Abstract
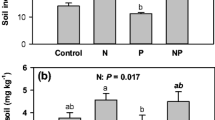
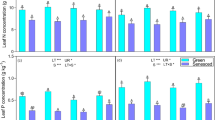
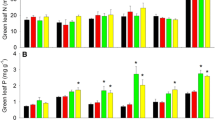
Resorption, the process of withdrawing foliar nutrients prior to leaf abscission, is one of the most important nutrient conservation mechanisms in trees. Along with foliar nutrient concentrations, foliar resorption can be used to infer nutrient limitation. We collected green and senesced leaves of five species in early successional stands in the White Mountains of New Hampshire. In unmanipulated controls, foliar N:P ratios ranged from 20 to 31 and litter N:P ratios ranged from 19 to 36. These values suggest P limitation, although this forest type has been assumed to be N-limited. Additionally, N:P resorption ratios in control plots were < 1, reflecting proportionately more conservation of P through resorption than N. Four years into a full-factorial N × P fertilization experiment, N and P additions had increased N and P concentrations in leaves; more importantly, P addition reduced N concentration, possibly indicating alleviation of growth limitation by P. Resorption of P was less proficient (indicated by the concentration of an element in leaf litter) with P addition, as expected. Resorption proficiency and efficiency (the proportion of leaf nutrients resorbed) of N increased with P addition, suggesting increased demand for N with alleviation of P limitation. Resorption of P was more proficient and efficient with N addition, consistent with exacerbated P limitation. Temperate forests on glaciated soils are generally thought to be N-limited, but long-term N × P manipulations in this biome are lacking. Our results suggest that decades of anthropogenic N deposition may have tipped the balance to P limitation in these forests.







Similar content being viewed by others
References
Aber JD, Goodale CL, Ollinger SV et al (2003) Is nitrogen deposition altering the nitrogen status of northeastern forests? AIBS Bull 53:375–389
Adams MB, Loughry LH, Plaugher LL (2003) Experimental forests and ranges of the USDA forest service
Aerts R (1990) Nutrient use efficiency in evergreen and deciduous species from heathlands. Oecologia 84:391–397
Aerts R (1996) Nutrient resorption from senescing leaves of perennials: are there general patterns? J Ecol 84:597–608
Aerts R, Chapin FS (1999) The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv Ecol Res 30:1–67
Ågren GI (2004) The C:N:P stoichiometry of autotrophs—theory and observations. Ecol Lett 7:185–191. https://doi.org/10.1111/j.1461-0248.2004.00567.x
Bai Y, Wu J, Clark CM et al (2010) Tradeoffs and thresholds in the effects of nitrogen addition on biodiversity and ecosystem functioning: evidence from inner Mongolia Grasslands. Glob Change Biol 16:358–372
Berendse F, Aerts R (1987) Nitrogen-use-efficiency: a biologically meaningful definition? JSTOR 1:293–296
Bloom AJ, Chapin FS III, Mooney HA (1985) Resource limitation in plants-an economic analogy. Annu Rev Ecol Syst 16:363–392
Buchanan-Wollaston V (1997) The molecular biology of leaf senescence. J Exp Bot 48:181–199
Chapin FS, Matson PA, Mooney HA (2002) Terrestrial decomposition. Springer, Berlin
Chapin FS III, Matson PA, Vitousek P (2011) Principles of terrestrial ecosystem ecology. Springer, Berlin
Danger M, Daufresne T, Lucas F et al (2008) Does Liebig’s law of the minimum scale up from species to communities? Oikos 117:1741–1751
Davidson EA, Reis de Carvalho CJ, Vieira IC et al (2004) Nitrogen and phosphorus limitation of biomass growth in a tropical secondary forest. Ecol Appl 14:150–163
dos Santos Jr UM, de Carvalho Gonçalves JF, Feldpausch TR (2006) Growth, leaf nutrient concentration and photosynthetic nutrient use efficiency in tropical tree species planted in degraded areas in central Amazonia. For Ecol Manag 226:299–309
Elser JJ, Bracken ME, Cleland EE et al (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142
Fahey TJ, Battles JJ, Wilson GF (1998) Responses of early successional northern hardwood forests to changes in nutrient availability. Ecol Monogr 68:183–212
Fei S, Steiner KC (2007) Evidence for increasing red maple abundance in the eastern United States. For Sci 53:473–477
Finzi AC, Austin AT, Cleland EE et al (2011) Responses and feedbacks of coupled biogeochemical cycles to climate change: examples from terrestrial ecosystems. Front Ecol Environ 9:61–67
Fisk MC, Ratliff TJ, Goswami S, Yanai RD (2014) Synergistic soil response to nitrogen plus phosphorus fertilization in hardwood forests. Biogeochemistry 118:195–204
Goswami S, Fisk MC, Vadeboncoeur MA et al (2018) Phosphorus limitation of aboveground production in northern hardwood forests. Ecology 99:438–449
Gradowski T, Thomas SC (2006) Phosphorus limitation of sugar maple growth in central Ontario. For Ecol Manag 226:104–109. https://doi.org/10.1016/j.foreco.2005.12.062
Grime JP (1979) Plant strategies and vegetation processes. Plant Strateg Veg Process 206:1176–1177
Güsewell S (2004) N:P ratios in terrestrial plants: variation and functional significance. New Phytol 164:243–266
Haase DL, Rose R (1995) Vector analysis and its use for interpreting plant nutrient shifts in response to silvicultural treatments. For Sci 41:54–66
Hagen-Thorn A, Varnagiryte I, Nihlgard B, Armolaitis K (2006) Autumn nutrient resorption and losses in four deciduous forest tree species. For Ecol Manag 228:33–39
Harpole WS, Ngai JT, Cleland EE et al (2011) Nutrient co-limitation of primary producer communities. Ecol Lett 14:852–862
Hedwall P-O, Bergh J, Brunet J (2017) Phosphorus and nitrogen co-limitation of forest ground vegetation under elevated anthropogenic nitrogen deposition. Oecologia 185:317–326. https://doi.org/10.1007/s00442-017-3945-x
Ji H, Wen J, Du B et al (2018) Comparison of the nutrient resorption stoichiometry of Quercus variabilis Blume growing in two sites contrasting in soil phosphorus content. Ann For Sci. https://doi.org/10.1007/s13595-018-0727-5
Killingbeck KT (1996) Nutrients in senesced leaves: keys to the search for potential resorption and resorption proficiency. Ecology 77:1716–1727
Kobe RK, Lepczyk CA, Iyer M (2005) Resorption efficiency decreases with increasing green leaf nutrients in a global data set. Ecology 86:2780–2792
Koerselman W, Meuleman AF (1996) The vegetation N: P ratio: a new tool to detect the nature of nutrient limitation. J Appl Ecol 1:1441–1450
Marklein AR, Houlton BZ (2012) Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol 193:696–704
McGroddy ME, Daufresne T, Hedin LO (2004) Scaling of C:N:P stoichiometry in forests worldwide: implications of terrestrial redfield-type ratios. Ecology 85:2390–2401
Menge DN, Field CB (2007) Simulated global changes alter phosphorus demand in annual grassland. Glob Change Biol 13:2582–2591
Mohren GMJ, Van Den Burg J, Burger FW (1986) Phosphorus deficiency induced by nitrogen input in Douglas fir in the Netherlands. Plant Soil 95:191–200
NADP Program Office (2017) National Atmospheric Deposition Program
Naples BK, Fisk MC (2010) Belowground insights into nutrient limitation in northern hardwood forests. Biogeochemistry 97:109–121
Niinemets Ü, Kull K (2005) Co-limitation of plant primary productivity by nitrogen and phosphorus in a species-rich wooded meadow on calcareous soils. Acta Oecol 28:345–356
Ostertag R (2010) Foliar nitrogen and phosphorus accumulation responses after fertilization: an example from nutrient-limited Hawaiian forests. Plant Soil 334:85–98
Rastetter EB, Shaver GR (1992) A model of multiple-element limitation for acclimating vegetation. Ecology 73:1157–1174
Rastetter EB, Yanai RD, Thomas RQ et al (2013) Recovery from disturbance requires resynchronization of ecosystem nutrient cycles. Ecol Appl 23:621–642
Reed SC, Townsend AR, Davidson EA, Cleveland CC (2012) Stoichiometric patterns in foliar nutrient resorption across multiple scales. New Phytol 196:173–180
Reich PB, Oleksyn J (2004) Global patterns of plant leaf N and P in relation to temperature and latitude. Proc Natl Acad Sci USA 101:11001–11006
Reich PB, Walters MB, Ellsworth DS (1997) From tropics to tundra: global convergence in plant functioning. Proc Natl Acad Sci 94:13730–13734
Reich PB, Wright IJ, Cavender-Bares J et al (2003) The evolution of plant functional variation: traits, spectra, and strategies. Int J Plant Sci 164:S143–S164
See CR, Yanai RD, Fisk MC et al (2015) Soil nitrogen affects phosphorus recycling: foliar resorption and plant–soil feedbacks in a northern hardwood forest. Ecology 96:2488–2498
Soudzilovskaia NA, Onipchenko VG, Cornelissen JH, Aerts R (2007) Effects of fertilisation and irrigation on ‘foliar afterlife’in alpine tundra. J Veg Sci 18:755–766
Staaf H (1982) Plant nutrient changes in beech leaves during senescence as influenced by site characteristics. ACTA OECOL OECOL PLANT 3:161–170
Townsend AR, Cleveland CC, Asner GP, Bustamante M (2007) Controls over foliar N:P ratios in tropical rain forests. Ecology 88:107–118
Treseder KK, Vitousek PM (2001) Effects of soil nutrient availability on investment in acquisition of N and P in Hawaiian rain forests. Ecology 82:946–954
Tripler C, Canham C, Inouye R, Schnurr J (2002) Soil nitrogen availability, plant luxury consumption, and herbivory by white-tailed deer. Oecologia 133:517–524
US Environmental Protection Agency Clean Air Markets Division (2017) Clean air status and trends network (CASTNET)
Vadeboncoeur MA (2010) Meta-analysis of fertilization experiments indicates multiple limiting nutrients in northeastern deciduous forests. Can J For Res 40:1766–1780
Vadeboncoeur MA, Hamburg SP, Blum JD et al (2012) The quantitative soil pit method for measuring belowground carbon and nitrogen stocks. Soil Sci Soc Am J 76:2241. https://doi.org/10.2136/sssaj2012.0111
Vadeboncoeur MA, Hamburg SP, Yanai RD, Blum JD (2014) Rates of sustainable forest harvest depend on rotation length and weathering of soil minerals. For Ecol Manag 318:194–205
Van Heerwaarden LM, Toet S, Aerts R (2003) Nitrogen and phosphorus resorption efficiency and proficiency in six sub-arctic bog species after 4 years of nitrogen fertilization. J Ecol 91:1060–1070
Venterink HO, Güsewell S (2010) Competitive interactions between two meadow grasses under nitrogen and phosphorus limitation. Funct Ecol 24:877–886
Vergutz L, Manzoni S, Porporato A et al (2012) Global resorption efficiencies and concentrations of carbon and nutrients in leaves of terrestrial plants. Ecol Monogr 82:205–220
Vitousek PM (1984) Litterfall, nutrient cycling, and nutrient limitation in tropical forests. Ecology 65:285–298
Vitousek PM, Sanford RL Jr (1986) Nutrient cycling in moist tropical forest. Annu Rev Ecol Syst 17:137–167
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol Appl 20:5–15
Walker TW, Syers JK (1976) The fate of phosphorus during pedogenesis. Geoderma 15:1–19
Wood Tana E, Deborah Lawrence, Wells Jessie A (2011) Inter-specific variation in foliar nutrients and resorption of nine canopy-tree species in a secondary neotropical rain forest. Biotropica 43:544–551. https://doi.org/10.1111/j.1744-7429.2010.00740.x
Wright IJ, Reich PB, Westoby M et al (2004) The worldwide leaf economics spectrum. Nature 428:821–827
Yanai RD (1992) Phosphorus budget of a 70-year-old northern hardwood forest. Biogeochemistry 17:1–22
Yang Y, Yanai RD, Fatemi FR et al (2016) Sources of variability in tissue chemistry in northern hardwood species. Can J For Res 46:285–296. https://doi.org/10.1139/cjfr-2015-0302
Yuan ZY, Chen HY (2015) Negative effects of fertilization on plant nutrient resorption. Ecology 96:373–380
Acknowledgements
This research was conducted on plots established by Matt Vadeboncoeur, Steve Hamburg, and others almost 15 years ago under funding to Yanai from the National Science Foundation (DEB 0235650). Beginning in 2010, Multiple Element Limitation in Northern Hardwood Ecosystems (http://www.esf.edu/melnhe) was funded (by DEB 0949324 to Yanai), guided by the early vision of Tim Fahey and Melany Fisk. Maintaining the sites and fertilizing them has involved 69 summer interns and 33 graduate students. We are especially grateful to Bali Quintero, Adam Wild, and Nora Yanai, who shot and processed green leaves in August 2014. Jerome Barner, Stephanie Suttenberg, and Alex Kuhn helped collect leaf litter in fall 2014. The paper was improved by comments from Keith Killingbeck, Craig See, and students in FOR 694 at SUNY-ESF. The Bartlett Experimental Forest is owned and operated by the USDA Forest Service. This paper is a product of the Hubbard Brook Long-Term Ecological Research network, funded by the National Science Foundation (DEB 1633026).
Funding
This study was funded by the National Science Foundation (DEB 0235650 and 0949324).
Author information
Authors and Affiliations
Contributions
The idea for this study was part of a much larger project led by RDY. KEG joined the project in her final semester as an undergraduate student, collecting samples and processing them. As a graduate student, KEG analyzed the data. KEG and RDY interpreted the results, wrote the paper, and enhanced its value through extended revision.
Corresponding author
Additional information
Communicated by Jason P. Kaye.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gonzales, K., Yanai, R. Nitrogen–phosphorous interactions in young northern hardwoods indicate P limitation: foliar concentrations and resorption in a factorial N by P addition experiment. Oecologia 189, 829–840 (2019). https://doi.org/10.1007/s00442-019-04350-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-019-04350-y