, super oxide dismutase, carbonic anhydrase and RNA polymerase [8]. Meanwhile oxidative stress parameters were decreased by ZnO NPs application [25]. Similar results are also reported by [26,27,28,29,30] that ZnO NPs enhance plant metabolism and physiological parameters. Zinc deficiency led plants to loss of many enzyme reactions because zinc is co-factor of many enzymes. For example, an enzyme named carbonic anhydrase increases the CO2 content in chloroplast which greatly affects the rate of photosynthesis by enhancing Rubisco contents. Moreover, zinc deficiency causes various developmental abnormalities like stunted growth and spikelet sterility in Poaceae family [31].
Application of iron oxide nanoparticles (FeO NPs) increased the plant growth parameters, physiological parameters and antioxidants up to 7–27% on 50 ppm concentration of iron oxide nanoparticles and then plant metabolism declined on 100 ppm. Results of study showed that FeO NPs regulate plant metabolism till threshold and after that nanoparticles become toxic [32,33,34]. Iron play’s critical role in many physiological and biochemical processes like photosynthesis, that’s why chlorophyll content and antioxidants increased till the threshold concentration, because iron also serves as component of cytochrome and also required for chlorophyll synthesis. Iron also promotes the biochemical processes like DNA replication and respiration due to which antioxidants might be increased [35, 36]. According to [34], iron oxide nanoparticles can improve nitrogen uptake and plant metabolism due to which it is possible that initial growth of cotton was increased. But higher concentration, i.e., 75 and 100 ppm of Feo NPs, there was possibility of iron accumulation in plants that may cause decrease in plant biomass and physiological parameters up to 7.9–23.6%. This decrease may be explained by hydroxyl ion radical production via Fenton reaction [37] and oxidative stress damage by ROS (reactive oxygen species) production which might destabilize the plant metabolism [38]. Actually, iron is essential nutrient for all organisms and insoluble in alkaline and aerobic soils. Iron is present in huge amounts in soil but unavailable to plants due to bonded nature of iron. Iron stimulates many physiological processes in plants, such as chlorophyll biosynthesis, respiration and redox reactions [39]. Iron toxicity is caused by accumulation of iron in plants; during iron stress iron acts catalytically and generates reactive oxygen radicals via Fenton radicals which damage proteins, lipids and DNA [40]. Iron deficiency and toxicity both cause physiological abnormalities in plants [41].
Copper nanoparticles (Cu NPs) promoted physical growth parameters, physiological parameters and antioxidants up to 6.8–37% at 50 ppm concentration of Cu NPs. Actually, copper as a nutrient required as co-factor for various metalloproteins which is responsible for increasing chlorophyll content [42, 43]. Results also indicated that phytotoxic threshold of Cu NPs is narrow because being micronutrient required in very small amount. Similar results are also reported by [44,45,46] that Cu NPs promoted plant growth within threshold. [47] reported that copper nanoparticles increase initial seed germination and causes vigorous seedling, which is possibly due to increasing enzymatic reactions because copper act as co-factors in various enzymes and Cu NPs toxicity causes inhibition of biomass and various processes like chlorophyll and respiration [48] and ultimately decrease in physical growth parameters, physiological parameters and antioxidants up to 9.6—14.9% due to accumulation of copper. Lee 2008 also reported phytotoxicity of copper nanoparticles on wheat and mung bean. Copper is important micronutrient for plants; it acts as an important catalyst for numerous metalloproteins. Various physiological processes like photosynthesis and respiration are driven by copper in plants. Copper stress causes stunted growth, necrosis, apoptosis and decolorization of leaf. Copper stress induces oxidative stress in plants by generating reactive oxygen species (ROS). Similarly, copper deficiency symptoms are appeared on newer leaves [44].
Silver nanoparticles (Ag NPs) have increased plant biomass and physiological parameters from 4 to 13.6% on 25 ppm concentration of Ag NPs by foliar application on cotton. Similar result was reported by [49] and [50]. According to [51], Ag NPs promoted the plant biomass of maize. But Ag NPs have narrow threshold as compared to other nanoparticles and the reason behind this is silver is not a micronutrient [52]. Ag NPs decreased plant growth parameters at 50, 75 and 100 ppm concentration of Ag NPs concentration due to toxicity. Ag NPs toxicity might decrease the uptake of other nutrients. Actually, silver ions accumulation causes inhibition of chlorophyll synthesis, decline in transpiration and nutrient uptake. So, ultimately plant growth and biomass reduced [53]. Meanwhile silver is known for antimicrobial activity in many studies. Although silver is not a micronutrient, it is used for mitigation of various diseases in plants [54]. Silver nanoparticles enhance plant growth [55].
Overall, the accumulation of metals when they exceeded from threshold could cause cell death in plants and it also blocks the binding site, thus competition created between metal ions and other nutrients. Nanoparticles have various applications in agriculture field. In this study, we tried to biosynthesize the four metallic nanoparticles of zinc oxide, iron oxide, copper and silver from leaf extract of Conocarpus erectus L. These nanoparticles were applied foliarly to cotton crop at different concentrations in order to determine the threshold concentration. Various studies have found that after a specific threshold concentration, nanoparticles become toxic and hence decreased the plant growth and disturb the plants antioxidant system. Meanwhile, instead of providing the beneficial effects on plant growth, overutilization of such nanoparticles showed metal toxicity, inhibiting plant growth and photosynthesis (Yusefi-Tanha., 2020). They can breakdown the protein synthesis and disturb the antioxidant defense by down regulating antioxidant enzymes like SOD, POD and CAT that reduce plant abiotic and biotic stress resistance (Kumar., 2020). The description of quantitative results is briefly given here.
Conclusion
The basic objective of this study is to find phytotoxic threshold of green routed Conocarpus-mediated zinc oxide, iron oxide, copper and silver nanoparticles on cotton. All the nanoparticles were applied in five concentrations (0, 25, 50, 75 and 100 ppm). Zinc oxide nanoparticles showed maximum plant biomass and antioxidant system. Iron oxide and copper nanoparticles showed maximum growth at 50 ppm concentration while silver nanoparticles showed maximum result at 25 ppm. The health of plant is indicated by chlorophyll content and antioxidants. Metal nanoparticles enhance enzymatic activity which positively regulates the plant growth and biomass. But after threshold nanoparticles accumulation downregulates plant growth and biomass.
Availability of data and materials
Other data could be made available upon request to the corresponding author.
References
Usman M, Farooq M, Wakeel A, Nawaz A, Cheema SA, et al. Nanotechnology in agriculture: current status, challenges and future opportunities. J Sci Total Environ. 2020;721:137778.
Sajid M, Płotka-Wasylka J. Nanoparticles: synthesis, characteristics, and applications in analytical and other sciences. Microchem J. 2020;154:104623.
Charbgoo F, Ahmad MB, Darroudi M. Cerium oxide nanoparticles: green synthesis and biological applications. J Int J Nanomed. 2017;12:1401.
Rajput V, Minkina T, Sushkova S, Behal A, Maksimov A, Blicharska E. ZnO and CuO nanoparticles: a threat to soil organisms, plants, and human health. J Environ Geochem Health. 2020;42(1):147–58.
Ahmad S, Munir S, Zeb N, Ullah A, Khan B, Ali J. Green nanotechnology: a review on green synthesis of silver nanoparticles—an ecofriendly approach. J Int J Nanomed. 2019;14:5087.
Arumugam M, Manikandan DB, Dhandapani E, Sridhar A, Balakrishnan K, Markandan M. Green synthesis of zinc oxide nanoparticles (ZnO NPs) using Syzygium cumini: potential multifaceted applications on antioxidants, cytotoxic and as nanonutrient for the growth of Sesamum indicum. J Environ Technol. 2021;23:101653.
Feng Y, Kreslavski VD, Shmarev AN, Ivanov AA, Zharmukhamedov SK, Kosobryukhov A. Effects of iron oxide nanoparticles (Fe3O4) on growth, photosynthesis, antioxidant activity and distribution of mineral elements in wheat (Triticum aestivum) Plants. J Plants. 2022;11(14):1894.
Rizwan M, Ali S, Ali B, Adrees M, Arshad M, Hussain A. Zinc and iron oxide nanoparticles improved the plant growth and reduced the oxidative stress and cadmium concentration in wheat. J Chemosphere. 2019;214:269–77.
Li J, Chang PR, Huang J, Wang Y, Yuan H, Ren H. Physiological effects of magnetic iron oxide nanoparticles towards watermelon. J Nanosci Nanotechnol. 2013;13(8):5561–7.
Moradbeygi H, Jamei R, Heidari R, Darvishzadeh R. Investigating the enzymatic and non-enzymatic antioxidant defense by applying iron oxide nanoparticles in Dracocephalum moldavica L. plant under salinity stress. J Sci Hortic. 2020;272:109537.
Liu G, Gao J, Ai H, Chen X. Applications and potential toxicity of magnetic iron oxide nanoparticles. J Small. 2013;9(9–10):1533–45.
Zuverza-Mena N, Medina-Velo IA, Barrios AC, Tan W, Peralta-Videa JR, Gardea-Torresdey JL. Copper nanoparticles/compounds impact agronomic and physiological parameters in cilantro (Coriandrum sativum). J Environ Sci Process Impacts. 2015;17(10):1783–93.
López-Vargas ER, Ortega-Ortíz H, Cadenas-Pliego G, de Alba RK, et al. Foliar application of copper nanoparticles increases the fruit quality and the content of bioactive compounds in tomatoes. J Appl Sci. 2018;8(7):1020.
Noman M, Shahid M, Ahmed T, Tahir M, Naqqash T, Muhammad S. Green copper nanoparticles from a native Klebsiella pneumoniae strain alleviated oxidative stress impairment of wheat plants by reducing the chromium bioavailability and increasing the growth. J Ecotoxicol Environ Safe. 2020;192:110303.
Mosa KA, El-Naggar M, Ramamoorthy K, Alawadhi H, Elnaggar A, Wartanian S. Copper nanoparticles induced genotoxicty, oxidative stress, and changes in superoxide dismutase (SOD) gene expression in cucumber (Cucumis sativus) plants. J Front Plant Sci. 2018;9:872.
Salama HM. Effects of silver nanoparticles in some crop plants, common bean (Phaseolus vulgaris L.) and corn (Zea mays L.). J Int Res J Biotechnol. 2012;3(10):190–7.
Zhang H, Du W, Peralta-Videa JR, Gardea-Torresdey JL, White JC, Keller A. Metabolomics reveals how cucumber (Cucumis sativus) reprograms metabolites to cope with silver ions and silver nanoparticle-induced oxidative stress. J Environ Sci Technol. 2018;52(14):8016–26.
Singh N, Bhuker A, Jeevanadam J. Effects of metal nanoparticle-mediated treatment on seed quality parameters of different crops. J Naunyn-Schmiedeberg’s Arch Pharmacol. 2021;394(6):1067–89.
Hasnain Z, Zafar S, Shafqat U, Perveen S, Iqbal N, Qaisrani SA. Antibacterial activity of eco-friendly zinc nanoparticles prepared from leaf extract of Mentha piperita L. PJPS. 2020;33:2413–6.
Palta JP. Leaf chlorophyll content. J Remote Sens Rev. 1990;5(1):207–13.
Dionisio-Sese ML, Tobita S. Antioxidant responses of rice seedlings to salinity stress. J Plant Sci. 1998;135(1):1–9.
Zhang X. The measurement and mechanism of lipid peroxidation and SOD, POD and CAT activities in biological system. J Res Methodol Crop Physiol. 1992.
Dhindsa RS, Plumb-Dhindsa P, Thorpe TA. Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J Exp Bot. 1981;32(1):93–101.
Zhang J, Kirkham M. Drought-stress-induced changes in activities of superoxide dismutase, catalase, and peroxidase in wheat species. J Plant Cell Physiol. 1994;35(5):785–91.
Emamverdian A, Ding Y, Mokhberdoran F, Ahmad Z, Xie Y. Determination of heavy metal tolerance threshold in a bamboo species (Arundinaria pygmaea) as treated with silicon dioxide nanoparticles. J Glob Ecol Conserv. 2020;24:e01306.
Tanveer Y, Yasmin H, Nosheen A, Ali S, Ahmad A. Ameliorative effects of plant growth promoting bacteria, zinc oxide nanoparticles and oxalic acid on Luffa acutangula grown on arsenic enriched soil. J Environ Pollut. 2022;300:118889.
Sun L, Wang Y, Wang R, Wang R, Zhang P, Ju Q. Physiological, transcriptomic, and metabolomic analyses reveal zinc oxide nanoparticles modulate plant growth in tomato. J Environ Sci Nano. 2020;7(11):3587–604.
Singh J, Kumar S, Alok A, Upadhyay SK, Rawat M, Tsang DC. The potential of green synthesized zinc oxide nanoparticles as nutrient source for plant growth. J Clean Product. 2019;214:1061–70.
Venkatachalam P, Priyanka N, Manikandan K, Ganeshbabu I, Indiraarulselvi P, Geetha N. Enhanced plant growth promoting role of phycomolecules coated zinc oxide nanoparticles with P supplementation in cotton (Gossypium hirsutum L.). J Plant Physiol Biochem. 2017;110:118–27.
Hafeez B, Khanif Y, Saleem M. Role of zinc in plant nutrition-a review. AJEA. 2013;3(2):374.
Du W, Yang J, Peng Q, Liang X, Mao H. Comparison study of zinc nanoparticles and zinc sulphate on wheat growth: from toxicity and zinc biofortification. J Chemosphere. 2019;227:109–16.
Noor R, Yasmin H, Ilyas N, Nosheen A, Hassan MN, Mumtaz S. Comparative analysis of iron oxide nanoparticles synthesized from ginger (Zingiber officinale) and cumin seeds (Cuminum cyminum) to induce resistance in wheat against drought stress. J Chemosphere. 2022;292: 133201.
Bidast S, Golchin A, Baybordi A, Naidu R. Effects of Fe oxide-based nanoparticles on yield and nutrient content of corn in Cobalt-contaminated soils. J Environ Technol Innov. 2022;26:102314.
Cao X, Yue L, Wang C, Luo X, Zhang C, Zhao X. Foliar application with iron oxide nanomaterials stimulate nitrogen fixation, yield, and nutritional quality of soybean. J ACS Nano. 2022;16(1):1170–81.
Jhanzab HM, Qayyum A, Bibi Y, Sher A, Hayat MT, Iqbal J, et al. Chemo-blended Ag & Fe nanoparticles effect on growth, physiochemical and yield traits of wheat (Triticum aestivum). J Agronomy. 2022;12(4):757.
Chen Y, Zhang G, Wang H. Enhancement of photosynthetic bacteria biomass production and wastewater treatment efficiency by zero-valent iron nanoparticles. J Int J Nanomed. 2020;130(3):306–10.
Gong X, Huang D, Liu Y, Zeng G, Wang R, Wan J. Stabilized nanoscale zerovalent iron mediated cadmium accumulation and oxidative damage of Boehmeria nivea (L.) Gaudich cultivated in cadmium contaminated sediments. J Environ Sci Technol. 2017;51(19):11308–16.
Sharma S, Singh VK, Kumar A, Mallubhotla S. Effect of nanoparticles on oxidative damage and antioxidant defense system in plants. J Mol Plant Abiotic Stress Biol Biotechnol. 2019.
Hussain A, Ali S, Rizwan M, et al. Responses of wheat (Triticum aestivum) plants grown in a Cd contaminated soil to the application of iron oxide nanoparticles. J Ecotoxicol Environ Safe. 2019;173:156–64.
Delias DS, Da-Silva CJ, Martins AC, de Oliveira DS, do Amarante L. Iron toxicity increases oxidative stress and impairs mineral accumulation and leaf gas exchange in soybean plants during hypoxia. J Environ Sci Pollut Res. 2022;29(15):22427–38.
Saaltink RM, Dekker SC, Eppinga MB, Griffioen J, Wassen MJ. Plant-specific effects of iron-toxicity in wetlands. J Plant Soil. 2017;416(1):83–96.
Rai M, Ingle AP, Pandit R, Paralikar P, Shende S, Gupta I. Copper and copper nanoparticles: role in management of insect-pests and pathogenic microbes. J Nanotechnol Rev. 2018;7(4):303–15.
Wang T, Long X, Cheng Y, Liu Z, Yan S. The potential toxicity of copper nanoparticles and copper sulphate on juvenile Epinephelus coioides. J Aquatic Toxicol. 2014;152:96–104.
Kasana RC, Panwar NR, Kaul RK, Kumar P. Biosynthesis and effects of copper nanoparticles on plants. J Environ Chem Lett. 2017;15(2):233–40.
Hafeez A, Razzaq A, Mahmood T, Jhanzab HM. Potential of copper nanoparticles to increase growth and yield of wheat. J Nanosci Adv Technol. 2015;1(1):6–11.
Shobha G, Moses V, Ananda S. Biological synthesis of copper nanoparticles and its impact. Int J Pharm Sci Invent. 2014;3(8):6–28.
Seregina T, Chernikova O, Mazhaysky Y, Ampleeva L. Features of the influence of copper nanoparticles and copper oxide on the formation of barley crop. 2020.
Pohanka M. Copper and copper nanoparticles toxicity and their impact on basic functions in the body. J Bratisl Lek Listy. 2019;120(6):397–409.
Sillen WM, Thijs S, Abbamondi GR, Janssen J, Weyens N, White JC. Effects of silver nanoparticles on soil microorganisms and maize biomass are linked in the rhizosphere. J Soil Biol Biochem. 2015;91:14–22.
Ali A, Mohammad S, Khan MA, Raja NI, Arif M, Kamil A. Silver nanoparticles elicited in vitro callus cultures for accumulation of biomass and secondary metabolites in Caralluma tuberculata. Artif Cells Nanomed Biotechnol. 2019;47(1):715–24.
Cao J, Feng Y, He S, Lin X. Silver nanoparticles deteriorate the mutual interaction between maize (Zea mays L) and arbuscular mycorrhizal fungi: a soil microcosm study. J Appl Soil Ecol. 2017;119:307–16.
Yang J, Jiang F, Ma C, Rui Y, Rui M, Adeel M. Alteration of crop yield and quality of wheat upon exposure to silver nanoparticles in a life cycle study. J Agric Food Chem. 2018;66(11):2589–97.
Siddiqui MH, Al-Whaibi MH, Firoz M, Al-Khaishany MY. Role of nanoparticles in plants. J Nanotechnol Plant Sci. 2015.
Dang F, Wang Q, Cai W, Zhou D, Xing B. Uptake kinetics of silver nanoparticles by plant: relative importance of particles and dissolved ions. J Nanotoxicol. 2020;14(5):654–66.
Ibrahim E, Fouad H, Zhang M, Zhang Y, Qiu W, Yan C. Biosynthesis of silver nanoparticles using endophytic bacteria and their role in inhibition of rice pathogenic bacteria and plant growth promotion. J RSC Adv. 2019;9(50):29293–9.
Acknowledgements
The authors would like to thank Mr. Muhammad Usama Yasin, Scientific Officer, Plant Pathology Research Institute, Ayub Agricultural Research Institute Faisalabad.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
All the authors wrote the original draft of manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Shafqat, U., Hussain, S., Shahzad, T. et al. Elucidating the phytotoxicity thresholds of various biosynthesized nanoparticles on physical and biochemical attributes of cotton. Chem. Biol. Technol. Agric. 10, 30 (2023). https://doi.org/10.1186/s40538-023-00402-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40538-023-00402-x