Abstract
Background
Alginate oligosaccharides (AOs) are the degradation products of alginate, a natural polysaccharide abundant in brown algae. AOs generated by enzymatic hydrolysis have diverse bioactivities and show broad application potentials. AOs production via enzymolysis is now generally with sodium alginate as the raw material, which is chemically extracted from brown algae. In contrast, AOs production by direct degradation of brown algae is more advantageous on account of its cost reduction and is more eco-friendly. However, there have been only a few attempts reported in AOs production from direct degradation of brown algae.
Results
In this study, an efficient Laminaria japonica-decomposing strain Pseudoalteromonas agarivorans A3 was screened. Based on the secretome and mass spectrum analyses, strain A3 showed the potential as a cell factory for AOs production by secreting alginate lyases to directly degrade L. japonica. By using the L. japonica roots, which are normally discarded in the food industry, as the raw material for both fermentation and enzymatic hydrolysis, AOs were produced by the fermentation broth supernatant of strain A3 after optimization of the alginate lyase production and hydrolysis parameters. The generated AOs mainly ranged from dimers to tetramers, among which trimers and tetramers were predominant. The degradation efficiency of the roots reached 54.58%, the AOs production was 33.11%, and the AOs purity was 85.03%.
Conclusion
An efficient, cost-effective and green process for AOs production directly from the underutilized L. japonica roots by using strain A3 was set up, which differed from the reported processes in terms of the substrate and strain used for fermentation and the AOs composition. This study provides a promising platform for scalable production of AOs, which may have application potentials in industry and agriculture.
Similar content being viewed by others
Background
Brown algae, including Laminaria japonica, Laminaria digitata, Undaria pinnatifida, Macrocystis pyrifera, Ascophyllum nodosum, Sargassum natans, and others, are a large group of marine seaweeds (~ 1800 species) with huge biomass [1, 2]. They have aroused a wide interest by virtue of their rapid growth, superior nutritional value and absence of recalcitrant lignin [3, 4]. Among brown algae, L. japonica is an important primary producer in the coastal ocean and is cultivated as a popular edible alga worldwide, especially in East Asia [5]. In food processing, while L. japonica blade is always in demand, its so-called “root”, which includes meristem and stipe, is usually discarded in the food industry, causing a waste of resources. Thus, techniques for the high-value use of L. japonica roots are worth exploring.
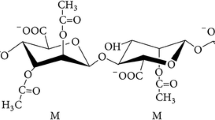
Alginate is an acid polysaccharide, consisting of mannuronic acid (M) and its epimer guluronic acid (G) linked via 1,4-O-glycosidic bonds. It is abundant in various brown algae, accounting for 30–60% of the dry weight [6], and thus is a significant marine carbon source. Alginate is frequently employed as a thickener, stabilizer, and gel-forming agent [7]. It is also the raw material for the production of alginate oligosaccharides (AOs).
AOs are the depolymerization products of alginate with low degrees of polymerization (DPs), which have recently received widespread attention because of their diverse bioactivities. As reported, AOs have various physiological activities, including antitumor, antioxidant, prebiotic activities, and others [8,9,10], and thus have broad therapeutic potentials. In agriculture, AOs can promote the root growth, improve the tolerance to drought and induce the immunity of plants [11,12,13]. In particular, AOs at DP2-4 have been shown to have a variety of bioactivities on plants [14], for instance, promoting the root development in Triticum aestivum L [15]. and increasing the yield of flowering Chinese cabbage [16]. The conventional method of AOs production is to hydrolyze alginate by heating in the presence of acid, such as hydrochloric acid, sulfuric acid and formic acid [17]. Acid hydrolysis is a rapid method with violent chemical reaction, leading to the directly break of glycosidic bonds between the C1 and C4 in alginate [18]. The use of acids and the requirement for high temperatures result in environmental pollution and energy waste [19]. By comparison, AOs production via enzymolysis using alginate lyases is milder, more environment-friendly and energy-saving as no acids or intense heating is required. Moreover, AOs derived from enzymatic degradation are unsaturated oligomers with lower DPs, which have stable structures and more biological activities [18]. As reported, unsaturated AOs prepared by enzymolysis have excellent antioxidant activity and better immunomodulatory effect on murine macrophage RAW264.7 cells compared with AOs prepared by acid hydrolysis [20, 21]. Thus, the enzymatic approach is an advantageous method for AOs preparation.
Alginate lyases, a class of polysaccharide lyases (PLs) distributed in 14 families in the Carbohydrate-Active enZYmes Database (http://www.cazy.org/), cleave the 1,4-glycosidic bonds between alginate monomers via the β-elimination reaction, leading to the formation of an unsaturated double bond at the non-reducing end [22]. So far, the conventional enzymatic approach for AOs production is to use alginate lyases to degrade sodium alginate. However, due to the multiple steps required to extract sodium alginate from brown algae [23], the high cost of using sodium alginate as substrate for the industrial AOs preparation is a non-negligible issue. Accordingly, producing AOs by directly degrading brown algae is more advantageous on account of its cost reduction. Till now, only 3 studies have reported the direct production of AOs from brown algae. Li et al. provided an approach for preparing AOs directly from brown seaweed via a combination of enzymatic hydrolysis and selective fermentation by utilizing an engineered Yarrowia lipolytica strain [24]. Wang et al. isolated an alginate lyase-producing Bacillus litoralis strain that can degrade Sargassum horneri to produce AOs [25]. Additionally, the alginate lyase AlgL7 from Microbulbifer sp. ALW1 that was heterologously expressed in Escherichia coli, was capable of producing AOs from L. japonica [26]. Therefore, more approaches for preparing AOs directly from brown seaweeds, which are simple, economic and efficient, need to be developed.
In this study, we screened a strain Pseudoalteromonas agarivorans A3 from L. japonica collected from a kelp farm in Weihai, China. Strain A3 can directly decompose L. japonica mainly relying on its secreted alginate lyases. We optimized the fermentation conditions for alginate lyase production of strain A3 and the hydrolysis parameters of the fermentation broth supernatant (FBS) on L. japonica roots. Moreover, we characterized the degradation products of L. japonica roots by the FBS. Based on our data, an efficient, cost-effective and green process for AOs production directly from L. japonica roots by utilizing the FBS of strain A3 was set up, providing a new enzymatic method for AOs production.
Results and discussion
Screening of an effective L. japonica-decomposing bacterial strain
To obtain bacteria with high L. japonica-decomposing ability, bacterial strains with alginate-hydrolyzing ability were first isolated from the L. japonica sample by detecting the clear halo around the bacterial colony on the screening plates containing alginate as the sole carbon source. Then, to further verify the utilization of alginate, 5 strains with a high ratio of the halo diameter (HD) to the colony diameter (CD) (the HD/CD ratio ≧ 3) were cultured in the liquid medium with sodium alginate as the sole carbon source. Among them, strain A3 showed the highest HD/CD ratio (5.3, Fig. 1A) and the fastest growth in the liquid medium, reaching the stationary phase after 12 h (Fig. 1B). When cultured in the medium containing a piece of L. japonica blade and 3% (w/v) sea salts, strain A3 showed the highest L. japonica-decomposing ability. After 12 h-cultivation, the L. japonica piece was completely degraded into debris (Fig. 1C), indicating that strain A3 is an effective L. japonica-decomposing strain. Thus, strain A3 was chosen for further study and its genome was sequenced.
Screening of the L. japonica-decomposing bacterial strain Pseudoalteromonas agarivorans A3. A the degradation of alginate by strain A3. Strain A3 was cultured on the screening plate at 25 °C for 5 days and the plate were dyed with Lugol’s solution to determine the degradation of alginate around the strain colony. B the growth curve of strain A3 cultured in the liquid medium containing 0.5% sodium alginate as the sole carbon source. The growth curve of strain A3 cultured in the liquid medium without sodium alginate was taken as the control. C decomposition of L. japonica by strain A3. Strain A3 was cultured at 25 °C in the medium containing a piece of L. japonica blade and 3% sea salts. The decomposition of L. japonica was observed at 0, 6, 12 and 24 h. The L. japonica blade-containing medium without strain A3 incubated at 25 °C, 180 rpm for 24 h was taken as the control. D the neighbor-joining phylogenetic tree of strain A3 based on the 16S rRNA gene sequences. The bootstrap values of each branch were tested by 1000 repetitions. E transmission electron micrograph of negatively stained cells of strain A3 cultured in 2216E marine broth at 25 °C for 12 h. F LTQ-Orbitrap-MS analysis of the degradation products of the L. japonica piece by the fermentation broth supernatant of strain A3. DP degree of polymerization, AO alginate oligosaccharide
Phylogenetic analysis based on 16S rRNA gene sequences revealed that strain A3 was affiliated with the Pseudoalteromonas genus (Fig. 1D), sharing the highest 16S rRNA gene sequence similarity (99.93%) with that of Pseudoalteromonas agarivorans DSM 14585, a strain capable of decomposing algal polysaccharides such as agar, alginate and carrageenan [27]. Transmission electron microscopy (TEM) analysis showed that cells of strain A3 were rod-shaped with a single polar flagellum, approximately 1.6–2.4 μm in length and 0.8–1 μm in width (Fig. 1E), which resembles the cell morphology of P. agarivorans DSM 14585 [27]. Therefore, strain A3 is a novel strain of Pseudoalteromonas agarivorans, designated as Pseudoalteromonas agarivorans A3.
Several brown alga-decomposing bacterial strains have been reported. Geobacillus thermodenitrificans OS27 degraded the brown alga Undaria pinnatifida into small pieces after 5 days [28] and Microbulbifer sp. 6532 A after 2 days [29]. Pseudoalteromonas sp. Alg6B showed obvious hydrolysis of L. japonica, transforming L. japonica pieces into particles after 24 h [30]. Comparatively, strain A3 showed a stronger L. japonica-decomposing ability, suggesting that strain A3 may have a potential in L. japonica degradation in industry.
Ability of strain A3 to produce AOs directly from L. japonica via its secreted alginate lyases
Represented by alginate, various polysaccharides are the major components of L. japonica. In the genome of strain A3, a number of genes encoding putative carbohydrate-active enzymes (CAZymes) were observed, including dozens of genes encoding enzymes responsible for the degradation of algae-specific polysaccharides (e.g. fucoidan, laminarin and alginate) and other plant polysaccharides (e.g. cellulose and starch). To investigate the enzymes secreted by strain A3 involved in the L. japonica decomposition, we analyzed the secretome of strain A3 cultured with L. japonica as the sole carbon source. In the secretome, 9 CAZymes were detected, including 4 glycoside hydrolases (GHs) and 5 PLs (Table 1). The 4 GHs belong to the GH13 and GH16 families. Thereinto, the 2 GH13 enzymes (OIZ54_07165 and OIZ54_18130) share the highest sequence similarities with an α-amylase (GenBank: P29957.3) and a pullulanase (GenBank: P07206.2) [31, 32], respectively, indicating that they may be involved in the degradation of the starch in L. japonica. A putative GH16 laminarinase (OIZ54_02445) was detected, which may hydrolyze the abundant laminarin in L. japonica. Another detected GH16 enzyme (OIZ54_00850) shares the highest sequence similarity with an agarase based on the amino acid sequence. Remarkably, all of the 5 putative alginate lyases of strain A3 were detected in the secretome, which are from the PL6, -7, -17, and -18 families (Table 1). Among them, the PL18 alginate lyase was the most abundant (48.90%), and the 5 alginate lyases had a total abundance of 70.88% (Table 1). These results suggested that the alginate lyases secreted by strain A3 played a crucial role in the L. japonica decomposition.
To investigate whether strain A3 can decompose L. japonica with the secreted enzymes to produce AOs, we utilized the FBS of strain A3 cultured with L. japonica to degrade L. japonica blade and analyzed the degradation products by LTQ-Orbitrap-MS. The main peaks all correspond to the molecular masses of unsaturated AOs (including dimers, trimers, tetramers and pentamers) (Fig. 1F). The degradation products of starch and laminarin, namely glucose monomers or oligomers, were not observed, which may be due to the low contents of starch and laminarin in L. japonica and the small amount of starch- and laminarin-degrading enzymes in the FBS of strain A3. These results showed that the products released from L. japonica by the FBS of strain A3 were mainly AOs. Thus, strain A3 may have a potential in AOs production from brown algae in industry.
Optimization of the alginate lyase production of strain A3 by single factor and response surface optimization (RSM) methods
Single factor assays were first performed to optimize the alginate lyase production of strain A3. The effects of fermentation temperature and time, L. japonica root powder content, and inorganic nitrogen sources on the alginate lyase production of strain A3 were investigated. The highest alginate lyase production of strain A3 cultured at 10 °C, 15 °C and 20 °C were almost identical, higher than that at 25 °C or 30 °C, and strain A3 reached the highest alginate lyase production the fastest (~ 12 h) at 20 °C (Fig. 2A). Thus, 20 °C and 12 h were chosen as the optimum fermentation temperature and time, respectively. In addition, for the alginate lyase production of strain A3, the optimum content of L. japonica root powder was 0.75% (Fig. 2B). Among the 4 inorganic nitrogen sources, NH4Cl, NH4NO3 and (NH4)2SO4 had promoting effects on the alginate lyase production of strain A3, and NH4NO3 was the optimal one (Fig. 2C). In contrast, the growth of strain A3 was seriously inhibited by NH4HCO3 with unknown reasons, resulting in a significant decrease in the alginate lyase production (Fig. 2C). After optimization, the optimal concentration of NH4NO3 for the alginate lyase production was 2% (Fig. 2D).
Optimization of the fermentation conditions of strain A3 for alginate lyase production by single factor method. A effects of fermentation temperature and time on the alginate lyase production. Strain A3 was cultured at 10, 15, 20, 25 or 30 °C in the basic fermentation medium. B effect of the content of L. japonica root powder on the alginate lyase production. Strain A3 was cultured in the medium containing 3% sea salts and different concentrations of L. japonica root powder (0.1, 0.25, 0.5, 0.75, 1 or 1.25%) at 20 °C for 24 h. C effects of inorganic nitrogen sources on the alginate lyase production. Strain A3 was cultured in the basic fermentation medium with an addition of 0.13% (w/v) NH4Cl, NH4HCO3, NH4NO3 or (NH4)2SO4 at 20 °C for 24 h. The alginate lyase production of strain A3 cultured in the basic fermentation medium at 20 °C for 24 h was taken as the control. D effect of the concentration of NH4NO3 on the alginate lyase production. Strain A3 was cultured in the basic fermentation medium containing different concentrations of NH4NO3 (0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5 or 4%) at 20 °C for 24 h
Based on the single factor experimental results, the fermentation parameters were further optimized by RSM, including the Plackett–Burman (PB) design, the steepest ascent (descent) experiment and the central composite design (CCD). In the PB design, 8 variables (NH4NO3 concentration, rotation speed, L. japonica root powder content, fermentation time, fermentation temperature, pH, inoculum size, and sea salt concentration) were investigated to determine the significant factors affecting the alginate lyase production of strain A3 (Table 2). Based on the analysis of variance (ANOVA) of the PB design results, the p-values of NH4NO3 concentration, fermentation temperature, pH and inoculum size were less than 0.05 (Additional file 1: Table S1), indicating that these 4 variables were the significant factors affecting the alginate lyase production and were selected for further optimization by performing the steepest ascent (descent) experiment [33]. The steepest ascent (descent) experiment started from the center point of the 4 key variables in the PB design and moved along the path, in which NH4NO3 concentration, pH and inoculum size increased, while fermentation temperature decreased. The highest response (8.56 U/ml) was reached at the experiment with 2% NH4NO3, 19.5 °C, pH 8.0 and 1.75% inoculum size, indicating that this point was closed to the maximum response region of alginate lyase production (Table 3). CCD was used to determine the optimal level of each key variable and the effect of their interactions for the alginate lyase production of strain A3. Thirty experiments were performed (Table 4), and the equation was established by multiple regression analysis (Additional file 1: Formula S1).
The ANOVA results showed that the determination coefficient R2, Model F-value, Lack of Fit p-value of the model were 0.9527, 21.57, and 0.3244, respectively, and the p-value of the model was less than 0.0001, suggesting that the regression equation had nice fitting result and this model had a high significance (Additional file 1: Table S2). As shown in Fig. 3, except for the contour map of response surface between NH4NO3 concentration and pH, those between other variables were elliptical or saddle, demonstrating that the interactions between the corresponding variables were significant. Based on the analysis using Design Expert 12 software, the maximum alginate lyase production was predicted to be 8.57 U/ml under the optimum conditions (2.03% NH4NO3, 0.6% L. japonica root powder, 3% sea salt, 2% inoculum size, pH 8.04, 19.56 °C and 120 rpm for 36 h). To validate the feasibility of the model, the fermentation of strain A3 was performed under the predicted optimum conditions. The alginate lyase production reached the maximum 8.77 ± 0.16 U/ml (649.76 ± 8.28 U/ml detected by the ultraviolet absorption (UA) method), approximately 10.4 times higher than that before optimization (0.84 ± 0.097 U/ml), indicating a good accordance with the prediction production and an effective optimization (Fig. 4).
Three-dimensional plots of the effect of four variables on the alginate lyase production of strain A3. A interaction of fermentation temperature and NH4NO3 concentration. B Interaction of pH and NH4NO3 concentration. C Interaction of inoculum size and NH4NO3 concentration. D Interaction of pH and fermentation temperature. E Interaction of inoculum size and fermentation temperature. F Interaction of inoculum size and pH. When the effect of 2 variables was plotted, the other variables were set at the central level
The alginate lyase production of strain A3 cultured under the conditions before and after optimization. Before optimization, strain A3 was cultured in the medium (pH 8.0) containing 1% (w/v) L. japonica root powder and 3% sea salts with 1% inoculum size at 20 °C, 180 rpm for 24 h. After optimization, strain A3 was cultured in the medium (pH 8.04) containing 2.03% NH4NO3, 0.6% L. japonica root powder and 3% sea salt with 2% inoculum size at 19.6oC, 120 rpm for 36 h. The graphs show data from triplicate experiments (mean ± S.D.)
Up to now, many alginate-degrading strains have been reported. Wang et al. isolated a strain Bacillus litoralis M3 that could secrete alginate lyases to degrade alginate. When strain M3 was cultured in the medium containing sodium alginate, the enzyme activity of the FBS was 0.4079 U/ml (the UA method) [25]. Li et al. screened an alginate-degrading strain Pseudoalteromonas sp. SM0524. The alginolytic activity of its fermentation broth reached 62.61 U/ml (the UA method) [34]. In addition, Zhu et al. screened 26 alginate-metabolizing strains, and the strain Microbulbifer sp. ALW1 showed the highest extracellular alginate lyase activity, reaching 0.18 U/ml (the DNS method) [35]. The alginate lyase production of strain A3 was much higher than that of the above strains, indicating that the FBS of strain A3 was a promising enzyme cocktail for the AOs preparation. Moreover, after optimization, the fermentation medium only contains NH4NO3, seawater, and L. japonica roots that are underutilized in the food industries, which is simple, accessible and low-cost, providing feasibility for industrial production of the FBS of strain A3.
Optimization of the hydrolysis parameters of the FBS of strain A3 for AOs preparation from L. japonica roots
To investigate the optimal hydrolysis conditions on L. japonica roots of the FBS, 3 enzymatic hydrolysis parameters, enzyme-substrate ratio (E/S ratio), hydrolysis time and hydrolysis temperature, were optimized. The hydrolysis efficiency reached the maximum when the E/S ratio was ≧ 25 U/g and the hydrolysis time was ≧ 3 h (Fig. 5A, B). The optimal temperature for the hydrolysis of L. japonica root powder by the FBS of strain A3 was 45 °C (Fig. 5C). Under the optimal conditions for hydrolysis (25 U/g for the E/S ratio, 3 h for hydrolysis time and 45 °C for hydrolysis temperature), the hydrolysis efficiency of L. japonica root powder reached 54.58 ± 0.79%, indicating that the L. japonica roots were efficiently degraded by the FBS of strain A3.
Optimization of the enzymatic hydrolysis parameters of L. japonica roots by the fermentation broth supernatant (FBS) of strain A3. A effect of E/S ratio on the hydrolysis efficiency. L. japonica roots were hydrolyzed by the FBS with different E/S ratios (0, 0.05, 0.15, 0.3125, 0.625, 6.25, 12.5, 25, 37.5, 50 or 62.5 U/g) at 45 °C. B effect of hydrolysis time on the hydrolysis efficiency. L. japonica roots were hydrolyzed by the FBS from 5 min to 9 h with an E/S ratio of 25 U/g. C effect of hydrolysis temperature on the hydrolysis efficiency. Hydrolysis of the FBS on L. japonica roots was performed at different temperatures (10, 20, 30, 40, 45, 50, 60 or 70 °C) with an E/S ratio of 25 U/g for 3 h
To characterize the hydrolysate generated from the hydrolysis of L. japonica roots by the FBS, the composition of the hydrolysate was analyzed by high performance liquid chromatography (HPLC) and LTQ-Orbitrap-MS. HPLC result indicated that the hydrolysate consisted of 4 components (Fig. 6A). Based on subsequent LTQ-Orbitrap-MS analysis, the 3 main components were identified as alginate dimers, trimers and tetramers (Fig. 6B–D). Another product, which was too small to be collected for a further LTQ-Orbitrap-MS analysis, corresponded to alginate pentamers based on the alginate oligomer standards (Fig. 6A). Among the products, alginate trimers and tetramers were the most abundant, accounting for 39.62% and 34.60% of the product amount, respectively, followed by alginate dimers (20.69%) and alginate pentamers (5.08%) (Fig. 6A).
Analyses of the degradation products of L. japonica roots by the fermentation broth supernatant (FBS) of strain A3. A HPLC analysis of the degradation products. L. japonica roots treated with the pre-inactivated FBS was taken as the control. Mannuronate oligomers from DP1 to DP6 were taken as the standards. The percentage numbers indicate the percentage of the AO area to the total chromatograph area of the AOs. B–D LTQ-Orbitrap-MS analysis of the major peaks in (A) in the negative-ion mode. DP degree of polymerization, AO alginate oligosaccharide
Till now, only 2 studies have reported the AOs preparation directly from brown algae by fermentation of microbial strains [24, 25]. The engineered Yarrowia lipolytica strain with an alginate lyase gene was reported to produce AOs with DP2 and DP3 after 72 h-fermentation in the L. japonica liquid medium pretreated by enzymes [24]. An alginate lyase-producing Bacillus litoralis strain can degrade Sargassum horneri water extract pretreated by autoclave to produce AOs mainly at DP2 after 12 h-fermentation [25]. In our study, L. japonica roots were used in AOs preparation. Compared with L. japonica, the cost of using L. japonica roots as the raw material is lower, and no pretreatment except for milling is necessary, saving energy. In addition, the preparation of AOs by the FBS of strain A3 is time-saving, requiring only 3 h. AOs with different DPs have been reported to have distinct bioactivities [36]. AOs produced from the L. japonica roots by the FBS of strain A3 are different from those produced in the reports above, indicating that they may have different functions. Especially, the AOs produced by the FBS of strain A3 may be applied in agriculture, as the bioactivities of AOs at DP2-4 have been extensively investigated in plants [14,15,16]. In addition, the advantages of the low fermentation cost and short time for AOs preparation by strain A3 are also beneficial for the large-scale agricultural usage of the prepared AOs.
The efficiency of the process for AOs preparation from L. japonica roots with the FBS on the laboratory scale
Based on the above results, a process for AOs preparation from L. japonica roots with the FBS of strain A3 on the laboratory scale were established (Fig. 7). In the process, the hydrolysis efficiency of L. japonica roots reached 54.58 ± 0.79%, as described above. The AOs production, the total sugar production and the AOs purity were then determined to characterize the L. japonica root hydrolysate obtained by the established process.
On account of the vast majority of uronic acids present in alginate in L. japonica [24], the AOs production was measured with the sulfamate/m-hydroxydiphenyl method, a common method for the determination of uronic acids [37]. The AOs production was determined to be 331.08 ± 8.20 mg/g L. japonica roots (Table 5), accounting for 80.36% of the total amount (41.20%) of alginate in the L. japonica roots, which indicated that the majority of alginate in L. japonica roots was degraded by the FBS in the process. Compared with the AOs production (131 mg/g L. japonica) of the alginate lyase AlgL7 on L. japonica [26], the AOs production of the FBS of strain A3 on L. japonica roots was much higher. The total sugar production was 389.36 ± 20.20 mg/g L. japonica roots. Thus, the purity of the prepared AOs, referring to the proportion of the AOs production to the total sugar production, was 85.03% (Table 5). These results showed that substantial AOs with considerable AOs purity were produced by the process. Therefore, the process is feasible for AOs production directly from L. japonica roots.
Conclusion
Herein, we screened a strain Pseudoalteromonas agarivorans A3 that can efficiently decompose L. japonica to generate AOs mainly relying on its secreted alginate lyases. By optimizing the alginate lyase production of the FBS and the hydrolysis parameters, both of which used the underutilized L. japonica roots in the food industry as raw material, we established an efficient, cost-effective and green process for AOs production. In this process, L. japonica roots were hydrolyzed efficiently and substantial AOs mainly at DP2-4 with considerable purity were obtained. The developed process is a practical process for AOs production directly from L. japonica roots with strain A3, which may have a promising application potential in industry and agriculture. This represents the first study on AOs production from direct degradation of L. japonica roots, which converts the leftover material into a high-value compound.
Materials and methods
Materials
The sun-dried L. japonica, including the roots and the blades, was obtained from the local seafood market (36.08° N, 120.35° E), which was first soaked and washed with water to remove dirt and salt. The washed L. japonica roots were then dried and milled into powder. The L. japonica root powder was passed through an 80-mesh sieve, and stored at room temperature until use. The content of alginate in the dried L. japonica blade and L. japonica root used in this study was determined to be 44.40% and 41.20%, respectively, according to the previously reported extraction method of alginate [38]. Sodium alginate was purchased from Sinopharm Co., Ltd (China). AO standards (purity ≧ 97%) were purchased from Zzstandard (China). Glucose, D-glucuronic acid and sea salt were purchased from Sigma (USA). The TEM 200 copper grid was purchased from Beijing Zhongjingkeyi Technology Co., Ltd (China).
Screening of L. japonica-decomposing bacteria
The fresh L. japonica sample was collected from a kelp farm in Weihai, China and stored in a sterile collection bag in ice for 3 days before being further processed. The sample (20 g) was cut into small pieces (1 cm × 1 cm) and put in 200 ml sterile seawater containing 3% (w/v) sea salts in a 500 ml flask, which was shaken at 180 rpm and 25 °C for 15 min. The resultant solution was serially diluted to 10−4 to 10−6 dilution and then each diluted solution was spread on the screening plates containing a medium composed of 0.5% (w/v) sodium alginate, 3% sea salts and 1.5% (w/v) agar [39]. The plates were incubated at 25 °C.
After incubation at 25 °C for 3–5 days, morphologically distinct colonies emerged on the plates, which were then separated by repeatedly streaking on the 2216E marine agar plates to obtain the purified isolates. The obtained isolates were re-streaked on the screening plates, incubated at 25 °C for 5 days and stained by Lugol’s solution [40]. The HD/CD ratio was used as the standard for a preliminary evaluation of the extracellular alginolytic activity of the isolates. Isolates with a HD/CD ratio ≧ 3 were then inoculated into the liquid medium containing 0.5% sodium alginate and 3% sea salt, and cultured at 25 °C for 24 h to further confirm the alginate-degrading ability. The screened alginate-degrading strains were inoculated into the 4 ml medium containing 3% sea salts and a piece of L. japonica blade (1 cm × 1 cm) and cultured at 180 rpm and 25 °C. The decomposition of L. japonica was observed at 0, 6, 12 and 24 h. Strain A3 showing the highest L. japonica-decomposing ability was screened for further study.
TEM analysis
Strain A3 was cultured in 2216E marine broth at 25 °C, 180 rpm for 12 h. The cells were collected and washed three times with sterile seawater. The cells were then transferred to a TEM 200 copper grid and incubated for 2 min. The grid was dried, negatively stained with 3% (w/v) aqueous uranyl acetate for 1.5 min. After air drying, the cells were observed and photographed using a Tecnai G2 F20 microscope (USA, FEI).
Alginate lyase assay
The alginolytic activity of the FBS was measured by the dinitrosalicylic acid (DNS) method [41] with glucose as the standard. A 200-µl reaction mixture containing 2 mg/ml sodium alginate, 0.5 M NaCl and 20 µl FBS in 50 mM Tris-HCl (pH 8.0) was incubated at 45 °C for 15 min. Then, the reaction was terminated after the addition of 100 µl DNS. The reaction mixture was boiled for 10 min for color development. After that, the absorbance of the mixture at 540 nm was measured. One unit (U) of enzyme activity was defined as the amount of enzyme required to release 1 µmol reducing sugars per min. The reaction system containing the pre-inactivated FBS under the same conditions was taken as the control.
Alginolytic activity of the optimized FBS was also determined by the UA method [42]. The reaction mixture containing 50 mM Tris-HCl (pH 8.0), 2 mg/ml sodium alginate, 0.5 M NaCl and 20 µl FBS was incubated at 45 °C for 15 min. The reaction was then terminated by boiling the mixture for 10 min. The increase in the absorbance at 235 nm (A235) resulted from the release of unsaturated uronic in the mixture was monitored. One unit (U) of enzyme activity was defined as the amount of enzyme required to cause an increase of 0.1 at 235 nm per minute.
Optimization of the fermentation conditions of strain A3 for alginate lyase production
Optimization with single factor method
To determine the optimal fermentation temperature and time for alginate lyase production, strain A3 was cultured for different times (0, 3, 6, 9, 12, 24, 36, 48, 60 or 72 h) at 10, 15, 20, 25 or 30 °C, 180 rpm in 50 ml basic fermentation medium containing 1% (w/v) L. japonica root powder and 3% sea salts. To optimize the content of L. japonica root powder in the fermentation medium, strain A3 was inoculated into the medium containing 3% sea salts and different concentrations of L. japonica root powder (0.1, 0.25, 0.5, 0.75, 1 or 1.25%) and cultured at 20 °C, 180 rpm for 24 h. To determine the optimal inorganic nitrogen source, an additional 0.13% (w/v) nitrogen source, NH4Cl, NH4HCO3, NH4NO3 or (NH4)2SO4, was added into the basic fermentation medium. To determine the optimum concentration of NH4NO3, NH4NO3 was added into the basic fermentation medium at different final concentrations (0, 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5 or 4%). Strain A3 was cultured in the media at 20 °C, 180 rpm for 24 h. After fermentation, the alginate lyase activity of the FBS was determined.
Optimization with RSM
RSM was further performed based on the single factor trials. The designation of the experiments and the data analysis were conducted by using the Design Expert 12 software.
In the PB design, the first step of RSM, 11 variables, including NH4NO3 concentration, rotation speed, L. japonica roots concentration, fermentation time, fermentation temperature, pH, inoculum size, sea salt concentration and 3 dummy variables, were selected in 12 experiments. Each variable was tested at low (− 1) and high (+ 1) levels (Table 2) [43]. The path of steepest ascent (descent) experiment started from the center point (“0” level) in the PB design and the moving directions of significant variables were related to the regression coefficients obtained by the PB design (Table 3) [44]. The levels where the alginate lyase production peaked were taken as “0” level for the following CCD. CCD with 5 coded levels (“− 2”, “− 1”, “0”, “+ 1”, and “+ 2”) was used for obtaining the true optimal fermentation conditions. A total of 30 experiments were performed with 6 replications at level “0” (Table 4), and the results were analyzed using a second-order equation (Additional file 1: Formula S2).
Optimization of the hydrolysis parameters of the FBS on L. japonica roots
To determine the optimal E/S ratio, 0.16 g L. japonica root powder was reacted with the FBS with different E/S ratios (0, 0.05, 0.15, 0.3125, 0.625, 6.25, 12.5, 25, 37.5, 50 or 62.5 U/g) for 12 h. To determine the optimal hydrolysis time, L. japonica root powder was hydrolyzed by the FBS from 5 min to 9 h with an E/S ratio of 25 U/g. To determine the optimal hydrolysis temperature, L. japonica root powder was hydrolyzed by the FBS at different temperatures (10, 20, 30, 40, 45, 50, 60 or 70oC) for 3 h with an E/S ratio of 25 U/g. After hydrolysis, the reaction mixture was centrifuged at 12,000 × g for 10 min. After centrifugation, the supernatant was collected as the L. japonica root hydrolysate, and the precipitated L. japonica root powder was freeze dried and weighed for calculating the hydrolysis efficiency of L. japonica root powder by the following equation:
where W1 and W2 are the weight of the L. japonica root powder samples before and after being hydrolyzed.
Degradation products analysis
The degradation products of the FBS of strain A3 towards L. japonica piece and L. japonica roots were identified by high-resolution LTQ-Orbitrap-MS (Thermo, USA) in the negative-ion mode. The source voltage and capillary temperature were set at 3.6 kV and 275 °C, respectively. The mass acquisition range was 160–1500 m/z or 200–1500 m/z.
The AOs composition in the L. japonica root hydrolysate produced by the FBS of strain A3 was analyzed by HPLC on a Superdex Peptide 10/300 GL column (GE Healthcare, USA) [45]. The AOs in the hydrolysate were eluted with 0.2 M NH4HCO3 at a flow rate of 0.3 ml/min and detected by a UV detector at 210 nm. The commercial saturated mannoronate oligomers were taken as the standards. The proportion of each AO was calculated according to the percentage of the AO area to the total chromatograph area of the AOs [46].
Total sugar content of the degradation products was measured by the phenol–sulfuric acid method [47] using d-glucuronic acid as the standard. The content of uronic acid in the degradation products was analyzed by the sulfamate/m-hydroxydiphenyl assay described by Filisetti-Cozzi [37] using d-glucuronic acid as the standard.
Genome sequencing and analysis
Genomic DNA of strain A3 was extracted with a bacterial DNA kit (Omega, USA) and sequenced on the Illumina platform (Biozeron, China). Genome assembly was performed using ABySS v2.1.5. The genome data of strain A3 was submitted to NCBI (GenBank: JAPCKL000000000.1). The 16S rRNA gene sequence of strain A3 was aligned on the Ezbiocloud database (http://www.ezbiocloud.net). The phylogenetic analysis was performed by MEGA X [48]. The putative CAZymes of strain A3 were predicted by dbCAN meta server [49].
Secretome analysis
Strain A3 was cultured in the 100 ml medium containing 25 pieces of L. japonica blade (~ 0.1 g/ml, 1 cm × 1 cm) and 3% sea salts at 25 °C for 18 h. The supernatant of the culture was collected after centrifugation (10,000×g) and proteins in the supernatant were processed and identified according to the method described by Cheng et al. [50]. The secretome data was uploaded to the ProteomeXchange Consortium (http://proteomecentral.proteomexchange.org) via the iProX partner repository [51] under the accession number PXD037880.
Availability of data and materials
All data generated or analyzed during this study are included in this article and the additional file.
References
Shen P, Gu Y, Zhang C, Sun C, Qin L, Yu C, Qi H. Metabolomic approach for characterization of polyphenolic compounds in Laminaria japonica, Undaria pinnatifida, Sargassum fusiforme and Ascophyllum nodosum. Foods. 2021;10(1):192. https://doi.org/10.3390/foods10010192.
Wei N, Quarterman J, Jin Y. Marine macroalgae: an untapped resource for producing fuels and chemicals. Trends Biotechnol. 2013;31(2):70–7. https://doi.org/10.1016/j.tibtech.2012.10.009.
Ji SQ, Wang B, Lu M, Li FL. Direct bioconversion of brown algae into ethanol by thermophilic bacterium Defluviitalea phaphyphila. Biotechnol Biofuels. 2016;9:81. https://doi.org/10.1186/s13068-016-0494-1.
Liu G, Qu Y. Integrated engineering of enzymes and microorganisms for improving the efficiency of industrial lignocellulose deconstruction. Eng Microbiol. 2021;1:100005. https://doi.org/10.1016/j.engmic.2021.100005.
Gao J, Lin L, Sun B, Zhao M. A comparison study on polysaccharides extracted from Laminaria japonica using different methods: structural characterization and bile acid-binding capacity. Food Funct. 2017;8(9):343–52. https://doi.org/10.1039/c7fo00218a.
Zhu B, Yin H. Alginate lyase: review of major sources and classification, properties, structure-function analysis and applications. Bioengineered. 2015;6(3):125–31. https://doi.org/10.1080/21655979.2015.1030543.
Florez-Fernandez N, Torres MD, Gonzalez-Munoz MJ, Dominguez H. Recovery of bioactive and gelling extracts from edible brown seaweed Laminaria ochroleuca by non-isothermal autohydrolysis. Food Chem. 2019;277:353–61. https://doi.org/10.1016/j.foodchem.2018.10.096.
Chen J, Hu Y, Zhang L, Wang Y, Wang S, Zhang Y, Guo H, Ji D, Wang Y. Alginate oligosaccharide DP5 exhibits antitumor effects in osteosarcoma patients following surgery. Front Pharmacol. 2017;8:623. https://doi.org/10.3389/fphar.2017.00623.
Falkeborg M, Cheong L, Gianfico C, Sztukiel KM, Kristensen K, Glasius M, Xu X, Guo Z. Alginate oligosaccharides: enzymatic preparation and antioxidant property evaluation. Food Chem. 2014;164:185–94. https://doi.org/10.1016/j.foodchem.2014.05.053.
Wang Y, Han F, Hu B, Li J, Yu W. In vivo prebiotic properties of alginate oligosaccharides prepared through enzymatic hydrolysis of alginate. Nutr Res. 2006;26(11):597–603. https://doi.org/10.1016/j.nutres.2006.09.015.
Zhang Y, Yin H, Zhao X, Wang W, Du Y, He A, Sun K. The promoting effects of alginate oligosaccharides on root development in Oryza sativa L. mediated by auxin signaling. Carbohydr Polym. 2014;113:446–54. https://doi.org/10.1016/j.carbpol.2014.06.079.
Liu H, Zhang Y, Yin H, Wang W, Zhao X, Du Y. Alginate oligosaccharides enhanced Triticum aestivum L. tolerance to drought stress. Plant Physiol Biochem. 2013;62:33–40. https://doi.org/10.1016/j.plaphy.2012.10.012.
Zhang C, Howlader P, Liu T, Sun X, Jia X, Zhao X, Shen P, Qin Y, Wang W, Yin H. Alginate Oligosaccharide (AOS) induced resistance to pst DC3000 via salicylic acid-mediated signaling pathway in Arabidopsis thaliana. Carbohydr Polym. 2019;225:115221. https://doi.org/10.1016/j.carbpol.2019.115221.
Zhang C, Wang W, Zhao X, Wang H, Yin H. Preparation of alginate oligosaccharides and their biological activities in plants: a review. Carbohydr Res. 2020;494:108056. https://doi.org/10.1016/j.carres.2020.108056.
Zhang Y, Liu H, Yin H, Wang W, Zhao X, Du Y. Nitric oxide mediates alginate oligosaccharides-induced root development in wheat (Triticum aestivum L.). Plant Physiol Biochem. 2013;71:49–56. https://doi.org/10.1016/j.plaphy.2013.06.023.
Zhang YH, Yin H, Liu H, Wang WX, Wu LS, Zhao XM, Du YG. Alginate oligosaccharides regulate nitrogen metabolism via calcium in Brassica campestris L. var. Utilis Tsen et Lee. J Hortic Sci Biotech. 2013;88(4):502–8. https://doi.org/10.1080/14620316.2013.11512998.
Xu X, Bi D, Wan M. Characterization and immunological evaluation of low-molecular-weight alginate derivatives. Curr Top Med Chem. 2016;16(8):874–87. https://doi.org/10.2174/1568026615666150827101239.
Bi D, Yang X, Lu J, Xu X. Preparation and potential applications of alginate oligosaccharides. Crit Rev Food Sci Nutr. 2022. https://doi.org/10.1080/10408398.2022.2067832.
Lee DW, Choi WS, Byun MW, Park HJ, Yu Y, Lee CM. Effect of γ-irradiation on degradation of alginate. J Agric Food Chem. 2003;51(16):4819–23. https://doi.org/10.1021/jf021053y.
Yang J, Cui D, Ma S, Chen W, Chen D, Shen H. Characterization of a novel PL 17 family alginate lyase with exolytic and endolytic cleavage activity from marine bacterium Microbulbifer sp. SH-1. Int J Biol Macromol. 2021;169:551–63. https://doi.org/10.1016/j.ijbiomac.2020.12.196.
Xu X, Wu X, Wang Q, Cai N, Zhang H, Jiang Z, Wan M, Oda T. Immunomodulatory effects of alginate oligosaccharides on murine macrophage RAW264.7 cells and their structure-activity relationships. J Agric Food Chem. 2014;62(14):3168–76. https://doi.org/10.1021/jf405633n.
Preiss J, Ashwell G. Alginic acid metabolism in bacteria. I. Enzymatic formation of unsaturated oligosaccharides and 4-deoxy-l-erythro-5-hexoseulose uronic acid. J Biol Chem. 1962;237:309–16. https://doi.org/10.1016/S0021-9258(18)93920-7.
Abka-khajouei R, Tounsi L, Shahabi N, Patel AK, Abdelkafi S, Michaud P. Structures, properties and applications of alginates. Mar Drugs. 2022;20(6):364. https://doi.org/10.3390/md20060364.
Li S, Wang Z, Wang L, Peng J, Wang Y, Han Y, Zhao S. Combined enzymatic hydrolysis and selective fermentation for green production of alginate oligosaccharides from Laminaria japonica. Bioresour Technol. 2019;281:84–9. https://doi.org/10.1016/j.biortech.2019.02.056.
Wang M, Chen L, Liu Z, Zhang Z, Qin S, Yan P. Isolation of a novel alginate lyase-producing Bacillus litoralis strain and its potential to ferment Sargassum horneri for biofertilizer. Microbiologyopen. 2016;5(6):1038–49. https://doi.org/10.1002/mbo3.387.
Li H, Huang X, Yao S, Zhang C, Hong X, Wu T, Jiang Z, Ni H, Zhu Y. Characterization of a bifunctional and endolytic alginate lyase from Microbulbifer sp. ALW1 and its application in alginate oligosaccharides production from Laminaria japonica. Protein Expr Purif. 2022;200:106171. https://doi.org/10.1016/j.pep.2022.106171.
Romanenko LA, Zhukova NV, Rohde M, Lysenko AM, Mikhailov VV, Stackebrandt E. Pseudoalteromonas agarivorans sp. nov., a novel marine agarolytic bacterium. Int J Syst Evol Microbiol. 2003;53(Pt 1):125–31. https://doi.org/10.1099/ijs.0.02234-0.
Fujii K, Tominaga Y, Okunaka J, Yagi H, Ohshiro T, Suzuki H. Microbial and genomic characterization of Geobacillus thermodenitrificans OS27, a marine thermophile that degrades diverse raw seaweeds. Appl Microbiol Biotechnol. 2018;102(11):4901–13. https://doi.org/10.1007/s00253-018-8958-9.
Wakabayashi M, Sakatoku A, Noda F, Noda M, Tanaka D, Nakamura S. Isolation and characterization of Microbulbifer species 6532A degrading seaweed thalli to single cell detritus particles. Biodegradation. 2012;23(1):93–105. https://doi.org/10.1007/s10532-011-9489-6.
Sun C, Zhou J, Duan G, Yu X. Hydrolyzing Laminaria japonica with a combination of microbial alginate lyase and cellulase. Bioresour Technol. 2020;311:123548. https://doi.org/10.1016/j.biortech.2020.123548.
Feller G, Lonhienne T, Deroanne C, Libioulle C, Van Beeumen J, Gerday C. Purification, characterization, and nucleotide sequence of the thermolabile alpha-amylase from the antarctic psychrotroph Alteromonas haloplanctis A23. J Biol Chem. 1992;267(8):5217–21. https://doi.org/10.1016/S0021-9258(18)42754-8.
Kornacker MG, Pugsley AP. Molecular characterization of pulA and its product, pullulanase, a secreted enzyme of Klebsiella pneumoniae UNF5023. Mol Microbiol. 1990;4(1):73–85. https://doi.org/10.1111/j.1365-2958.1990.tb02016.x.
Zhou WW, He YL, Niu TG, Zhong JJ. Optimization of fermentation conditions for production of anti-TMV extracellular ribonuclease by Bacillus cereus using response surface methodology. Bioprocess Biosyst Eng. 2010;33(6):657–63. https://doi.org/10.1007/s00449-009-0330-0.
Li J, Dong S, Song J, Li C, Chen X, Xie B, Zhang Y. Purification and characterization of a bifunctional alginate lyase from Pseudoalteromonas sp. SM0524. Mar Drugs. 2011;9(1):109–23. https://doi.org/10.3390/md9010109.
Zhu Y, Wu L, Chen Y, Ni H, Xiao A, Cai H. Characterization of an extracellular biofunctional alginate lyase from marine Microbulbifer sp. ALW1 and antioxidant activity of enzymatic hydrolysates. Microbiol Res. 2016;182:49–58. https://doi.org/10.1016/j.micres.2015.09.004.
Mrudulakumari Vasudevan U, Lee OK, Lee EY. Alginate derived functional oligosaccharides: recent developments, barriers, and future outlooks. Carbohydr Polym. 2021;267:118158. https://doi.org/10.1016/j.carbpol.2021.118158.
Filisetti-Cozzi TM, Carpita NC. Measurement of uronic acids without interference from neutral sugars. Anal Biochem. 1991;197(1):157–62. https://doi.org/10.1016/0003-2697(91)90372-Z.
Khajouei RA, Keramat J, Hamdami N, Ursu A, Delattre C, Laroche C, Gardarin C, Lecerf D, Desbrières J, Djelveh G, Michaud P. Extraction and characterization of an alginate from the iranian brown seaweed Nizimuddinia zanardini. Int J Biol Macromol. 2018;118:1073–81. https://doi.org/10.1016/j.ijbiomac.2018.06.154.
Xue Z, Sun XM, Chen C, Zhang XY, Chen XL, Zhang YZ, Fan SJ, Xu F. A novel alginate lyase: identification, characterization, and potential application in alginate trisaccharide preparation. Mar Drugs. 2022;20(3):159. https://doi.org/10.3390/md20030159.
Schlesner H, Bartels C, Sittig M, Dorsch M, Stackebrandt E. Taxonomic and phylogenetic studies on a new taxon of budding, hyphal Proteobacteria, Hirschia baltica gen. nov., sp. nov. Int J Syst Bacteriol. 1990;40(4):443–51. https://doi.org/10.1099/00207713-40-4-443.
Miller GL. Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem. 1959;31(3):426–8. https://doi.org/10.1021/ac60147a030.
Xu F, Chen X, Sun X, Dong F, Li C, Li P, Ding H, Chen Y, Zhang Y, Wang P. Structural and molecular basis for the substrate positioning mechanism of a new PL7 subfamily alginate lyase from the arctic. J Biol Chem. 2020;295(48):16380–92. https://doi.org/10.1074/jbc.ra120.015106.
Plackett RL, Burman JP. The design of optimum multifactorial experiments. Biometrika. 1946;33(4):305–25. https://doi.org/10.1093/biomet/33.4.305.
Sun M, Zhao F, Zhang X, Zhang X, Zhang Y, Song X, Chen X. Improvement of the production of an Arctic bacterial exopolysaccharide with protective effect on human skin cells against UV-induced oxidative stress. Appl Microbiol Biotechnol. 2020;104(11):4863–75. https://doi.org/10.1007/s00253-020-10524-z.
Wang Y, Jiang W, Zhang Y, Cao H, Zhang Y, Chen X, Li C, Wang P, Zhang Y, Song X, Li P. Structural insight into chitin degradation and thermostability of a novel endochitinase from the glycoside hydrolase family 18. Front Microbiol. 2019;10:2457. https://doi.org/10.3389/fmicb.2019.02457.
Cheng JH, Zhang XY, Wang Z, Zhang X, Liu SC, Song XY, Zhang YZ, Ding JM, Chen XL, Xu F. Potential of thermolysin-like protease A69 in preparation of bovine collagen peptides with moisture-retention ability and antioxidative activity. Mar Drugs. 2021;19(12):676. https://doi.org/10.3390/md19120676.
DuBois M, Gilles KA, Hamilton JK, Rebers PA, Smith F. Colorimetric method for determination of sugars and related substances. Anal Chem. 1956;28(3):350–6. https://doi.org/10.1021/ac60111a017.
Kumar S, Stecher G, Li M, Knyaz C, Tamura K. MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol. 2018;35(6):1547–9. https://doi.org/10.1093/molbev/msy096.
Zhang H, Yohe T, Huang L, Entwistle S, Wu P, Yang Z, Busk PK, Xu Y, Yin Y. dbCAN2: a meta server for automated carbohydrate-active enzyme annotation. Nucleic Acids Res. 2018;46(W1):W95–W101. https://doi.org/10.1093/nar/gky418.
Cheng JH, Wang Y, Zhang XY, Sun ML, Zhang X, Song XY, Zhang YZ, Zhang Y, Chen XL. Characterization and diversity analysis of the extracellular proteases of thermophilic Anoxybacillus caldiproteolyticus 1A02591 from deep-sea hydrothermal vent sediment. Front Microbiol. 2021;12:643508. https://doi.org/10.3389/fmicb.2021.643508.
Ma J, Chen T, Wu S, Yang C, Bai M, Shu K, Li K, Zhang G, Jin Z, He F, Hermjakob H, Zhu Y. iProX: an integrated proteome resource. Nucleic Acids Res. 2019;47(D1):D1211-7. https://doi.org/10.1093/nar/gky869.
Acknowledgements
We thank Jing Zhu, Guannan Lin, Jingyao Qu and Zhifeng Li from State Key laboratory of Microbial Technology of Shandong University for assistance in mass spectrum.
Funding
This work was supported by the National Science Foundation of China (grants 32270047, 42076151 and U2006205), the Key Research and Development Program of Shandong Province (2021CXGC010502), and the Program of Shandong for Taishan Scholars (tspd20181203).
Author information
Authors and Affiliations
Contributions
XS performed all the experiments. XS, FX and XC wrote the original draft. FX, XC and YZ designed the research. YZ and XZ directed the experiments. XW, XZ and XS assisted in experiments. MS contributed to the statistical analysis.
Corresponding authors
Ethics declarations
Ethics approval and consent to participation
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
The analysis of variance (ANOVA) results of the model for alginate lyase production of strain A3 by the Plackett–Burman (PB) design. Table S2. The ANOVA results of the model for alginate lyase production of strain A3 by the central composite design (CCD). Formula S1. The second-order equation of central composite design (CCD) established by multiple regression analysis. Formula S2. The second-order equation used for the analysis of the central composite design (CCD) result.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Sun, XH., Chen, XL., Wang, XF. et al. Cost-effective production of alginate oligosaccharides from Laminaria japonica roots by Pseudoalteromonas agarivorans A3. Microb Cell Fact 22, 179 (2023). https://doi.org/10.1186/s12934-023-02170-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12934-023-02170-7