Abstract
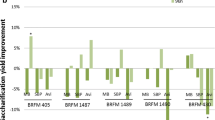
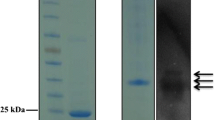
Lignocellulolytic enzymes from a novel Myceliophthora verrucosa (5DR) strain was found to potentiate the efficacy of benchmark cellulase during saccharification of acid/alkali treated bagasse by ~ 2.24 fold, indicating it to be an important source of auxiliary enzymes. The De-novo sequencing and analysis of M. verrucosa genome (31.7 Mb) revealed to encode for 7989 putative genes, representing a wide array of CAZymes (366) with a high proportions of auxiliary activity (AA) genes (76). The LC/MS QTOF based secretome analysis of M. verrucosa showed high abundance of glycosyl hydrolases and AA proteins with cellobiose dehydrogenase (CDH) (AA8), being the most prominent auxiliary protein. A gene coding for lytic polysaccharide monooxygenase (LPMO) was expressed in Pichia pastoris and CDH produced by M. verrucosa culture on rice straw based solidified medium were purified and characterized. The mass spectrometry of LPMO catalyzed hydrolytic products of avicel showed the release of both C1/C4 oxidized products, indicating it to be type-3. The lignocellulolytic cocktail comprising of in-house cellulase produced by Aspergillus allahabadii strain spiked with LPMO & CDH exhibited enhanced and better hydrolysis of mild alkali deacetylated (MAD) and unwashed acid pretreated rice straw slurry (UWAP), when compared to Cellic CTec3 at high substrate loading rate.






Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Adney B, Baker J (2008) Measurement of Cellulase Activities. Laboratory Analytical Procedure (LAP), Issue Date, 08/12/1996. National Renewable Energy Laboratory. https://www.nrel.gov/docs/gen/fy08/42628. (Accessed 25 December 2019)
Adsul M, Sandhu SK, Singhania RR, Gupta R, Puri SK, Mathur A (2020) Designing a cellulolytic enzyme cocktail for the efficient and economical conversion of lignocellulosic biomass to biofuels. Enzyme Microb Technol 133:109442. https://doi.org/10.1016/J.ENZMICTEC.2019.109442
Agger JW, Isaksen T, Várnai A, Vidal-Melgosa S, Willats WGT, Ludwig R, Horn SJ, Eijsink VGH, Westereng B (2014) Discovery of LPMO activity on hemicelluloses shows the importance of oxidative processes in plant cell wall degradation. Proc Natl Acad Sci USA 111:6287–6292. https://doi.org/10.1073/PNAS.1323629111
Agrawal D, Basotra N, Balan V, Tsang A, Chadha BS (2020a) Discovery and expression of Thermostable LPMOs from Thermophilic Fungi for producing efficient lignocellulolytic enzyme cocktails. Appl Biochem Biotechnol 191:463–481. https://doi.org/10.1007/S12010-019-03198-5
Agrawal D, Kaur B, Brar K, Chadha BS (2020b) An innovative approach of priming lignocellulosics with lytic polysaccharide mono-oxygenases prior to saccharification with glycosyl hydrolases can economize second generation ethanol process. Bioresour Technol 308:123257. https://doi.org/10.1016/j.biortech.2020.123257
Alikhan NF, Petty NK, Ben Zakour NL, Beatson SA (2011) BLAST Ring Image Generator (BRIG): simple prokaryote genome comparisons. BMC Genomics 12:402. https://doi.org/10.1186/1471-2164-12-402
Auxenfans T, Crônier D, Chabbert B, Paës G (2017) Understanding the structural and chemical changes of plant biomass following steam explosion pretreatment. Biotechnol Biofuels 10:1–16. https://doi.org/10.1186/s13068-017-0718-z
Badhan AK, Chadha BS, Sonia KG, Saini HS, Bhat MK (2004) Functionally diverse multiple xylanases of thermophilic fungus myceliophthora sp. IMI 387099. Enzyme Microb Technol 35:460–466. https://doi.org/10.1016/J.ENZMICTEC.2004.07.002
Barbosa FC, Silvello MA, Goldbeck R (2020a) Cellulase and oxidative enzymes: new approaches, challenges and perspectives on cellulose degradation for bioethanol production. Biotechnol Lett 42:875–884. https://doi.org/10.1007/S10529-020-02875-4
Barbosa FC, Kendrick E, Brenelli LB, Arruda HS, Pastore GM, Rabelo SC, Damasio A, Franco TT, Leak D, Goldbeck R (2020b) Optimization of cello-oligosaccharides production by enzymatic hydrolysis of hydrothermally pretreated sugarcane straw using cellulolytic and oxidative enzymes. Biomass Bioenergy 141:105697. https://doi.org/10.1016/J.BIOMBIOE.2020.105697
Basotra N, Dhiman SS, Agrawal D, Sani RK, Tsang A, Chadha BS (2019) Characterization of a novel Lytic Polysaccharide Monooxygenase from Malbranchea cinnamomea exhibiting dual catalytic behavior. Carbohydr Res 478:46–53. https://doi.org/10.1016/J.CARRES.2019.04.006
Basotra N, Kaur B, Raheja Y, Agrawal D, Sharma G, Chadha BS (2021) Developing and evaluating lignocellulolytic hyper producing deregulated strains of Mycothermus Thermophilus for hydrolysis of lignocellulosics. Biomass Convers Biorefinery 13:5059–5071. https://doi.org/10.1007/s13399-021-01539-1
Behera S, Arora R, Nandhagopal N, Kumar S (2014) Importance of chemical pretreatment for bioconversion of lignocellulosic biomass. Renew Sustainable Energy Rev 36:91–106. https://doi.org/10.1016/j.rser.2014.04.047
Bernardes A, Pellegrini VOA, Curtolo F, Camilo FCM, Mello BL, Johns MA, Scott JL, Guimaraes FEC, Polikarpov I (2019) Carbohydrate binding modules enhance cellulose enzymatic hydrolysis by increasing access of cellulases to the substrate. Carbohydr Polym 211:57–68. https://doi.org/10.1016/J.CARBPOL.2019.01.108
Bernt M, Donath A, Jühling F, Externbrink F, Florentz C, Fritzsch G, Pütz J, Middendorf M, Stadler PF (2013) MITOS: Improved de novo metazoan mitochondrial genome annotation. Mol Phylogenetics Evol 69:313–319. https://doi.org/10.1016/j.ympev.2012.08.023
Berrin JG, Rosso MN, Abou Hachem M (2017) Fungal secretomics to probe the biological functions of lytic polysaccharide monooxygenases. Carbohydr Res 448:155–160. https://doi.org/10.1016/J.CARRES.2017.05.010
Bertini L, Breglia R, Lambrughi M, Fantucci P, De Gioia L, Borsari M, Sola M, Bortolotti CA, Bruschi M (2018) Catalytic mechanism of Fungal Lytic Polysaccharide Monooxygenases investigated by first-principles calculations. Inorg Chem 57:86–97. https://doi.org/10.1021/ACS.INORGCHEM.7B02005
Bey M, Zhou S, Poidevin L, Henrissat B, Coutinho PM, Berrin JG, Sigoillot JC (2013) Cello-oligosaccharide oxidation reveals differences between two lytic polysaccharide monooxygenases (family GH61) from Podospora Anserina. Appl Environ Microbiol 79:488–496. https://doi.org/10.1128/AEM.02942-12
Bissaro B, Røhr ÅK, Müller G, Chylenski P, Skaugen M, Forsberg Z, Horn SJ, Vaaje-Kolstad G, Eijsink VGH (2017) Oxidative cleavage of polysaccharides by monocopper enzymes depends on H2O2. Nat Chem Biol 13:1123–1128. https://doi.org/10.1038/NCHEMBIO.2470
Bissaro B, Várnai A, Røhr ÅK, Eijsink VGH (2018) Oxidoreductases and reactive oxygen species in Conversion of Lignocellulosic Biomass. Microbiol Mol Biol Rev 82:e00029–e00018. https://doi.org/10.1128/MMBR.00029-18
Breslmayr E, Hanžek M, Hanrahan A, Leitner C, Kittl R, Šantek B, Oostenbrink C, Ludwig R (2018) A fast and sensitive activity assay for lytic polysaccharide monooxygenase. Biotechnol Biofuels 11:79. https://doi.org/10.1186/S13068-018-1063-6
Buchfink B, Xie C, Huson DH (2015) Fast and sensitive protein alignment using DIAMOND. Nat Methods 12:59–60. https://doi.org/10.1038/nmeth.3176
Chadha BS, Rubinder K, Saini HS (2005) Constitutive α-amylase producing mutant and recombinant haploid strains of thermophilic fungus Thermomyces Lanuginosus. Folia Microbiol 50:133–140. https://doi.org/10.1007/BF02931462
Chandel AK, Albarelli JQ, Santos DT, Chundawat SP, Puri M, Meireles MAA (2019) Comparative analysis of key technologies for cellulosic ethanol production from Brazilian sugarcane bagasse at a commercial scale. Biofuels Bioprod Biorefining 13:994–1014. https://doi.org/10.1002/BBB.1990
Chen K, Liu X, Long L, Ding S (2017) Cellobiose dehydrogenase from Volvariella Volvacea and its effect on the saccharification of cellulose. Process Biochem 60:52–58. https://doi.org/10.1016/J.PROCBIO.2017.05.023
Chylenski P, Petrović DM, Müller G, Dahlström M, Bengtsson O, Lersch M, Siika-Aho M, Horn SJ, Eijsink VGH (2017) Enzymatic degradation of sulfite-pulped softwoods and the role of LPMOs. Biotechnol Biofuels 10:177. https://doi.org/10.1186/s13068-017-0862-5
Cregg JM, Tolstorukov I, Kusari A, Sunga J, Madden K, Chappell T (2009) Expression in the yeast Pichia pastoris. Methods Enzymol 463:169–189. https://doi.org/10.1016/S0076-6879(09)63013-5
Daou M, Bisotto A, Haon M, Correia LO, Cottyn B, Drula E, Garajová S, Bertrand E, Record E, Navarro D, Raouche S, Baumberger S, Faulds CB (2021) A putative lignin copper oxidase from Trichoderma reesei. J Fungi 7:643. https://doi.org/10.3390/JOF7080643
Diner BA, Lasio J, Camp CE, David Rosenfeld H, Fan J, Fox BC (2015) Gaseous ammonia pretreatment lowers the required energy input for fine milling-enhanced enzymatic saccharification of switchgrass. Biotechnol Biofuels 8:139. https://doi.org/10.1186/s13068-015-0315-y
Eijsink VGH, Petrovic D, Forsberg Z, Mekasha S, Røhr ÅK, Várnai A, Bissaro B, Vaaje-Kolstad G (2019) On the functional characterization of lytic polysaccharide monooxygenases (LPMOs). Biotechnol Biofuels 12:1–16. https://doi.org/10.1186
Felice AKG, Schuster C, Kadek A, Filandr F, Laurent CVFP, Scheiblbrandner S, Schwaiger L, Schachinger F, Kracher D, Sygmund C, Man P, Halada P, Oostenbrink C, Ludwig R (2021) Chimeric cellobiose dehydrogenases reveal the function of cytochrome domain mobility for the Electron Transfer to Lytic Polysaccharide Monooxygenase. ACS Catal 11:517–532. https://doi.org/10.1021/ACSCATAL.0C05294
Forsberg Z, Røhr AK, Mekasha S, Andersson KK, Eijsink VGH, Vaaje-Kolstad G, Sørlie M (2014) Comparative study of two chitin-active and two cellulose-active AA10-type lytic polysaccharide monooxygenases. Biochem 53:1647–1656. https://doi.org/10.1021/bi5000433
Garajova S, Mathieu Y, Beccia MR, Bennati-Granier C, Biaso F, Fanuel M, Ropartz D, Guigliarelli B, Record E, Rogniaux H, Henrissat B, Berrin JG (2016) Single-domain flavoenzymes trigger lytic polysaccharide monooxygenases for oxidative degradation of cellulose. Sci Rep 6:28276. https://doi.org/10.1038/srep28276
Gavaseraei HR, Hasanzadeh R, Afsharnezhad M, Kalurazi AF, Shahangian SS, Aghamaali MR, Aminzadeh S (2021) Identification, heterologous expression and biochemical characterization of a novel cellulase-free xylanase B from the thermophilic bacterium Cohnella sp. A01. Process Biochem 107:48–58. https://doi.org/10.1016/j.procbio.2021.05.002
Haddad Momeni M, Fredslund F, Bissaro B, Raji O, Vuong TV, Meier S, Nielsen TS, Lombard V, Guigliarelli B, Biaso F, Haon M, Grisel S, Henrissat B, Welner DH, Master ER, Berrin JG, Abou Hachem M (2021) Discovery of fungal oligosaccharide-oxidising flavo-enzymes with previously unknown substrates, redox-activity profiles and interplay with LPMOs. Nat Commun 12:2132. https://doi.org/10.1038/s41467-021-22372-0
Häkkinen M, Valkonen MJ, Westerholm-Parvinen A, Aro N, Arvas M, Vitikainen M, Penttilä M, Saloheimo M, Pakula TM (2014) Screening of candidate regulators for cellulase and hemicellulase production in Trichoderma reesei and identification of a factor essential for cellulase production. Biotechnol Biofuels 7:14. https://doi.org/10.1186/1754-6834-7-14
Harris PV, Welner D, McFarland KC, Re E, Poulsen JCN, Brown K, Salbo R, Ding H, Vlasenko E, Merino S, Xu F, Cherry J, Larsen S, Leggio LL (2010) Stimulation of lignocellulosic biomass hydrolysis by proteins of glycoside hydrolase family 61: structure and function of a large, enigmatic family. Biochem 49:3305–3316. https://doi.org/10.1021/bi100009p
Hemsworth G, Henrissat B, Davies GJ, Walton PH (2014) Discovery and characterization of a new family of lytic polysaccharide monooxygenases. Nat Chem Biol 10:122–126. https://doi.org/10.1038/nchembio.1417
Hoff KJ, Lomsadze A, Borodovsky M, Stanke M (2019) Whole-genome annotation with BRAKER. In: Kollmar M (ed) Gene Prediction. Methods in Molecular Biology. Humana, New York, pp 65–95. https://doi.org/10.1007/978-1-4939-9173-0_5
Isaksen T, Westereng B, Aachmann FL, Agger JW, Kracher D, Kittl R, Ludwig R, Haltrich D, Eijsink VGH, Horn SJ (2014) A C4-oxidizing lytic polysaccharide monooxygenase cleaving both cellulose and cello-oligosaccharides. J Biol Chem 289:2532–2642. https://doi.org/10.1074/jbc.M113.530196
Jain KK, Kumar A, Shankar A, Pandey D, Chaudhary B, De Sharma KK (2020) novo transcriptome assembly and protein profiling of copper-induced lignocellulolytic fungus Ganoderma lucidum MDU-7 reveals genes involved in lignocellulose degradation and terpenoid biosynthetic pathways. Genomics 112:184–192. https://doi.org/10.1016/j.ygeno.2019.01.012
Johansen KS (2016) Discovery and industrial applications of lytic polysaccharide mono-oxygenases. Biochem Soc Trans 44:143–149. https://doi.org/10.1042/BST20150204
Karlsson J, Saloheimo M, Siika-aho M, Tenkanen M, Penttilä M, Tjerneld F (2001) Homologous expression and characterization of Cel61A (EG IV) of Trichoderma reesei. Eur J Biochem 268:6498–6507. https://doi.org/10.1046/j.0014-2956.2001.02605.x
Karnaouri A, Topakas E, Antonopoulou I, Christakopoulos P (2014) Genomic insights into the fungal lignocellulolytic system of Myceliophthora Thermophila. Front Microbiol 5:281. https://doi.org/10.3389/FMICB.2014.00281/XML/NLM
Kolbusz MA, Di Falco M, Ishmael N, Marqueteau S, Moisan MC, Baptista C, da Powlowski J, Tsang S A (2014) Transcriptome and exoproteome analysis of utilization of plant-derived biomass by Myceliophthora Thermophila. Fungal Genet Biol 72:10–20. https://doi.org/10.1016/j.fgb.2014.05.006
Kracher D, Ludwig R (2016) Cellobiose dehydrogenase: an essential enzyme for lignocellulose degradation in nature - A review. Bodenkultur 67:145–163. https://doi.org/10.1515/BOKU-2016-0013
Kuhn EM, Chen X, Tucke MP (2020) Deacetylation and mechanical refining (DMR) and deacetylation and dilute acid (DDA) pretreatment of corn stover, switchgrass, and a 50: 50 corn stover/switchgrass blend. ACS Sustain Chem Eng 8:6734–6743. https://doi.org/10.1021/acssuschemeng.0c00894
Li X, Zheng Y (2018) Investigation of dynamic changes of substrate features on enzymatic hydrolysis of lignocellulosic biomass. Ind Crops Prod 111:414–421. https://doi.org/10.1016/J.INDCROP.2017.10.063
Lo Leggio L, Simmons T, Poulsen JC, Frandsen KE, Hemsworth GR, Stringer MA, Von Freiesleben P, Tovborg M, Johansen KS, De Maria L, Harris PV (2015) Structure and boosting activity of a starch-degrading lytic polysaccharide monooxygenase. Nat Commun 6:5961 (2015). https://doi.org/10.1038/ncomms6961
Ma L, Liu Z, Kong Z, Wang M, Li T, Zhu H, Wan Q, Liu D, Shen Q (2021) Functional characterization of a novel copper-dependent lytic polysaccharide monooxygenase TgAA11 from Trichoderma Guizhouense NJAU 4742 in the oxidative degradation of chitin. Carbohydr Polym 258:117708. https://doi.org/10.1016/J.CARBPOL.2021.117708
Makhlouf J, Carvajal-Campos A, Querin A, Tadrist S, Puel O, Lorber S, Oswald IP, Hamze M, Bailly JD, Bailly S (2019) Morphologic, molecular and metabolic characterization of Aspergillus section Flavi in spices marketed in Lebanon. Sci Rep 9:5623. https://doi.org/10.1038/s41598-019-41704-1
Mankar AR, Pandey A, Modak A, Pant KK (2021) Pretreatment of lignocellulosic biomass: a review on recent advances. Bioresour Technol 334:125235. https://doi.org/10.1016/J.BIORTECH.2021.125235
Morais ARC, Zhang J, Dong H, Otto WG, Mokomele T, Hodge D, Balan V, Dale BE, Lukasik RM, da Costa Sousa L (2022) Development of an ammonia pretreatment that creates synergies between biorefineries and advanced biomass logistics models. Green Chem 24:4443–4462. https://doi.org/10.1039/D2GC00488G
Müller G, Várnai A, Johansen KS, Eijsink VGH, Horn SJ (2015) Harnessing the potential of LPMO-containing cellulase cocktails poses new demands on processing conditions. Biotechnol Biofuels 8:187. https://doi.org/10.1186/S13068-015-0376
Navarro D, Rosso MN, Haon M, Olivé C, Bonnin E, Lesage-Meessen L, Chevret D, Coutinho PM, Henrissat B, Berrin JG (2014) Fast solubilization of recalcitrant cellulosic biomass by the basidiomycete fungus Laetisaria arvalis involves successive secretion of oxidative and hydrolytic enzymes. Biotechnol Biofuels 7:143. https://doi.org/10.1186/S13068-014-0143-5
Novozymes Cellic[®] CTec3 (2022) Secure your plant’s lowest total cost [WWW Document]. https://www.yumpu.com/en/document/read/50573332/novozymes-cellicar-ctec3-secure-your-plants-lowest-total-cost, 2022. (Accessed 2 October 2022)
Ogunyewo OA, Randhawa A, Gupta M, Kaladhar VC, Verma PK, Yazdani SS (2020) Synergistic action of a Lytic Polysaccharide Monooxygenase and a cellobiohydrolase from Penicillium Funiculosum in Cellulose Saccharification under High-Level substrate loading. Appl Environ Microbiol 86:e01769–e01720. https://doi.org/10.1128/AEM.01769-20
Østby H, Hansen LD, Horn SJ, Eijsink VGH, Várnai A (2020) Enzymatic processing of lignocellulosic biomass: principles, recent advances and perspectives. J Ind Microbiol Biotechnol 47:623–657. https://doi.org/10.1007/S10295-020-02301-8
Pal S, Joy S, Trimukhe KD, Kumbhar PS, Varma AJ, Padmanabhan S (2016) Pretreatment and enzymatic process modification strategies to improve efficiency of sugar production from sugarcane bagasse. 3 Biotech 6:126. https://doi.org/10.1007/S13205-016-0446-2
Pasari N, Gupta M, Eqbal D, SYazdani SS (2019) Genome analysis of Paenibacillus polymyxa A18 gives insights into the features associated with its adaptation to the termite gut environment. Sci Rep 9:6091. https://doi.org/10.1038/s41598-019-42572-5
Payne CM, Knott BC, Mayes HB, Hansson H, Himmel ME, Sandgren M, Stahlberg J, Beckham GT (2015) Fungal cellulases. Chem Rev 115:1308–1448. https://doi.org/10.1021/cr500351c
Perez-Riverol Y, Bai J, Bandla C, Hewapathirana S, García-Seisdedos D, Kamatchinathan S, Kundu D, Prakash A, Frericks-Zipper A, Eisenacher M, Walzer M, Wang S, Brazma A, Vizcaíno JA (2022) The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res 50(D1):D543 D552 (PubMed ID: 34723319)
Quinlan RJ, Sweeney MD, Lo Leggio L, Otten H, Poulsen JCN, Johansen KS, Krogh KBRM, Jørgensen CI, Tovborg M, Anthonsen A, Tryfona T, Walter CP, Dupree P, Xu F, Davies GJ, Walton PH (2011) Insights into the oxidative degradation of cellulose by a copper metalloenzyme that exploits biomass components. Proc Natl Acad Sci USA 108:15079–15084. https://doi.org/10.1073/PNAS.1105776108
Raheja Y, Kaur B, Falco M, Tsang A, Chadha BS (2020) Secretome analysis of Talaromyces Emersonii reveals distinct CAZymes profile and enhanced cellulase production through response surface methodology. Ind Crops Prod 152:112554. https://doi.org/10.1016/J.INDCROP.2020.112554
Raheja Y, Singh V, Kaur B, Basotra N, Di Falco M, Tsang A, Chadha BS (2022) Combination of system biology and classical approaches for developing biorefinery relevant lignocellulolytic Rasamsonia emersonii strain. Bioresour Technol 351:127039. https://doi.org/10.1016/J.BIORTECH.2022.127039
Rai R, Kaur B, Singh S, Di Falco M, Tsang A, Chadha BS (2016) Evaluation of Secretome of highly efficient lignocellulolytic Penicillium sp. Dal 5 isolated from rhizosphere of conifers. Bioresour Technol 216:958–967. https://doi.org/10.1016/J.BIORTECH.2016.06.040
Rai R, Basotra N, Kaur B, Di Falco M, Tsang A, Chadha BS (2020) Exoproteome profile reveals thermophilic fungus crassicarpon thermophilum (strain 6GKB; syn. Corynascus Thermophilus) as a rich source of cellobiose dehydrogenase for enhanced saccharification of bagasse. Biomass Bioenergy 132:105438. https://doi.org/10.1016/J.BIOMBIOE.2019.105438
Ryu JS, Shary S, Houtman CJ, Panisko EA, Korripally P, St. John FJ, Crooks C, Siika-aho M, Magnuson JK, Hammel KE (2017) Proteomic and functional analysis of the cellulase system expressed by Postia placenta during brown rot of solid wood. Appl Environ Microbiol 77:7933–7941. https://doi.org/10.1128/AEM.05496-11
Saldarriaga-Hernández S, Velasco-Ayala C, Leal-Isla Flores P, de Jesús Rostro-Alanis M, Parra-Saldivar R, Iqbal HMN, Carrillo-Nieves D (2020) Biotransformation of lignocellulosic biomass into industrially relevant products with the aid of fungi-derived lignocellulolytic enzymes. Int J Biol Macromol 161:1099–1116. https://doi.org/10.1016/J.IJBIOMAC.2020.06.047
Semenova MV, Gusakov AV, Volkov PV, Matys VY, Nemashkalov VA, Telitsin VD, Rozhkova AM, Sinitsyn AP (2019) Enhancement of the enzymatic cellulose saccharification by Penicillium Verruculosum multienzyme cocktails containing homologously overexpressed lytic polysaccharide monooxygenase. Mol Biol Rep 46:2363–2370. https://doi.org/10.1007/S11033-019-04693
Shallom D, Shoham Y (2003) Microbial hemicellulases. Curr Opin Microbiol 6:219–228. https://doi.org/10.1016/S1369-5274(03)00056-0
Sharma M, Chadha BS, Kaur M, Ghatora SK, Saini HS (2008) Molecular characterization of multiple xylanases producing thermophilic/thermotolerant fungi isolated from composting materials. Lett Appl Microbiol 46:526–535. https://doi.org/10.1111/J.1472-765X.2008.02357.X
Sharma G, Kaur B, Raheja Y, Agrawal D, Basotra N, Di Falco M, Tsang A, Chadha BS (2022) Lignocellulolytic enzymes from aspergillus allahabadii for efficient bioconversion of rice straw into fermentable sugars and biogas. Bioresour Technol 360:127507. https://doi.org/10.1016/J.BIORTECH.2022.127507
Srivastava S, Jhariya U, Purohit HJ, Dafale NA (2021) Synergistic action of lytic polysaccharide monooxygenase with glycoside hydrolase for lignocellulosic waste valorization: a review. Biomass Convers Biorefinery 1–19. https://doi.org/10.1007/S13399-021-01736-Y
Tõlgo M, Hüttner S, Rugbjerg P, Thuy NT, Thanh VN, Larsbrink J, Olsson L (2021) Genomic and transcriptomic analysis of the thermophilic lignocellulose-degrading fungus thielavia terrestris LPH172. Biotechnol Biofuels 14:131. https://doi.org/10.1186/s13068-021-01975-1
Villares A, Moreau C, Bennati-Granier C, Garajova S, Foucat L, Falourd X, Saake B, Berrin JG, Cathala B (2017) Lytic polysaccharide monooxygenases disrupt the cellulose fibers structure. Sci Rep 71:40262. https://doi.org/10.1038/srep40262
Xu H, Kong Y, Peng J, Song X, Liu Y, Su Z, Li B, Gao C, Tian W (2021) Comprehensive analysis of important parameters of choline chloride-based deep eutectic solvent pretreatment of lignocellulosic biomass. Bioresour Technol 319:124209. https://doi.org/10.1016/J.BIORTECH.2020.124209
Yadav V, Nighojkar S (2023) Cellobiose dehydrogenase from Schizophyllum commune Bcc26414: purification and characterization. Biosci Biotechnol Res Asia 20:897–906. https://doi.org/10.13005/bbra/3141
Acknowledgements
The grant received from the project “Developing thermophilic fungal strains as source of cellulase and auxiliary enzymes for 2-G ethanol from lignocellulosics” sponsored by AMAAS (ICAR) and PRAJ industries ltd. Pune for providing the acid pretreated slurry is duly acknowledged.
Funding
This work was funded by AMAAS grant from the ICAR (NBAIM/AMASS/2017-20/PF/30/135).
Author information
Authors and Affiliations
Contributions
G.S. Conceptualization, Investigation, Writing – original draft. B.K. and V.S. Data curation, validation, formal analysis. Y.R. Data curation, validation. M.D.F. and A.T. Methodology, Software. B.C. Project administration, Resources, Supervision, Writing - review & editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
It is declared that no competing interest among all the authors exists.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible editor: Prof Yusuf Akhter.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, G., Kaur, B., Singh, V. et al. Genome and secretome insights: unravelling the lignocellulolytic potential of Myceliophthora verrucosa for enhanced hydrolysis of lignocellulosic biomass. Arch Microbiol 206, 236 (2024). https://doi.org/10.1007/s00203-024-03974-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03974-w