Abstract
Background
The prevalence of understory removal and anthropogenic nitrogen (N) deposition has significantly altered the ecological processes of forest ecosystems at both regional and global scales. However, it remains a pressing challenge to understand how N deposition and understory removal affect leaf nutrient dynamics, nutrient resorption, litter decomposition, and their linkages for better managing forest ecosystems under nutrient imbalances induced by N enrichment. To address this research gap, a field manipulation experiment was carried out in a subtropical Cunninghamia lanceolata plantation with four treatments including: control (CK), canopy N addition (CN), understory removal (UR), and canopy N addition plus understory removal (CN × UR). Green and senesced leaf N and phosphorus (P) concentrations, N and P resorption efficiencies, litter decomposition, and their correlations were measured.
Results
The results revealed that the average N concentrations of green early and late leaves in UR were increased by 6.61 and 18.89% compared to CK. UR had the highest whereas CN had the lowest P concentrations in green leaves across the two sampling seasons. Following this, UR, leaf type, season, and their interactions significantly affected leaf N, P, and N:P (P < 0.05). The highest leaf N resorption (32.68%) and P resorption efficiencies (63.96%) were recorded in UR. Litter decomposition was significantly retarded in UR (P < 0.01) relative to CN. The regression analysis demonstrated that leaf nutrient status was significantly interconnected with leaf nutrient resorption efficiencies. In addition, leaf nutrient dynamics were strongly correlated with litter nutrients, indicating that both were coupled.
Conclusion
These findings can deepen our knowledge of biogeochemical cycling and reveal contrasting nutrient-acquisition strategies on N and P limitation in response to UR and CN. Considering the P limitation, it is important to note that P was resorbed more efficiently, illustrating a remarkable nutrient preservation approach for nutrient-limitations. Resorption may be a crucial mechanism for keeping nutrients in these forests, so better understory management practices are required to prevent reliance on external nutrient pools. Overall, this study sheds meaningful insights into the ability of forest adaptation in response to global climatic change.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The Anthropocene has drastically altered global atmospheric nitrogen (N) deposition, leading to substantial nutrient alteration and elemental imbalances in forest ecosystems (Gundale 2022). The emission of total reactive N around the world has increased by 7.3 Tg N yr−1 or 30% in recent years (Tian et al. 2020). China has emerged as a major hotspot for reactive N emissions (Cui et al. 2013), which are anticipated to reach 63 Tg N yr−1 by 2050 (IPCC 2022). Despite the fact that N is a fundamental nutrient that restricts plant growth and alters leaf nutrient resorption efficiencies, as well as plant stoichiometry (Moreau et al. 2019), excessive N accumulation on the other hand, has a cascading impact on the stability and functioning of forest ecosystems (Liu et al. 2019). Thus, understanding the effects of increased N deposition on plant nutritional status and resorption efficiencies would provide more realistic and integrated insights into nutrient reactions to global environmental challenges.
Long-term management practices such as understory plant removal exhibited strong influence on the functioning of forest ecosystems by directly or indirectly modifying soil biogeochemical processes (Zhang et al. 2022a). However, understory removal and its consequences on subtropical ecosystem processes where atmospheric N deposition is rising are typically ignored mostly in ecological research (Kumar et al. 2018). On the one hand, understory plants were eliminated to prevent competition for resources utilization between trees and the understory plants (Giuggiola et al. 2018; Yang et al. 2018; Yu et al. 2022). On the other hand, understory vegetation is a pivotal stratum of forest ecosystems that has a considerable impact on key processes like forest regeneration, soil nutrient and carbon cycling (Landuyt et al. 2019). Numerous studies have demonstrated a strong connection between understory plants and belowground ecological processes such as altering organic carbon, microbial biomass, litter decomposition, nutrient inputs, and retention (Trentini et al. 2018; Zhang et al. 2022a). Understory vegetation usually has faster nutrient turnover rates compared with the canopy (Landuyt et al. 2019). Thus, understory management should be of greater concern in order to comprehensively understand their mechanistic linkages in subtropical ecosystem processes.
Acquisition of leaf nutrients and the recycling of nutrients are key ecological processes that are significantly regulated by anthropogenic N deposition in forest ecosystems. Increased N input generally leads to higher foliar N concentrations and fulfills the plant N demands in N-limited forests (Minocha et al. 2015; You et al. 2018a). Meanwhile, this situation raises environmental concerns as it negatively affects ecosystem function and stability (Liu et al. 2019; Gundale 2022). The excessive influx of N can create a shift in nutrient limitation patterns from N to P, causing intense competition for scarce nutrients in N-saturated ecosystems. For instance, the addition of N can modify the leaf N:P ratio, further exacerbating phosphorus (P) limitation and potentially shifting it from N to P limitation (You et al. 2018b). Although extensive research on leaf nutrient dynamics and N deposition on ecosystem processes (Zhou et al. 2017a; Tong et al. 2021; You et al. 2021) had been conducted by adding N directly to the forest understory or soil (Wortman et al. 2012; Zhang et al. 2022b), most of these experiments failed to account for some key processes that occur in the forest canopy, such as canopy N interception and uptake (Liu et al. 2021). Recent literature has revealed that understory addition of N considerably overstated the detrimental effects of N deposition by 375–472% compared to canopy N-addition (Zheng et al. 2018). Therefore, emphasizing the importance of canopy N addition is a more realistic strategy for modeling the actual processes of atmospheric N deposition and will provide reliable information about the impact of ecological changes on nutrient cycling in forest ecosystems.
Nutrient resorption and litter decomposition play a significant role in controlling nutrient budgets and plant nutrient use efficiency in forest ecosystems (Wan et al. 2021). Nutrient resorption allows plants to withdraw nutrients such as N and P before abscission from senescent structures to developing tissues (Prieto and Querejeta 2020). Nutrient resorption reduces nutrient loss and minimizes plant reliance on soil nutrient availability (Li et al. 2020). Approximately 31% and 40% of the annual N and P requirements of plants, respectively, are fulfilled via the resorption process (Xu et al. 2017). In addition, a global meta-analysis estimated that nitrogen and phosphorus resorption efficiencies (NRE and PRE) were 62.1% and 64.9%, respectively (Vergutz et al. 2013). It has been found that anthropogenic N addition can have positive (Shen et al. 2018; Luo et al. 2022), negative (Lü et al. 2016), or neutral effects (Kou et al. 2017) on nutrient resorption efficiencies. Moreover, it is still debated whether plants in nutrient-poor environments resorb more nutrients than those in nutrient-rich environments (See et al. 2015). These inconsistent patterns are most likely a response to nutrient limitations on plant growth (Yan et al. 2015). For instance, under low nutrient availability conditions, plants would increase variability in their own nutrient concentration and sustain high resorption efficiency (Additional file 1: Fig. S1a) (Chen et al. 2021). Similarly, in an environment of unbalanced nutrition supply, plants resorb nutrients based on their level of nutrient limitation status (Sun et al. 2023), a process known as “relative nutrient limitation” (Additional file 1: Fig. S1b). If a plant’s growth is limited by N, it will absorb more N than other nutrients. In contrast, if the plant is P-limited, more P will be resorbed. Furthermore, stoichiometric control of leaf multiple nutrient resorptions could also be regulated by “stoichiometry control” (Additional file 1: Fig. S1c) (Sun et al. 2023). To date, research on the impacts of canopy N addition and understory removal on nutrient resorption is relatively limited, which impedes our understanding of plant nutrient conservation strategies and nutrient recycling in forest ecosystems.
The N enrichment considerably affects leaf litter fall kinetics and decomposition by changing the soil or litter quality (Pichon et al. 2020). Litter decomposition is tightly linked with nutrient resorption, in general, high nutrient resorption capacities are usually associated with lower nutrient concentrations and slower decomposition rates in plants (Killingbeck 1996; Deng et al. 2018). Litter-derived nutrient is often considered to be an important factor influencing decomposition processes due to the nutrient demands of the decomposing microbiota (Tong et al. 2021). Specifically, the litter-derived C stimulates the decomposition of soil organic C by influencing the structure and activity of the soil microbial community (Zhang et al. 2023). Inversely, the rate of litter decomposition might be influenced by soil microbial biomass, which contributes to ensure optimal nutrient use efficiency (Müller et al. 2017). The effects of N addition on litter decomposition have been debated since inconsistent patterns have been observed (Keeler et al. 2009; Zhou et al. 2017b; Ren et al. 2018). Increased N-deposition can result in escalation of litter decomposition rates, because of the abundance of N that is available to decomposers in soil ecosystems (Gill et al. 2022). In addition, understory vegetation removal hinders the rate of litter decomposition by reducing substrate availability for soil microbiota (Additional file 1: Fig. S1) (Wu et al. 2011; Wan et al. 2021). On the contrary, Jonathan et al. (2016) pointed out that understory vegetation removal hastens the decomposition of the Pinus sylvestris litter. Therefore, it is imperative to develop conceptual frameworks for the better understanding of ecological processes by predicting the interspecific variabilities of foliar litter decomposition and nutrient uptake among various forest ecosystems in response to N deposition. However, how and to what extent increased atmospheric N deposition and management practices affect the multiple nutrient-associated processes such as leaf nutrient dynamics, nutrient resorption, litter decomposition, and their linkages remains unclear in forest ecosystems.
In this study, we conducted canopy N deposition and understory removal in Cunninghamia lanceolata (Chinese fir) forests since they are high-yielding, evergreen conifers with leaves that typically grow and senesce simultaneously (Wu et al. 2019; Liu et al. 2022). The relationships between various nutrient-interlinked processes were detected to provide comprehensive empirical evidence such as leaf nutrient dynamics, nutrient resorption, and litter decomposition in response to canopy N addition and understory removal in subtropical Chinese fir plantations. We tested the following hypotheses: (i) Understory removal would significantly increase leaf nutrient concentrations and leaf nutrient stoichiometry by reducing field nutrient competition; (ii) Canopy N addition might increase litter decomposition while decreases NRE due to the increased N input, similarly leaf P concentration decreases and PRE increases because of N-induced acidification and lower P supply; (iii) The relationships between foliar nutrients, resorption, litter nutrients, and their stoichiometry should be coupled due to plant nutrient acquisition and conservation strategies in response to nutrient addition.
Materials and methods
Study area
The experimental site was situated in Sanming city, at the Guanzhuang National Forestry Farm (26° 30′ N and 117° 43′ E), Fujian Province, China. The ecosystem experiences typical subtropical monsoon climate, with mean annual rainfall between 1606 and 1650 mm and mean annual temperatures between 18.8 and 19.6 °C. Soil is classified as an acrisol in the U.S. soil taxonomy. The basic soil properties were as follows: mean soil organic carbon 18.38 g kg−1, soil bulk density 1.06 g cm−3 and soil pH 4.68. The average annual wet N deposition in this region from rainfall (including NO3− and NH4+) was 4.2–5.7 g N m−2 yr−1 (Wu et al. 2019). The study was carried out in a Cunninghamia lanceolata (Chinese fir) plantation forest that was planted in 2008 at a density of 1660 trees ha−1. At the time of the study, the mean height and diameter at breast height were 5.11 m and 7.34 cm, respectively. The predominant species of understory were Ardisia punctate, Ficus hirta, Arachniodes hasseltii, and Smilax china.
Experimental design
In this study, a randomized block experiment was deployed with 4 replicates when the plantation of C. lanceolata was 6 years old. In June 2014, eight blocks (40 m × 40 in size) were established over a 6-ha section of the plantation, and each block was divided into two 15 m × 15 m plots. Each plot was inset with a 5 m × 5 m subplot for understory removal treatment. There were two factors, with four treatments randomly assigned to each of the four replicated blocks including: (i) CK, control; (ii) CN, canopy nitrogen addition (25 kg N ha−1 yr−1); (iii) UR, understory removal; and (iv) CN × UR, canopy nitrogen addition plus understory removal. A 3–8 m buffer strip was used to avoid cross-contamination between adjacent plots. For each CN plot, the required amount of NH4NO3 (269 g) was dissolved in 15 L of tap water, and the solution was sprayed over the forest canopy inside the plots. In order to apply the N on forest canopy, a high-pressure spraying equipment was placed in the center of each plot. Briefly, N solutions were pumped to a height of 5 m above the forest canopy using PVC pipes (10 cm in diameter) coupled to a tall supporting tower. The N solutions were sprayed evenly (rotating at 360°) over the canopy using crane sprinklers. The solution was sprayed once every 2 months for 15 months, starting in June 2014 and lasting until sampling in December 2016. The control plots received an equal amount of water without NH4NO3. For understory removal subplots, the existing and germinating understory vegetation were manually pulled out each month using a machete and hoe. The collected aboveground biomass of understory plants was completely removed from the experimental plots to avoid their onsite decomposition.
Field sampling and analysis
Soil samples from 0 to 20 cm were randomly collected by taking three soil cores (3 cm diameter) from each plot. Before taking the cores, plant debris was scraped off from the top of the soil. One sample per subplot was created using the three combined cores. Stones and small, discernible plant residues were manually removed after the soil samples had been air-dried. Before measuring the soil properties, all samples were grounded to pass through a 2 mm sieve. The pH of soil was measured using 1:2.5 (w/v) ratio of soil to deionized water. Fresh soil was dried at 105 °C for 24 h in order to determine the gravimetric soil moisture content.
Both green and senesced leaves were sampled in late April and December 2016, a representative set of mature green and freshly senesced but still connected leaf were sampled. Plant samples were oven dried at 75 °C for 48 h and ground by using a ball mill (NM200, Retsch) prior to chemical analysis. For C, N, and P analysis, dried leaves were crushed into a fine powder and passed through a 0.15-mm sieve. The modified Walkley–Black acid–dichromate FeSO4 titration technique was used to determine the organic C concentration of soil and plant samples (Bao 2000). A flow injection auto analyzer was utilized to determine the total N concentration following micro-Kjeldahl digestion. Following the digestion of pulverized leaf material in H2SO4–HClO4, the total P concentrations were determined colorimetrically using the molybdate/ascorbic acid procedure (Bao 2000).
The leaf litter decay experiment was performed by following the method of Wu et al. (2011). The fresh Chinese fir litter was collected from the tested plots and mixed to create a uniform mixture before filling the 1-mm mesh nylon litter bags (10 g of dried leaf litter per bag, 20 cm × 20 cm). In each subplot of the four treatments, litter bags were randomly allocated across the surface soil in May 2016. Litter bags were collected (one per subplot) from each subplot every 2 months for a total of seven times between May 2016 and July 2017. To determine the percentage of litter mass loss, the retrieved litters were cleansed with forceps, dried for 72 h at 75 °C in an oven, and weighed. The C, N, and P concentrations were chemically analyzed using the same methods as described above (Bao 2000).
Calculations and statistical analysis
Nutrient resorption efficiency (NRE/PRE) is defined as the percentage of the mature leaf nutrient pool that is resorbed from senescent leaves (Killingbeck 1996). It was calculated as follows
where N/Pgreen and N/Psenesced are the mass-based N or P concentrations of green and senescent leaves, respectively. The MLCF (mass loss correction factor) of 0.745 was used for the compensation of mass loss correction (Vergutz et al. 2013).
Prior to analysis, Shapiro–Wilk tests were run to examine the normality and homogeneity of the data. The effects of treatments on leaf nutrient concentrations and nutrient resorption efficiencies were examined discretely by one-way ANOVAs along with LSD tests. Two-way ANOVAs were applied to determine the main and interaction effects of CN and UR on foliar nutrient concentrations, resorption efficiency, and N:P ratio. Three-factor ANOVAs were carried out to analyze the main and interactional effects of CN, UR, and season (S; April and December) on leaf nutrient resorption efficiencies. Similarly, a four-factor ANOVA was employed to investigate the main and interactional effects of CN, UR, leaf type (LT, Green vs Senesced), and season (S) on the concentrations of N, P, and N:P. Furthermore, the impacts of CN, UR, and their various interrelations on litter nutrient (N, P, and C) dynamics and litter decomposition were also investigated using repeated measures ANOVA. The various relationships between leaf nutrients, resorptions, and decomposition of litter were analyzed using the function of linear regression. The statistical analysis was carried out using SPSS 25 (SPSS Inc., Chicago, IL, USA), and a confidence level of P ≤ 0.05 was used to determine significance. All illustrations and analyses were accomplished by SigmaPlot 14.0 (Systat Software, Inc., San Jose, CA) and GraphPad Prism 10.0 software (GraphPad Software, Inc., San Diego, CA, USA).
Results
Response of leaf nutrient concentration and resorption
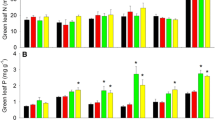
UR (P = 0.02), leaf type (LT, P < 0.01), season (S, P < 0.01), and their interactions significantly influenced leaf N, P, and N:P of early and late sampling (Fig. 1 and Additional file 1: Table S3). In contrast, CN and its interaction with UR had no significant effect on leaf nutrient concentrations but had a remarkable impact on N:P ratios and nutrient resorption efficiencies as determined by two-way ANOVAs (Additional file 1: Tables S1, S2). The concentrations of N in green early and late leaves were increased by 6.61% and 18.89% in UR and by 3.56% and 11.24% in CN compared to the control (Fig. 1a, b). In addition, UR had the highest, whereas CN had the lowest leaf P concentration in green early and late leaves (Fig. 1c, d and Additional file 1: Table S3). The highest level of senesced leaf P concentration was observed in the combined treatment of CN × UR (Fig. 1c, d).
Response of nitrogen and phosphorus concentrations in green and senesced leaves to canopy nitrogen addition (CN) and understory removal (UR) across two seasons. Data are presented as means ± SD, with four replicates. Within each of the four-bar groups (Green or Senesced treatment) values with a different letter are significantly different based on ANOVA with LSD (P < 0.05). The lower and upper cases represent green and senesced subplots, respectively. CK, control; UR, understory removal; CN, canopy nitrogen addition; CN × UR, canopy nitrogen addition plus understory removal. Main effects of CN, UR, leaf types (green vs senesced), season (S) and their interactions were evaluated with four-way ANOVA analyses. *P < 0.05; ***P < 0.001
Leaf nutrient resorption efficiencies varied dramatically with interactions across early and late leaves (Fig. 2 and Additional file 1: Table S4). The average NREs and PREs were 32.68% and 63.96% for UR and 27.64% and 55.66%, respectively, for CN. UR recorded the highest while CN plus UR recorded the lowest leaf NRE and PRE across the sampling seasons (Fig. 2a). PRE was higher than NRE without statistical significance difference (P > 0.05, Additional file 1: Table S4).
Effects of canopy N addition and understory removal on nutrient resorption efficiencies across two seasons. Data are the means ± SD of four replicates. Within each group, values with a different letter are significantly different based on ANOVA with LSD (P < 0.05). The lower and upper cases represent NRE and PRE subplots, respectively. NRE: nitrogen resorption efficiency; PRE: phosphorus resorption efficiency; CK: control; UR: understory removal; CN: canopy nitrogen addition; CN × UR: canopy nitrogen addition with understory removal
Response of leaf litter decomposition and nutrient dynamics
Two-way repeated measures ANOVA revealed that UR (P < 0.01) significantly retarded leaf litter decomposition, whereas CN recorded remarkably faster decomposition rate (Fig. 3 and Additional file 1: Table S5). There was an interactive effect between CN and UR (P = 0.02). The mean leaf litter C, N, and P concentrations were 500.3 g kg−1, 7.1 g kg−1, and 0.26 g kg−1, respectively. The effect of UR was more pronounced on C release relative to CN (Fig. 4a). Conversely, the high levels of residual N and P were usually noted in the UR plots (Fig. 4b, c). The average leaf litter stoichiometry ratios of C:N, C:P, and N:P were 65.8, 2600.9, and 38.4, respectively (Fig. 4d, f). UR, sampling date (T), and the interactions between CN × UR and UR × T significantly impacted leaf litter P during the decomposition process (P < 0.05, Additional file 1: Table S5).
The effects of canopy nitrogen addition (N), understory removal (UR), and their interactions on litter decomposition rate after 12 months in Cunninghamia lanceolata plantation. Data were presented as the mean ± standard deviation (SD) of four replicates at a significance level of P < 0.05 (The inserted p-values were from two-way repeated measures ANOVA). CK: control; UR: understory removal; CN: canopy N addition; CN × UR: canopy N addition plus understory removal
Dynamics of the remaining C, N, and P and the C:N, C:P and N:P ratios in leaf litter of Cunninghamia lanceolata plantation (a–e). CK: control; UR: understory removal; CN: canopy N addition; CN × UR: canopy N addition plus understory removal. Error bars represent the standard deviations of the means (n = 4)
Relationships among leaf nutrient concentration, resorption, litter decomposition, and their stoichiometry
Leaf litter organic carbon was positively correlated, while total N and P concentrations were negatively correlated with litter dry mass (P < 0.001 for all, Fig. 5a–c). Litter dry mass and the stoichiometric ratios of C:N, C:P, and N:P were all positively correlated (P < 0.001, Fig. 5d–f). Furthermore, green leaf P concentrations increased linearly with litter TN (P = 0.018, Fig. 6a), whereas the relationships between the N:P of green and senesced leaves and the litter TN were negative (P = 0.007 and P = 0.019, respectively, Fig. 6b, c). Contrastingly, the senesced leaf P was positively correlated, while N:P was negatively correlated with litter P (P < 0.01, Fig. 6d, e). The [N]g, [N]s, and NRE did not correlate with litter TN, TP, and OC (P > 0.05 for all, Additional file 1: Figs. S2 and S3). Litter OC decreased with increased [P]g and [P]s (Fig. 6f, g), whereas leaf N:P increased with litter OC (P < 0.001, Fig. 6h, i).
Strong correlations between NRE and PRE were observed (P < 0.001, Fig. 6j). Also, significant correlations among leaf and litter N:P ratios were found (P < 0.001, Fig. 6l). The relationship between PRE and litter N was positive (P = 0.048, Fig. 6k), while NRE showed a negative relationship with senescent leaf N and a positive relationship with green leaf N (Fig. 6m, n). The correlations between PRE with green leaf P and senescent leaf N:P were positive (Fig. 6o, p).
Discussion
Dynamics of leaf nutrient concentration and resorption efficiencies
Our findings indicated that UR increased N and P concentrations in green early and late leaves (Fig. 1), which supported our first hypothesis and the nutrient limitation theory (Gruber et al. 2018; De Lombaerde et al. 2021). The proposed mechanism is attributed to modifying soil microclimate and enhancing the availability of soil nutrients with UR (Wu et al. 2011; Li et al. 2020). First, UR increases soil water availability and temperature by allowing more solar radiation to reach the forest floor, which has the potential to increase nutrient availability by influencing soil microbial activities (Giuggiola et al. 2018). Second, soil nutrient availability was potentially increased as a result of the faster decomposition rate of the remaining detritus from understory plants (Fang et al. 2021). Understory vegetation removal allows faster growth rates of Cunninghamia lanceolata plantations due to the release of competition for other resources (Chen et al. 2022). For example, UR can facilitate tree growth via speeding up N mineralization (Fang et al. 2021).
CN substantially increased both green and senesced leaf N and N:P ratios but decreased leaf P concentration (Fig. 1 and Additional file 1: Table S3). High N deposition generally raises leaf N concentrations (Zhang et al. 2022b), while decreasing leaf P concentrations (Deng et al. 2017). The favorable responses of leaf N following N addition are well studied (Gurmesa et al. 2016; You et al. 2018a). However, CN aggravated ecosystem P limitations in our study. The first reason would be that the significant decreases in N and P concentrations of senesced early and late leaves of Chinese fir plantations occurred as a result of homeostatic control and nutrient resorption (Chen et al. 2015). Green leaves in some plants retain their stability better than their senescent leaves because of internal homeostatic regulation (Zeng et al. 2021). Second, due to N-rich environments in the subtropical forests, N-induced soil acidification would decrease available P, especially in soils with P limitations (Deng et al. 2017; Luo et al. 2022). The evidence includes the values of N resorption (average 32.68%) in our study were notably lower than the mean NRE (62.1%), while P resorption (average 63.96%) was similar to the average PRE (64.9%) at a global scale estimation (Vergutz et al. 2013). Based upon this threshold, Cunninghamia lanceolata plantations exhibit effective P and ineffective N resorption in early and late leaves, respectively. Our data affirms the second hypothesis and showed that CN considerably reduced leaf NRE since more N from soils will be absorbed by plants and minimize reliance on internal N recycling from senescing leaves (You et al. 2018b; Hu et al. 2022). According to Killingbeck (1996), plants use P efficiently when the P concentration in senescent leaves is less than 0.5 g kg−1. Consistently, P concentration in senesced leaves were less than 0.5 g kg−1, indicating that Cunninghamia lanceolata had a greater P use efficiency in this study.
We also found the higher leaf PRE in both early and late leaf sampling with UR (Fig. 2 and Additional file 1: Table S4). This might be because: (i) P is abundantly present in leaves in an organic form (Estiarte and Peñuelas 2015), which likely makes the process of P resorption less expensive. (ii) P is frequently scarce because it is less soluble and mobile in subtropical soils (Vitousek et al. 2010). The twin stresses of soil P restriction and atmospheric N deposition affect forest ecosystems frequently in subtropical areas (Liu et al. 2013). Consequently, N deposition can change soil from N limitations to P limitations (Deng et al. 2017) and lead to a more conservative use of P (i.e., greater P resorption) in plantations (Luo et al. 2022). Concretely, higher PRE compared to NRE may indicate that Chinese fir is most likely to be limited by P rather than N and follows the mechanism of nutrient limitation control (Additional file 1: Fig. S1b).
Dynamics of leaf litter decomposition and nutrient stoichiometry
In this study, UR significantly (P = 0.01) retarded the rate of litter decomposition in Chinese fir plantations (Fig. 3). Previous studies have also found that removing the understory has a detrimental impact on litter decomposition in subtropical plantations (Wu et al. 2011; He et al. 2019; Wan et al. 2021). Since the rates of litter decomposition are influenced by substrate quality and soil decomposers (Wu et al. 2019), the explanation should be that UR would hinder litter decomposition because the lower plant species resulted in varied soil microbial diversity and community composition. Although soil microbial community was not directly determined in this experiment, recent published literature (Xi et al. 2022) and other evidence (Yang et al. 2018; Qiu et al. 2021) revealed that removing the understory reduced substrate availability to soil microbes, which inhibited the microbial decomposition functions (Wu et al. 2011; Wan et al. 2021). Furthermore, Cunninghamia lanceolata litter frequently releases allelochemicals like cyclic dipeptide during decomposition (Xia et al. 2015), which can prevent the microbial degradation performance in understory removal. Consistent with our second hypothesis, CN slightly accelerated litter decomposition rates of Cunninghamia lanceolata (Fig. 3 and Additional file 1: Table S5) and supported the N limitation theory that N deposition would speed up the rate of decomposition (Gill et al. 2022). CN potentially increased the soil fungal diversity, which alternatively accelerated the litter decomposition process (Wu et al. 2019; Liu et al. 2021). In addition, the net N release only happens when the litter average C:N ratio is lower than 40 (Parton et al. 2007). In this study, the C:N ratio of Cunninghamia lanceolata litter was higher than 40 (Fig. 4d), resulting in a higher decomposition rate after N addition. Likewise, according to Dziadowiec (1987), P will release if the litter C:P values are in the range of 200 to 480, the obtained value was far lower than those in our study (Fig. 4e). Therefore, litter decomposition was enhanced and net P immobilization occurred.
Relationships among leaf resorption, nutrient stoichiometry, and litter decomposition
In this study, the relationships between green leaf N/P and NRE/PRE were significantly positive (Fig. 6). In general, the efficiency of nutrient absorption rises with lower soil nutrient levels and declines significantly when the corresponding nutrients are more readily available in soils (Xu et al. 2021). The higher resorption indicated a higher nutrient transformation from green leaves to senescent leaves (Brant and Chen 2015). These findings are compatible with some studies (Chen et al. 2021), but contradict other results (Liu et al. 2014; See et al. 2015). For instance, NRE substantially increased with leaf N concentration in the non-karst forest (Chen et al. 2021). However, Liu et al. (2014) noted that, as the concentrations of N and P in green leaves decreased, the efficiency of N and P resorption increased. The discrepancies about the nutrient resorption efficiencies would be attributed to the initial leaf and soil nutrient status (Xu et al. 2020; Chen et al. 2021). NRE and PRE had poor relationships with litter nutrient status in our study, which also supported these findings. Theoretically, if foliar concentrations indicate nutrient constraints and resorption represents a nutrient limitation, PRE will increase and NRE will decrease as leaf N:P increases (Sorrell et al. 2011). However, other than a positive relationship between leaf N:P ratios and PRE, we did not observe such a phenomenon (Fig. 6p). These unexpected results imply the possibility of additional plant strategies, such as the stoichiometry control strategy (Sun et al. 2023).
There was a strong positive relationship between NRE and PRE (Fig. 6j), which indicated that resorptions of N and P were coupled and increased concurrently. For example, a similar significant interrelation between NRE and PRE was noted in two 50-year-old forests (Chen et al. 2021). This was also supported by Liebig’s law of the minimum (Marschner 2012), that multiple nutrient balances as well as nutrient availability are necessary for plant growth. Moreover, no significant relationships were noted between the stoichiometric ratios and the resorption efficiencies, which did not support our third hypothesis. The reason would be that the nutrients N and P in plants are coupled (Fig. 6l), while resorption is dominated by their concentrations but not their ratios, adding more concrete support for the nutrient limitation control (Killingbeck 1996; Sun et al. 2023). Litter OC was positively correlated with senesced leaf N:P and green leaf N:P, while litter TN and TP had a significant negative relationship with senesced leaf N:P and green leaf N:P (Fig. 6). Although these results appeared to be incongruous, they were also logical in light of the improved N affordance and P availability. Litter N:P ratios and leaf N:P ratios were significantly interrelated. It is confirmed by the retranslocation of nutrients between soil and plant, which is constrained by soil availability (Bui and Henderson 2013).
Conclusion
The present study evaluated the unique empirical evidence of multiple nutrient-associated mechanisms of foliar nutrient status, resorption, litter decomposition, and their linkages in response to the increasing N deposition and understory removal in Cunninghamia lanceolata plantations. To our knowledge, this study is the first of its kind that sheds light on the intricate interrelationships amongst multiple nutrient cycling processes in subtropical ecosystems. Our results suggested that CN and UR can greatly affect ecosystem nutrient cycling and resorption efficiencies. We first found that UR consistently increased the concentration of foliar nutrients and resorption efficiencies. In contrast, CN enhanced foliar N concentration while decreasing P concentration and NRE. NRE and PRE changed across treatments, being lower in CN and greater in UR, suggesting that UR is better capable of obtaining nutrients and may save more resources through absorption from senescing leaves. Second, UR appeared to be the dominant factor for retarded litter mass loss, and CN promoted leaf litter decomposition. UR provided a high concentration of C, N, and P from leaf litter decomposition processes. Except for the tight relationship between leaf N:P ratio and litter N:P ratio, few relationships were found between resorption efficiency and N, P, and C stoichiometric ratios. We conclude that nutrient resorption may be a key mechanism for nutrient conservation in these plantations, where P is the most limiting nutrient. Therefore, effective understory management is crucial for nutrient conservation cycling and enhancing nutrient uptake in subtropical plantation forests. However, exploring the long-lasting legacy impacts of understory removal by further edge cutting technologies merits further studies on global ecological challenges.
Availability of data and materials
This paper is original and contains all crucial data produced or analyzed throughout this research. However more detailed information related to this current study will be provided on a reasonable request from the corresponding author.
References
Bao SD (2000) Soil and agricultural chemistry analysis, 3rd edn. China Agriculture Press, Beijing
Brant AN, Chen HYH (2015) Patterns and mechanisms of nutrient resorption in plants. Crit Rev Plant Sci 34:471–486. https://doi.org/10.1080/07352689.2015.1078611
Bui EN, Henderson BL (2013) C:N:P stoichiometry in Australian soils with respect to vegetation and environmental factors. Plant Soil 373:553–568. https://doi.org/10.1007/s11104-013-1823-9
Chen FS, Niklas KJ, Liu Y, Fang XM, Wan SZ, Wang HM (2015) Nitrogen and phosphorus additions alter nutrient dynamics but not resorption efficiencies of Chinese fir leaves and twigs differing in age. Tree Physiol 35:1106–1117. https://doi.org/10.1093/treephys/tpv076
Chen H, Reed SC, Lü X, Xiao K, Wang K, Li D (2021) Coexistence of multiple leaf nutrient resorption strategies in a single ecosystem. Sci Total Environ 772:144951. https://doi.org/10.1016/j.scitotenv.2021.144951
Chen W, Su F, Pang Z, Mao Q, Zhong B, Xiong Y, Mo J, Lu X (2022) The removal of understory vegetation can rapidly alter the soil microbial community structure without altering the community assembly in a primary tropical forest. Geoderma 429:116180. https://doi.org/10.1016/j.geoderma.2022.116180
Cui S, Shi Y, Groffman PM, Schlesinger WH, Zhu Y (2013) Centennial-scale analysis of the creation and fate of reactive nitrogen in China (1910–2010). Proc Natl Acad Sci USA 110:2052–2057. https://doi.org/10.1073/pnas.1221638110
De Lombaerde E, Baeten L, Verheyen K, Perring MP, Ma S, Landuyt D (2021) Understorey removal effects on tree regeneration in temperate forests: a meta-analysis. J Appl Ecol 58:9–20. https://doi.org/10.1111/1365-2664.13792
Deng Q, Hui DF, Dennis S, Reddy KC (2017) Responses of terrestrial ecosystem phosphorus cycling to nitrogen addition: a meta-analysis. Glob Ecol Biogeogr 26:713–728. https://doi.org/10.1111/geb.12576
Deng M, Liu L, Jiang L, Liu W, Wang X, Li S, Yang S, Wang B (2018) Ecosystem scale trade-off in nitrogen acquisition pathways. Nat Ecol Evol 2:1724–1734. https://doi.org/10.1038/s41559-018-0677-1
Dziadowiec H (1987) The decomposition of plant litterfall in an oak-linden-hornbeam forest and an oak-pine mixed forest of the Białoweża National Park. Acta Soc Bot Pol 56:169–185. https://doi.org/10.5586/asbp.1987.019
Estiarte M, Peñuelas J (2015) Alteration of the phenology of leaf senescence and fall in winter deciduous species by climate change: effects on nutrient proficiency. Glob Change Biol 21:1005–1017. https://doi.org/10.1111/gcb.12804
Fang XM, Wang GG, Xu ZJ, Zong YY, Zhang XL, Li JJ, Wang H, Chen FS (2021) Litter addition and understory removal influenced soil organic carbon quality and mineral nitrogen supply in a subtropical plantation forest. Plant Soil 460:527–540. https://doi.org/10.1007/s11104-020-04787-8
Gill A, Adler PB, Borer ET, Buyarski CR, Cleland EE, D’Antonio CM, Davies KF, Gruner DS, Harpole WS, Hofmockel KS, MacDougall AS et al (2022) Nitrogen increases early-stage and slows late-stage decomposition across diverse grasslands. J Ecol 110:1376–1389. https://doi.org/10.1111/1365-2745.13878
Giuggiola A, Zweifel R, Feichtinger LM, Vollenweider P, Bugmann H, Haeni M, Rigling A (2018) Competition for water in a xeric forest ecosystem—effects of understory removal on soil micro-climate, growth and physiology of dominant Scots pine trees. For Ecol Manag 409:241–249. https://doi.org/10.1016/j.foreco.2017.11.002
Gruber A, Oberhuber W, Wieser G (2018) Nitrogen addition and understory removal but not soil warming increased radial growth of Pinus cembra at treeline in the Central Austrian Alps. Front Plant Sci 9:711. https://doi.org/10.3389/fpls.2018.00711
Gundale MJ (2022) The impact of anthropogenic nitrogen deposition on global forests: negative impacts far exceed the carbon benefits. Glob Change Biol 28:690–692. https://doi.org/10.1111/gcb.15959
Gurmesa GA, Lu X, Gundersen P, Mao Q, Zhou K, Fang Y, Mo J (2016) High retention of 15N-labeled nitrogen deposition in a nitrogen saturated old-growth tropical forest. Glob Change Biol 22:3608–3620. https://doi.org/10.1111/gcb.13327
He W, Xu X, Zhang CC, Ma ZY, Xu JY, Ten MJ, Yan ZG, Wang B, Wang PC (2019) Understory vegetation removal reduces the incidence of non-additive mass loss during leaf litter decomposition in a subtropical Pinus massoniana plantation. Plant Soil 446:529–541. https://doi.org/10.1007/s11104-019-04378-2
Hu WJ, Yuan ZY, Shi XR, Lock TR, Kallenbach RL (2022) A global meta-analysis reveals that nitrogen addition alters plant nutrient concentration and resorption in grassland ecosystems. J Soil Sci Plant Nutr 22:4960–4971. https://doi.org/10.1007/s42729-022-00973-y
IPCC (2022) Impacts, adaptation, and vulnerability. Contribution of working group II to the sixth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge, p 3056. https://doi.org/10.1017/9781009325844
Jonathan R, Dorrepaal E, Kardol P, Nilsson MC, Teuber LM, Wardle DA (2016) Understory plant functional groups and litter species identity are stronger drivers of litter decomposition than warming along a boreal forest post-fire successional gradient. Soil Biol Biochem 98:159–170. https://doi.org/10.1016/j.soilbio.2016.04.009
Keeler BL, Hobbie SE, Kellogg LE (2009) Effects of long-term nitrogen addition on microbial enzyme activity in eight forested and grassland sites: implications for litter and soil organic matter decomposition. Ecosystems 12:1–15. https://doi.org/10.1007/s10021-008-9199-z
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727. https://doi.org/10.2307/2265777
Kou L, Wang H, Gao W, Chen W, Yang H, Li S (2017) Nitrogen addition regulates tradeoff between root capture and foliar resorption of nitrogen and phosphorus in a subtropical pine plantation. Trees 31:77–91. https://doi.org/10.1007/s00468-016-1457-7
Kumar P, Chen HY, Thomas SC, Shahi C (2018) Linking resource availability and heterogeneity to understorey species diversity through succession in boreal forest of Canada. J Ecol 106:1266–1276. https://doi.org/10.1111/1365-2745.12861
Landuyt D, Lombaerde ED, Perring MP, Hertzog LR, Ampoorter E, Maes SL, Frenne PD, Ma S, Proesmans W, Blondeel H, Sercu BK, Wang B, Wasof S, Verheyen K (2019) The functional role of temperate forest understory vegetation in a changing world. Glob Change Biol 25:3625–3641. https://doi.org/10.1111/gcb.14756
Li Q, Zhou DW, Denton MD (2020) Plant nitrogen and phosphorus resorption in response to varied legume proportions in a restored grassland. Plants 9:292. https://doi.org/10.3390/plants9030292
Liu J, Huang W, Zhou G, Zhang D, Liu S, Li Y (2013) Nitrogen to phosphorus ratios of tree species in response to elevated carbon dioxide and nitrogen addition in subtropical forests. Glob Change Biol 19:208–216. https://doi.org/10.1111/gcb.12022
Liu CC, Liu YG, Guo K, Wang SJ, Yang Y (2014) Concentrations and resorption patterns of 13 nutrients in different plant functional types in the karst region of south-western China. Ann Bot 113:873–885. https://doi.org/10.1093/aob/mcu005
Liu J, Li X, Ma Q, Zhang X, Chen Y, Isbell F, Wang D (2019) Nitrogen addition reduced ecosystem stability regardless of its impacts on plant diversity. J Ecol 107:2427–2435. https://doi.org/10.1111/1365-2745.13187
Liu S, Behm JE, Wan SQ, Yan JH, Ye Q, Zhang W, Yang XD, Fu SL (2021) Effects of canopy nitrogen addition on soil fauna and litter decomposition rate in a temperate forest and a subtropical forest. Geoderma 382:114703. https://doi.org/10.1016/j.geoderma.2020.114703
Liu WF, Duan HL, Shen FF, Liao YC, Li Q, Wu J (2022) Effects of long-term nitrogen addition on water use by Cunninghamia lanceolata in a subtropical plantation. Ecosphere 13:e4033. https://doi.org/10.1002/ecs2.4033
Lü X, Reed SC, Yu Q, Han X (2016) Nutrient resorption helps drive intra-specific coupling of foliar nitrogen and phosphorus under nutrient-enriched conditions. Plant Soil 398:111–120. https://doi.org/10.1007/s11104-015-2642-y
Luo M, Moorhead DL, Ochoa-Hueso R, Mueller CW, Ying SC, Chen J (2022) Nitrogen loading enhances phosphorus limitation in terrestrial ecosystems with implications for soil carbon cycling. Funct Ecol 36:2845–2858. https://doi.org/10.1111/1365-2435.14178
Marschner P (2012) Mineral nutrition of higher plants, 3rd edn. Elsevier/Academic Press, Waltham. https://doi.org/10.1016/C2009-0-63043-9
Minocha R, Turlapati SA, Long S, McDowell WH, Minocha SC (2015) Long-term trends of changes in pine and oak foliar nitrogen metabolism in response to chronic nitrogen amendments at Harvard Forest, MA. Tree Physiol 35:894–909. https://doi.org/10.1093/treephys/tpv044
Moreau D, Bardgett RD, Finlay RD, Jones DL, Philippot L (2019) A plant perspective on nitrogen cycling in the rhizosphere. Funct Ecol 33:540–552. https://doi.org/10.1111/1365-2435.13303
Müller K, Marhan S, Kandeler E, Poll C (2017) Carbon flow from litter through soil microorganisms: from incorporation rates to mean residence times in bacteria and fungi. Soil Biol Biochem 115:187–196. https://doi.org/10.1016/j.soilbio.2017.08.017
Parton W, Silver WL, Burke IC, Grassens L, Harmon ME, Currie WS, Jennifer YK, Adair EC, Brandt LA, Stephen CH, Fasth B (2007) Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 315:361–364. https://doi.org/10.1126/science.1134853
Pichon NA, Cappelli SL, Soliveres S, Hölzel N, Klaus VH, Kleinebecker T, Allan E (2020) Decomposition disentangled: A test of the multiple mechanisms by which nitrogen enrichment alters litter decomposition. Funct Ecol 34:1479–1490. https://doi.org/10.1111/1365-2435.13560
Prieto I, Querejeta JI (2020) Simulated climate change decreases nutrient resorption from senescing leaves. Glob Change Biol 26:1795–1807. https://doi.org/10.1111/gcb.14914
Qiu L, Zhang Y, Mao R, Chen F, Liu J, Yang G, Wan S (2021) Understory removal accelerates nucleic phosphorus release but retards residual phosphorus release in decomposing litter of Phyllostachys edulis in subtropical China. Land Degrad Dev 32:2695–2703. https://doi.org/10.1002/ldr.3942
Ren HY, Qin J, Yan BL, Alata B, Han GD (2018) Mass loss and nutrient dynamics during litter decomposition in response to warming and nitrogen addition in a desert steppe. Front Agric Sci Eng 5:64–70. https://doi.org/10.15302/j-fase-2017194
See CR, Yanai RD, Fisk MC, Vadeboncoeur MA, Quintero BA, Fahey TJ (2015) Soil nitrogen affects phosphorus recycling: foliar resorption and plant-soil feedbacks in a northern hardwood forest. Ecology 96:2488–2498. https://doi.org/10.1890/15-0188.1
Shen Y, Yang X, Sun X, Chen W, Yang G, Liu N, Zhang Y (2018) Increased precipitation modulates the influence of nitrogen and litter inputs on the nutrient resorption proficiency rather than efficiency of Leymus chinensis. Plant Ecol 219:217–230. https://doi.org/10.1007/s11258-017-0790-2
Sorrell BK, Chagué-Goff C, Basher LM, Partridge TR (2011) N:P ratios, δ15N fractionation and nutrient resorption along a nitrogen to phosphorus limitation gradient in an oligotrophic wetland complex. Aquat Bot 94:93–101. https://doi.org/10.1016/j.aquabot.2010.11.006
Sun X, Li D, Lü XT, Fang Y, Ma Z, Wang Z, Chu C, Li M, Chen H (2023) Widespread controls of leaf nutrient resorption by nutrient limitation and stoichiometry. Funct Ecol 37:1653–1662. https://doi.org/10.1111/1365-2435.14318
Tian H, Xu R, Canadell JG, Thompson RL, Winiwarter W, Suntharalingam P, Davidson EA, Ciais P, Jackson RB, Janssens-Maenhout G, Prather MJ, Régnier P, Pan N, Pan S, Peters GP, Shi H, Tubiello FN, Zaehle S, Zhou F et al (2020) A comprehensive quantification of global nitrous oxide sources and sinks. Nature 586:248–256. https://doi.org/10.1038/s41586-020-2780-0
Tong R, Zhou B, Jiang L, Ge X, Cao Y, Shi J (2021) Leaf litter carbon, nitrogen and phosphorus stoichiometry of Chinese fir (Cunninghamia lanceolata) across China. Glob Ecol Conserv 27:e01542. https://doi.org/10.1016/j.gecco.2021.e01542
Trentini C, Villagra M, Pámies DG, Laborde VB, Bedano J, Campanello P (2018) Effect of nitrogen addition and litter removal on understory vegetation, soil mesofauna, and litter decomposition in loblolly pine plantations in subtropical Argentina. For Ecol Manag 429:133–142. https://doi.org/10.1016/j.foreco.2018.07.012
Vergutz L, Manzoni S, Porporato A, Novais RF, Jackson RB (2013) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82:205–220. https://doi.org/10.1890/11-0416.1
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol Appl 20:5–15. https://doi.org/10.1890/08-0127.1
Wan S, Fu S, Zhang C, Liu J, Zhang Y, Mao R (2021) Effects of understory removal and litter addition on leaf and twig decomposition in a subtropical Chinese fir plantation. Land Degrad Dev 32:5004–5011. https://doi.org/10.1002/ldr.4086
Wortman E, Tomaszewski T, Waldner P, Schleppi P, Thimonier A, Eugster W, Buchmann N, Sievering H (2012) Atmospheric nitrogen deposition and canopy retention influences on photosynthetic performance at two high nitrogen deposition Swiss forests. Tellus B Chem Phys Meteorol 64:17216. https://doi.org/10.3402/tellusb.v64i0.17216
Wu JP, Liu ZF, Wang XL, Sun YX, Zhou LX, Lin YB, Fu SL (2011) Effects of understory removal and tree girdling on soil microbial community composition and litter decomposition in two Eucalyptus plantations in South China. Funct Ecol 25:921–931. https://doi.org/10.1111/j.1365-2435.2011.01845.x
Wu JP, Liu W, Zhang W, Shao Y, Duan H, Chen B, Wei X, Fan H (2019) Long-term nitrogen addition changes soil microbial community and litter decomposition rate in a subtropical forest. Appl Soil Ecol 142:43–51. https://doi.org/10.1016/j.apsoil.2019.05.014
Xi D, Jin SF, Wu JP (2022) Soil bacterial community is more sensitive than fungal community to canopy nitrogen deposition and understory removal in a Chinese fir plantation. Front Microbiol 13:1015936. https://doi.org/10.3389/fmicb.2022.1015936
Xia ZC, Kong CH, Chen LC, Wang SL (2015) Allelochemical-mediated soil microbial community in long-term monospecific Chinese fir forest plantations. Appl Soil Ecol 96:52–59. https://doi.org/10.1016/j.apsoil.2015.07.012
Xu S, Zhou GY, Tang XL, Wang WT, Wang GX, Ma KP, Han SJ, Du S, Li SG, Yan JH, Ma YX (2017) Different spatial patterns of nitrogen and phosphorus resorption efficiencies in China’s forests. Sci Rep 7:10584. https://doi.org/10.1038/s41598-017-11163-7
Xu JW, Lin G, Liu B, Mao R (2020) Linking leaf nutrient resorption and litter decomposition to plant mycorrhizal associations in boreal peatlands. Plant Soil 448:413–424. https://doi.org/10.1007/s11104-020-04449-9
Xu L, Xing A, Du E, Shen H, Yan Z, Jiang L, Tian D, Hu H, Fang J (2021) Effects of nitrogen addition on leaf nutrient stoichiometry in an old-growth boreal forest. Ecosphere 12:e03335. https://doi.org/10.1002/ecs2.3335
Yan Z, Kim N, Han W, Guo Y, Han T, Du E, Fang J (2015) Effects of nitrogen and phosphorus supply on growth rate, leaf stoichiometry, and nutrient resorption of Arabidopsis thaliana. Plant Soil 388:147–155. https://doi.org/10.1007/s11104-014-2316-1
Yang Y, Zhang X, Zhang C, Wang H, Fu XL, Chen FS, Wan SZ, Sun XM, Wen XF, Wang JF (2018) Understory vegetation plays the key role in sustaining soil microbial biomass and extracellular enzyme activities. Biogeosciences 15:4481–4494. https://doi.org/10.5194/bg-15-4481-2018
You CM, Wu FZ, Yang WQ, Xu ZF, Tan B, Yue K, Ni XY (2018a) Nutrient-limited conditions determine the responses of foliar nitrogen and phosphorus stoichiometry to nitrogen addition: a global meta-analysis. Environ Pollut 241:740–749. https://doi.org/10.1016/j.envpol.2018.06.018
You C, Wu F, Yang W, Xu Z, Tan B, Zhang L, Yue K, Ni X, Li H, Chang C, Fu C (2018b) Does foliar nutrient resorption regulate the coupled relationship between nitrogen and phosphorus in plant leaves in response to nitrogen deposition? Sci Total Environ 645:733–742. https://doi.org/10.1016/j.scitotenv.2018.07.186
You C, Peng C, Xu Z, Liu Y, Zhang L, Yin R, Liu L, Li H, Wang L, Liu S, Tan B, Kardol B (2021) Nitrogen addition mediates the response of foliar stoichiometry to phosphorus addition: a meta-analysis. Ecol Process 10:58. https://doi.org/10.1186/s13717-021-00329-x
Yu LL, Zhang XH, Shi FX, Mao R (2022) Effect of shrub encroachment on leaf nutrient resorption in temperate wetlands in the Sanjiang Plain of Northeast China. Ecol Process 11:67. https://doi.org/10.1186/s13717-022-00413-w
Zeng Q, Liu Y, Yang J, Chen Y, Ma L, An S (2021) Nutrients of green and senesced leaves of a Robinia pseudoacacia plantation along a latitudinal gradient on the Loess Plateau, China. J For Res 32:2499–2506. https://doi.org/10.1007/s11676-020-01273-w
Zhang S, Yang X, Li D, Li S, Chen Z, Wu J (2022a) A meta-analysis of understory plant removal impacts on soil properties in forest ecosystems. Geoderma 426:116116
Zhang J, Tang ZY, Wang WT, Zhang HF, Liu Y, Xin YH, Zhao LJ, Li H (2022b) Nutrient resorption responses of plant life forms to nitrogen addition in temperate shrublands. Ecosphere 13:e4143. https://doi.org/10.1002/ecs2.4143
Zhang L, Liu J, Yin R, Xu Z, You C, Li H, Wang L, Liu S, Xu H, Xu L, Liu Y, Wang Y, Tan B (2023) Soil fauna accelerated litter C and N release by improving litter quality across an elevational gradient. Ecol Process 12:47. https://doi.org/10.1186/s13717-023-00459-4
Zheng M, Zhang W, Luo Y, Wan S, Fu S, Wang S, Liu N, Ye Q, Yan J, Zou B, Fang C, Ju Y, Ha D, Zhu L, Mo J (2018) The inhibitory effects of nitrogen deposition on asymbiotic nitrogen fixation are divergent between a tropical and a temperate forest. Ecosystems 22:955–967. https://doi.org/10.1007/s10021-018-0313-6
Zhou L, Addo-Danso SD, Wu P, Li S, Zou X, Zhang Y, Ma X (2017a) Leaf resorption efficiency in relation to foliar and soil nutrient concentrations and stoichiometry of Cunninghamia lanceolata with stand development in southern China. J Soils Sediments 16:1448–1459. https://doi.org/10.1007/s11368-016-1352-2
Zhou S, Huang C, Han B, Xiao Y, Tang J, Xiang Y, Luo C (2017b) Simulated nitrogen deposition significantly suppresses the decomposition of forest litter in a natural evergreen broad-leaved forest in the Rainy Area of Western China. Plant Soil 420:135–145. https://doi.org/10.1007/s11104-017-3383-x
Acknowledgements
We thank Dr. William J. Gale from Kansas State University, USA, and Dr. Sharafat Ali from Linnaeus University, Sweden, for their valuable comments and assistance in making modifications to the earlier version of the manuscript.
Funding
This research was funded by National Natural Science Foundation of China (Nos. 32371733, 31570444 and 32201368), the Xingdian Scholar Fund of Yunnan Province, and the Double Top University Fund of Yunnan University.
Author information
Authors and Affiliations
Contributions
Jawad Ali Shah, Jianping Wu and Wenfei Liu conceived the idea, suggestions for data selection, preparation, analysis and interpretation. Jawad Ali Shah and Saif Ullah conducted data analysis. Jianping Wu, Fangfang Shen, Yingchun Liao, contributed ideas to analyses. Jawad Ali Shah and Jianping Wu wrote the manuscript. Jawad Ali Shah, Wenfei Liu, Saif Ullah, Syed Turab Raza, Fangfang Shen, Yingchun Liao, Honglang Duan, Jianping Wu contributed to the shaping of the manuscript, and made edits and suggestions leading to the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict of competing interest for the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1.
A schematic diagram showing various leaf nutrient resorption control strategies under different nutrient limitation scenarios and litter decomposition processes in response to canopy nitrogen (CN) addition and understory ermoval (UR) in Cunninghamia lanceolata plantation. In the uppermost panel, (a) nutrient concentration control, the role of foliar nutrients in influencing resorption patterns is depicted. In the middle panel, (b) nutrient limitation control, certain nutrient restriction has the potential to influence resorption patterns. (c) stoichiometry control, this panel emphasizes the role of foliar N:P ratios in determining how much N versus P are resorbed during senescence. The CN addition follows the nutrient concentration control whereas UR follows the nutrient limitation control. The stoichiometric control is proposed for various relationships of leaf nutrient contents, nutrient resorption and litter decomposition. The CN addition accelerates litter decomposition process by boosting substrate availability for soil biota which in turn results in higher litter nutrient release in soil ecosystem. By contrast UR slows down the process of litter decomposition by decreasing the availability of substrate for soil biota and hence results in slow nutrient release into the soil ecosystem. Figure S2. Relationships between resorption efficiencies of N and P with leaf and litter nutrient contents in Chinese fir plantations. Figure S3. Relationships between N and P concentrations in green leaves ([N]g and [P]g), senesced leaves ([N]s and [P]s), with litter nutrient contents in Chinese fir plantations. Table S1. Results of two-way ANOVAs for leaf nutrient concentrations, leaf N:P ratio, and nutrient resorption parameters sampled in April, as dependent on canopy nitrogen addition (CN), understory removal (UR), and their interaction (CN × UR). Table S2. Results of two-way ANOVAs for leaf nutrient concentrations, leaf N:P ratio, and nutrient resorption parameters sampled in December, as depended on canopy nitrogen addition (CN), understory removal (UR), and their interaction (CN × UR). Table S3. Results of four-way ANOVAs showing the effects of canopy nitrogen addition (CN), understory removal (UR), leaf types {(LT) (Green vs Senesced)}, seasons {(S) (April and December)} and their interactions on N, P concentrations and N:P ratios in a Chinese fir plantation. Table S4. Effects of canopy nitrogen addition (CN), understory removal (UR), season {S (April and December)} and their interactions on leaf nutrient resorption parameters (NRE, and PRE) in a Chinese fir plantation. Table S5. Results of repeated measures ANOVA showing the effects of canopy nitrogen addition (CN), understory removal (UR), sampling dates (T), and their interactions on leaf litter nutrient content of N, P and N:P during 2016–2017.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Shah, J.A., Liu, W., Ullah, S. et al. Linkages among leaf nutrient concentration, resorption efficiency, litter decomposition and their stoichiometry to canopy nitrogen addition and understory removal in subtropical plantation. Ecol Process 13, 27 (2024). https://doi.org/10.1186/s13717-024-00507-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13717-024-00507-7