Abstract
Background
Evolutionarily conserved in plants, the enzyme D-myo-inositol-3-phosphate synthase (MIPS; EC 5.5.1.4) regulates the initial, rate-limiting reaction in the phytic acid biosynthetic pathway. They are reported to be transcriptional regulators involved in various physiological functions in the plants, growth, and biotic/abiotic stress responses. Even though the genomes of most legumes are fully sequenced and available, an all-inclusive study of the MIPS family members in legumes is still ongoing.
Results
We found 24 MIPS genes in ten legumes: Arachis hypogea, Cicer arietinum, Cajanus cajan, Glycine max, Lablab purpureus, Medicago truncatula, Pisum sativum, Phaseolus vulgaris, Trifolium pratense and Vigna unguiculata. The total number of MIPS genes found in each species ranged from two to three. The MIPS genes were classified into five clades based on their evolutionary relationships with Arabidopsis genes. The structural patterns of intron/exon and the protein motifs that were conserved in each gene were highly group-specific. In legumes, MIPS genes were inconsistently distributed across their genomes. A comparison of genomes and gene sequences showed that this family was subjected to purifying selection and the gene expansion in MIPS family in legumes was mainly caused by segmental duplication. Through quantitative PCR, expression patterns of MIPS in response to various abiotic stresses, in the vegetative tissues of various legumes were studied. Expression pattern shows that MIPS genes control the development and differentiation of various organs, and have significant responses to salinity and drought stress.
Conclusion
The MIPS genes in the genomes of legumes have been identified, characterized and their expression was analysed. The findings pave way for understanding their molecular functions and evolution, and lead to identify the putative MIPS genes associated with different cell and tissue development.
Similar content being viewed by others
Background
Myo-inositol-1-phosphate synthase (MIPS; EC 5.5.1.4), is a key, rate-limiting enzyme for the initial reaction in the lipid-independent pathway of phytic acid biosynthesis, wherein D-glucose-6-phosphate (G6P) is converted to inositol phosphate, which further is dephosphorylated to Myo-inositol (Ins), by Myo-inositol monophosphatase (IMP) [1]. MIPS genes are evolutionarily conserved and found in various species, including higher eukaryotes viz. humans and higher plants, as well as cyanobacteria, eubacteria, and archaea [2, 3]. It has also been found in a number of plants, including Arabidopsis thaliana [4], Oryza sativa [5], kiwi fruit [6], Glycine max [7, 8], Phaseolus vulgaris [9], and Sesamum indicum [10]. The eukaryotic MIPS family is homogeneous and yet distinct from the bacterial MIPS proteins in their sequence [11, 12]. This could be explained by the eukaryotic MIPS genes’ monophyletic origin. The MIPS sequences (approximately 510 amino acids), in all the eukaryotes, exhibit considerable conservation at the nucleotide level [13], signifying the crucial functions that MIPS play in various biological processes such as seed germination and embryogenesis. The MIPS proteins have four amino acid domains, GWGGNNG (domain 1), LWTANTERY (domain 2), NGSPQNTFVPGL (domain 3) and SYNHLGNNDG (domain 4) that are highly conserved [14]. These domains perform crucial roles in MIPS protein binding for the catalysis of enzymatic processes and thus regulate MIPS activities [15].
The MIPS regulates the biosynthesis of Ins and its derivatives such as phosphoinositide phosphates (PtdInsPs), Ins polyphosphates (IPs), and phospholipid phosphatidylinositol (PtdIns), which play important and varied roles in cell division, plant growth, organ development as well as biotic and abiotic stress responses [16]. This gene was reported for the first time in Archaeoglobus fulgidus and was observed to function at high temperatures [17]. Earlier studies have shown that three MIPS genes play important roles in Arabidopsis embryo development [18]. Ma et al. [19] showed how the direct binding of two light-signaling protein molecules (FHY3 and FAR1) to the MIPS1 promoter stimulated inositol synthesis in response to light-induced oxidative stress. Surprisingly, Latrasse et al. [20] discovered that MIPS1, in addition to its role in enzymatic metabolic activity, is also transported to the nucleus where it regulates transcription. Donahue et al. discovered that mips2 loss-of-function mutants of Arabidopsis are more susceptible to fungal, viral and bacterial diseases, whereas mips1 mutants exhibited higher resistance to oomycete infections but also spontaneous cell death [1]. In addition, mips1 mutants were also susceptible to severe light stress [21]. There were no major phenotypic differences in the single mips mutants, but the mips1 mips2 double mutant as well as the mips1 mips2 mips3 triple mutant were fatal to the embryo [22]. Significant suppression of plant growth was observed when the activity of MIPS was greatly reduced [15, 23]. Lower IP6 levels in the potatoes produced by RNA interference (RNAi)-mediated MIPS suppression has altered the leaf shape, promoted leaf senescence, and reduced tuber yield [24].
Legumes (Fabaceae) occupy about 27% of the primary production of the world and are grown on about 12% of the planet’s arable land [25, 26]. They help in fixing atmospheric nitrogen through the nitrogen-fixing bacteria that live symbiotically in their root nodules and thus are important nitrogen sources in crop systems [27]. Legumes are also gaining traction for the production of beneficial secondary metabolites that act as signaling molecules for attracting pollinators and seed dispersing animals and as chemical defenses against herbivores and microbes [28]. More efforts should be made to find and characterize genes associated with legume growth, development, and stress responses because they are among the world’s most important crops [29, 30]. Currently, knowledge about the MIPS gene family in Fabaceae and their contribution in legume growth and development is very little. A comparative analysis was carried out between the MIPS gene family in nonlegume (Arabidopsis and rice) and legume crops, to comprehend their roles in legumes, especially in seed germination, plant growth, and development, which included molecular traits, phylogenetic classifications, chromosome distribution, conserved motifs and synteny analysis. This is the first work on the MIPS gene family in legumes, which will support its functional research in related crops.
Methods
MIPS gene identification and characterization
The genomes and annotation files of 10 legume species including Arachis hypogea (peanut), Cicer arietinum (chickpea), Cajanus cajan (pigeon pea), Glycine max (soybean), Lablab purpureus (Dolichos bean/hyacinth bean), Medicago truncatula (barrel medic), Pisum sativum (pea), Phaseolus vulgaris (French bean/common bean), Trifolium pratense (clover), and Vigna unguiculata (cowpea) were retrieved from the Legumes database (https://plants.ensembl.org/index.html (accessed July 16, 2023)) and the Phytozome v13 database (http://www.phytozome.net (accessed July 16, 2023)) [31], and was used for the identification and annotation of MIPS genes (more information is available in Table 1). Further, three MIPS protein sequences (IDs: AT5G10170, AT4G39800 and AT2G22240) of Arabidopsis were obtained from the TAIR database (https://www.arabidopsis.org/). These sequences were used as query to perform BLASTp (e-value < 1 × 10–5) against various legumes to retrieve MIPS sequences. Thereafter, all the retrieved MIPS proteins were analyzed using the Pfam (http://pfam.xfam.org) [32], CDD (https://www.ncbi.nlm.nih.gov/cdd, [33] (accessed June 20, 2022)) and SMART (http://smart.embl-heidelberg.de (accessed June 20, 2022)) [34] databases, to validate the presence of the signature domains (Inos-1-P Synth; PF01658 and NAD_5 Binding; PF07994) in MIPS.
The DNA and protein characteristics of MIPS, including DNA sequence length, protein molecular weight, protein length, pI, and signal peptide prediction, were generated using ExPASy protparam (https://web.expasy.org/protparam, (accessed July 18, 2023)) [35], Phytozome v13 and UniProt (https://www.uniprot.org/ (accessed July 18, 2023)) [36].
Multiple sequence alignment and phylogenetic analysis
To investigate the evolutionary relationships among the MIPS peptide sequences of legumes, multiple sequence alignment (MSA) of the amino acid sequences of all the ten legumes along with three MIPS proteins from Arabidopsis, was performed using CLUSTALW in MEGA version 11.0.13 [37]. The phylogenetic tree was constructed in MEGA using the following parameters: (1) Scope: all selected taxa, (2) phylogenetic test: bootstrap technique, (3) neighbour-joining (NJ) statistical approach, (4) 1000 bootstrap replicates, (5) Substitution types: amino acid, (6) Poisson model, (7) Uniform rates applied across all sites, (8) Homogeneous (same) ancestry pattern, and (9) Pairwise elimination for the treatment of gaps/missing. To visualize the phylogenetic tree, the iTOL45 web server (https://itol.embl.de/, [38]) was used.
Gene structures, organization of motif, and prediction of domain
The Gene Structure Display Server (GSDS 2.0; http://gsds.cbi.pku.edu.cn/Gsds_abou.php) program was used to compare the CDS sequences with their corresponding genomic DNA sequences, to analyze the organization of the exonic and intronic regions as well as the untranslated region of the MIPS genes. The Motif Elicitation (MEME) program was employed to envisage the conserved motif with the parameters set to find out 20 motifs (http://meme-suit.org; [39]) and their structures were visualized using the TBtool software [40].
Gene nomenclature, duplication, chromosomal location, and analysis of cis-regulatory elements
The MIPS genes reported in Arabidopsis were used to determine the nomenclature of the genes in each legume species. The closest orthologs to the MIPS genes of Arabidopsis in the evolutionary tree were used to determine the nomenclature of the genes in each legume species. From NCBI, the chromosome coordinates and the gene and protein IDs were obtained. The position of each gene on each chromosome was plotted on chromosome maps generated for each species using the MG2C tool (http://mg2c.iask.in). For the analysis of the cis-regulatory elements (CREs) of MIPS, the sequences upstream (2000 bp) to the start codon were also retrieved from the corresponding database. The PlantCARE online server (https://bio.tools/plantcare) was used to identify the CREs in MIPS genes [41].
Nuclear localization signals, GO analysis and subcellular localization
To predict the MIPS NLS, the online tool cNLS Mapper (https://nls-mapper.iab.keio.ac.jp) [42] was used. The PANNZER server (Protein ANNotation with Z-scoRE) (http://ekhidna2.biocenter.helsinki.fi/sanspanz) [43] was employed to get the gene ontology (GO) annotation of the legume MIPS using their respective gene ID. Further, for predicting subcellular localization of legume MIPS, online resource WoLF PSORT II (https://www.genscript.com/wolf-psort.html?src=leftbar) [44] was used.
Plant materials and optimum growing conditions
Dolichos bean (Dolichos lablab L. cv. Hima) seeds were grown at ambient temperature (25 °C) in the greenhouse with a light/dark cycle of 14 h/10 h in pots filled with sterile mixtures (1:1) of agro-peat and vermiculite. The tissue-specific gene expression was analysed, for which the bud, leaf and root tissues were collected at the flowering stage (three months). All tissues were collected in three biological replicates and kept at -80 °C until used. PEG-6000 (20%) was used to induce drought stress in dolichos bean plants.
Isolation of total RNA, cDNA preparation and RT-qPCR validation
Purelink® Plant RNA purification reagent was used to isolate the total RNA, from 100 mg of tissue, following the manufacturer’s guidelines. From this, 5.0 µg of total RNA was treated using the RNase-Free DNase Set (Qiagen) for removing the contaminating DNA. Using nano-volume spectrophotometer (NanoDrop Technologies) and Bioanalyzer (Agilent Technologies), the quantity and quality of the RNA samples were evaluated. Only RNA samples with the following characteristics were selected for analysis: 260/230 absorbance ratios between 2.0 and 2.4, 260/280 ratios between 1.9 and 2.1, and an RNA integrity number (RIN) larger than 7.0. The RevertAid First Strand cDNA Synthesis Kit (ThermoScientific) was used to create cDNA from the total RNA, following the manufacturer’s instructions. A reaction mix of 10 µL containing 2X SyBr Green Master mix (5.0 µL) (Applied Biosystems™), 10 mM of each primer, and cDNA (100 ng) was used to perform the real-time PCR assay on the Applied Biosystems™ qPCR, and the temperature profile for the reaction was 95 °C (2 min), 40 cycles of 95 °C (15 s) and 60 °C for 1 min. Supplementary Table S1 provides the details of the primers used in this study. Three each of biological and technical replicates were made in each reaction. The 2−△△Ct technique was followed to analyze the data and calculate the relative expression.
Results
Identification and characterization of MIPS genes from legume genomes
To find the genes encoding proteins containing the MIPS domain, genome-wide searches were conducted by BLASTp. Further, HMMER was used for a thorough search and identification of MIPS genes in each of the legume proteomes. Results obtained were confirmed for the presence of the MIPS domain using Pfam, SMART, and NCBI Conserved Domains Database. A few protein sequences that were incomplete in the N-terminal region of the characteristic MIPS domain were excluded. In total, we found 24 potential MIPS genes and 47 different transcripts from 10 legume species (Supplementary Table S2). We gave each MIPS protein a unique name based on the MIPS genes reported in Arabidopsis and their chromosomal or scaffold position, using the species abbreviation as a prefix. Each legume had 2–3 MIPS genes, except soybean, which had four MIPS genes (Table 1, Supplementary Table S2).
The basic parameters of the identified proteins such as protein length, isoelectric point, molecular weight, subcellular localization, and functional annotation are presented in Table 1 and Supplementary Table S2. The length of MIPS genes in different legumes ranged from 2498 (PsMIPS1) to 5237 bp (LpMIPS2), and their average was 3770 bp. The length of MIPS protein sequences showed a bimodal distribution, ranging from 126 aa (TpMIPS4) to 529 aa (PsMIPS1) and the average was 479 aa. The length of the MIPS proteins fell into any one of two categories: (i) 509–529 amino acid residues for 21 MIPS proteins or (ii) 129–414 amino acid residues for three MIPS proteins. The predicted molecular weight of the proteins ranged from 14.07 (TpMIPS2) to 58.90 kDa (GmMIPS1), and the average molecular weight was 53.01 kDa. The range of isoelectric points of the proteins was 5.24 to 8.52.
The analysis of subcellular locations had shown that the proteins are localized in multiple organelles and most were located in the cytoplasm, plasma membrane, and vacuole (Fig. 1).
Phylogenetic analysis and classification of MIPS genes
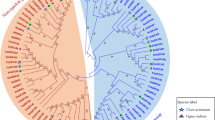
To comprehend the evolutionary relationships of MIPS genes, a multiple sequence alignment of 24 full-length peptide sequences from the legumes was performed and a phylogenetic tree was constructed using the neighbour-joining method (Fig. 2A). The MIPS proteins were thus classified into five clusters. Clusters B, C and D had bootstrap values > 80% and hence considered to have different phylogenetic lineages. The clusters A and E had bootstrap values less than 60%, having large variability among the members. To understand the phylogenetic relationship among the MIPS genes of legumes and Arabidopsis, an evolutionary tree was constructed with 24 sequences from legumes and three from Arabidopsis. The MIPS proteins from these species were distributed across almost all the branches. The evolutionary tree was divided into five subclades. Among them, subclades MIPS1 and MIPS2 have accommodated one sequence each, while MIPS4 had 15 and subclades MIPS3 and MIPS2 had seven and three sequences, respectively (Fig. 2B).
Two unrooted phylogenetic trees of MIPS genes were constructed by MEGA11: the evolutionary tree of the MIPS family was constructed using the neighbor-joining method, and the interspecific evolutionary tree of MIPS was constructed using the maximum likelihood method. A Phylogenetic tree of the MIPS family protein sequences in legume; B Phylogenetic relationships of 24 MIPS proteins from 10 legumes, and Arabidopsis
Gene structure and conserved motif analysis
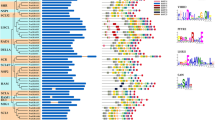
Clear gene structure patterns were seen, which varied across groups but conserved within a group (Fig. 3A). The most common number of exons was ten, found in 17 MIPS genes (70.83%) (Fig. 3B). In total, there were 17 genes with nine introns each (70.83%), two with four introns (8.33%), four with eight introns (16.66%) and one gene (TpMIPS4) with no introns (4.16%). A, C, D and E group genes had eight to nine introns. On the contrary, genes in group B varied greatly with four to zero introns.
The validation of the classification of MIPS using MEME analysis [45] showed 10 conserved motifs that were statistically significant (E value lower than e−100) (Fig. 3C, Supplementary Table S3). The amino acid motif length ranged from 29 to 50. The consensus sequences of these conserved motifs and their amino acid lengths are shown in Supplementary Table S3. These ten motifs were observed in all legume genes except CaMIPS1, TpMIPS2 and TpMIPS3 and the pattern in which the motifs were arranged was also similar in 21 proteins (Fig. 3C). This suggests that MIPS is highly conserved in legumes.
Location of MIPS on the chromosomes and gene duplication events
The distribution of the MIPS genes on the chromosomes of legumes was mapped with the programme MapInspect. We also examined the events of gene duplication in the MIPS gene family, and the output showed that MIPS gene pairs arose by tandem and segmental duplication and are connected by lines or shown in grey (Fig. 4). The legume chromosomes had an uneven distribution of MIPS genes. Tandem duplication contributed only slightly to the gene expansion of the MIPS family in legumes, but segmental duplication still had a significant impact. Seven pairs of genes evolved through tandem duplication and 17 pairs through segmental duplication were detected.
Chromosomal localization and gene duplication of MIPS genes in Arachis hypogea, Cicer arietinum, Cajanus cajan, Glycine max, Lablab purpureus, Medicago truncatula, Pisum sativum, Phaseolus vulgaris, Trifolium pratense and Vigna unguiculata., and tandem duplication of gene pairs during evolution is shown by lines
Out of all these legumes, it was observed that the gene duplication (including tandem and segmental duplication) of soybean was the most complicated, possibly due to hybridization [46, 47]. However, the gene expansion of the MIPS family in the lablab genome was only due to segmental duplication, whereas in the peanut genome, we did not find any indication of either tandem or segmental duplication of MIPS genes. Interestingly, many genes were duplicated both through tandem and segmental methods. For instance, ‘TpMIPS1 and TpMIPS2’ and ‘GmMIPS1 and GMMIPS4’ are gene pairs that denote tandem duplicates, while ‘CcMIPS2 and CcMIPS1’ is a gene pair resulting from chromosomal segmental duplication.
MIPS gene pairs that were segmentally duplicated are observed in all five groups (from A to E) of the phylogenetic tree, but such segmental duplication events are more prevalent between sister groups or within the same phylogenetic group. Fifteen of the 24 gene pairs that were segmentally duplicated had both the gene pairs in the same cluster and the remaining genes were mostly in pairs between two sister groups, such as ‘B and C’ or ‘D and E’. However, one exceptional gene pairing was between the MtMIPS1 in group A in red clover, which paired with TpMIPS1 in group B and TpMIPS5 in group E. Tandem gene duplication was observed only in phylogenetic groups D and E, where the genes were paired only within the same phylogenetic group. Furthermore, the sequence similarity of all the tandemly duplicated gene pairs was comparatively higher (> 75%). Among them, “CaMIPS1 and CaMIPS2” and “PsMIPS2 and PsMIPS3” showed more than 90% similarity.
Moreover, to find out the explanation for the legume MIPS gene expansion, the gene duplications were examined in the legume genomes, and a total of 43 duplication pairs were observed. The Ka/Ks value for the duplicated gene pairs was less than 1 for all gene pairs, with a mean value of 0.205 (Supplementary Table S2). These results indicate that in legume species, the MIPS genes are constantly evolving through purifying selection.
Analysis of the cis -regulatory elements in MIPS
To comprehend the likely role of MIPS genes in modulating growth and development of legume species, the possible CREs were searched within 2000 bp upstream of MIPS genes. A total of 6797 CREs, representing 88 types of CREs, were observed in the 24 MIPS genes (Fig. 5). These cis-acting elements were mainly elements related to plant growth (992; 44.22%), elements responsive to abiotic stress (728; 32.45%), followed by elements controlling hormone regulation (655; 15.55%), and elements responsive to light (288; 12.83%). CREs of legume MIPS genes involved in plant growth and development include the AAGAA motif, the TATA box, the CAT box, the A box, the AT -rich element and the CTAG motif, which are predominant in legume MIPS genes compared to other growth-related elements. Plant hormone-related CREs include TCA element (salicylic acid responsive elements), abscisic acid responsive elements (ABRE), gibberellin responsive elements (P-box, GARE motif), methyl jasmonate responsive elements (CGTCA motif, TGACG motif), auxin responsive elements (AuxRR core, TGA element) and ethylene responsive elements (ERE), which were the third most abundant cis-elements. These results suggest that MIPS genes may play an important role in regulating plant growth, and also in various abiotic stress responses.
GO annotation of MIPS gene
GO enrichment was also carried out for the MIPS genes to learn more about their roles and the results show that the MIPS gene was involved in the synthesis, metabolism and phospholipid metabolism of inositol (phytic acid) (Fig. 6). According to this GO-annotation, they are proven to be crucial for the production of phytic acid and drought tolerance.
MIPS gene expression profile
To examine the probable biological role of the legume MIPS genes, an expression analysis was carried out in Dolichos bean. Among leaf, stem, flower bud and flower, the stem tissue had the least expression for DlMIPS and flower bud had maximum expression (52.55-fold higher compared to the stem). The DlMIPS expression in flower and leaf tissues was 20.68-fold and 14.25-fold higher, respectively compared to that in stem (Fig. 7A).
Relative expression level of MIPS in different plant tissue, during drought stress. A Relative expression level of LpMIPS2 and LpMIPS3 in different plant tissue. B The relative expression level of LpMIPS in leaf tissue at different times (0 h, 6 h, 12 h, and 24 h) treated with 20% PEG and the effect of drought stress. Paired Student’s t-test was used to evaluate the significance of the differences between the samples, with p-values of *p < 0.05 and **p < 0.01 being considered significant
Expression analysis of MIPS gene in dolichos bean plants subjected to drought induction using 20% PEG-6000, was done through RT-qPCR. The expression of the MIPS gene under drought stress was normal until 24 h and subsequently up-regulated by 10.65-fold, compared to the control (Fig. 7B).
Discussion
The L-myo-inositol-1-phosphate synthase, also known as D-myo-inositol-3-phosphate synthase or MIPS (EC 5.5.1.4), is a crucial enzyme in the biosynthesis of inositol and phosphoinositides and is considered an ancient protein, as it is found in a variety of organisms with different evolutionary histories, including fungi, bacteria, animals, and plants [13]. The myo-inositol biosynthetic pathway (MIB) is controlled by MIPS, which converts the precursor glucose-6-phosphate into free myo-inositol. Myo-inositol and its derivatives are vital in plants for several growth and developmental processes, including auxin storage and transport, membrane biogenesis, biosynthesis of phytic acid, signal transduction, etc. [48,49,50]. The MIPS fulfils the basic physiological functions and is involved in a number of plant stress responses [51,52,53]. Myo-inositol, synthesised through the MIB pathway, is essential for the formation of phosphoinositide molecules used in evolutionarily well-defined signalling pathways, particularly in the signalling of osmotic stress [54].
The MIPS genes have been studied in various plants including, wheat [15], cotton [55], Brachypodium distachyon [56], Soybean [7], and Rose [57]. However, the comprehensive identification and characterization of these genes in the legume family has not been done. In this study, a total of 24 genes were identified and described in 10 bean species. In previous studies, 3, 12, and 51 MIPS genes were found in A. thaliana, Gossypium hirsutum, and Rosa chinensis, respectively [8, 14, 57]. With the exception of soybean, which contains four MIPS genes, the MIPS gene number in the rest of the legumes is similar to that in A. thaliana, which ranges from two to three. This is most likely due to the tetraploid genome of soybean, which is partially diploidised and may have undergone many duplications [46]. However, the disparity in the number of MIPS genes between plant species is not entirely due to genome size. The genome of rice, for example, is only about 430 Mb in size but has 38 MIPS genes [58]. Avena sativa, on the other hand, has a genome size of up to 11 Gb, but only two MIPS genes [59]. Hence, it is crucial to find and describe more MIPS genes from different species of plants to understand the several gene expansion methods in various plants.
Phylogenetic analysis revealed that 24 MIPS from 10 different legume species are divided into five different clades. Interestingly, all clades had MIPS genes from all the species studied, proving that these were conserved and evolved even before the divergence of legume species. Scattered occurrence of the majority of proteins from Arabidopsis and legumes, across the subclades, shows that MIPS proteins are functionally conserved in dicots. In addition, most of the legume genes shared MIPS exon–intron, motif, and domain arrangements. The amino acid sequence of MIPS has numerous important catalytic domains that are largely conserved across evolutionary lineages, suggesting that they are catalytically active [60]. All the ten species had the four amino acid stretches that are highly conserved, namely LWTANTERY, GWGGNG, GIKPLSIASYN, and INGSPQNTFVPG. These ten motifs were observed in all legume genes except CaMIPS1, TpMIPS2 and TpMIPS3 and the pattern in which the motifs were arranged was also similar in 21 proteins (Fig. 3C). This suggests that MIPS is highly conserved in legumes.
Studies on subcellular localization revealed that most MIPS are positioned in the cytoplasm. Previous studies in various plant species have shown that MIPS is generally found in the cytoplasm but can also be detected in the plasma membrane, endomembrane, and nucleus. These results suggest that MIPS may be associated with signal transduction, membrane trafficking, and gene expression regulation [14, 57].
The genomes of the land plants and gene family sizes have been shaped by genome duplication followed by whole-genome fractionation [61]. Multiple tandem and segmental duplication events also had a key function in the formation of the MIPS gene family, as revealed by a gene duplication study of MIPS genes in the whole-genomes of Arabidopsis and legumes. Recognition of MIPS gene orthology may be impacted by the high frequency of gene duplications. Through this study, we could predict 88 types of CREs located on the promoter region of legume MIPS genes, which were linked with hormonal regulation, response to abiotic stress, plant development, and photosensitivity. Drought-responsive CREs (ABRE, DRE, ARE, MYC, MYB, and F-box) were predominant in the legume MIPS gene when compared to other stress-responsive elements, which points to the fact that legume MIPS genes may have a role in stress response in addition to regulating growth and development in plants. Similar studies in other plant species yielded similar results [51, 62, 63]. For example, Gangwar et al. [57] reported that among the various CREs involved in abiotic stress response, CREs related to drought response were the most abundant.
MIPS genes are associated with the control of plant growth and development [57]. The wide variation (up to 52.55-fold) of MIPS in various plant tissues show that this gene also plays essential roles in growth and development of Lablab plants. MIPS has also been shown to play a critical role in tolerance to drought [64] and heat [65]. As per the GO analysis, it was observed that MIPS genes are mainly involved in the synthesis, metabolism and phospholipid metabolism of inositol (phytic acid). Myoinositol and its derivatives (e.g., pinitol, ononitol, galactinol, and raffinose) play important dual functions as both signals and essential metabolites for osmotic adjustment and ROS scavenging in response to drought stress [64]. In peanut, the MIPS gene was identified as a drought-responsive gene that was elevated in transgenic lines that were more drought resistant [66]. Similarly, MIPS gene expression was increased during drought stress in Nicotiana glauca [67], rose [57], cotton [14], Arabidopsis [68], and many others. Overexpressing the HhMIPS1D gene of cotton in transgenic Arabidopsis improved drought tolerance [14]. Under drought stress, the MIPS gene (OsMIPS1) splice variants were also increased in rice [65]. Zhai et al. [2016] reported that a myo‐inositol‐1‐phosphate synthase gene, IbMIPS1 has enhanced the tolerance to salinity and drought and resistance to stem nematodes in transgenic sweet potato [64]. Tan et al. [2013] found that through the ectopic expression of MfMIPS1 (Medicago falcata MIPS) in tobacco, tolerance to drought is increased [69]. In the current study, a substantial increase in the MIPS gene expression was detected under the drought stress in Lablab, indicating a positive function of the MIPS gene for drought resistance in lablab. In addition, we detected drought-responsive elements CREs (i.e. DRE, ABRE, MYC and MYB) in the promoter sequence of MIPS, suggesting the involvement of MIPS in plant tolerance under low-water conditions.
Conclusion
We discovered 24 MIPS genes belonging to five clades, from ten legume species. Total length of the gene was 6085 bp and gene structure analysis had shown one to nine exons. The presence of multiple components in the promoter sequence, including the growth- and stress-sensitive elements, suggests that MIPS plays a critical role in many developmental processes as well as abiotic stresses in legumes. Analysis of LpMIPS expression has confirmed the importance of MIPS in plant development and drought stress resistance. Current results provide precise data on MIPS genes, which could serve as a solid platform for the functional characterization of this important gene family.
Availability of data and materials
All the results, obtained through this study are included in the article/Supplementary Material. Further clarifications can be received from the corresponding author.
Abbreviations
- aa:
-
Amino acids
- BLASTp:
-
Protein-to-protein Basic Local Alignment Search Tool
- bp:
-
Base pair
- CDD:
-
Conserved Domain Database
- cDNA:
-
Complementary DNA
- CDS:
-
Coding sequence
- CREs:
-
Cis-regulatory elements
- G6P:
-
D-glucose-6-phosphate
- GO:
-
Gene Ontology
- GSDS:
-
Gene Structure Display Server
- HMM:
-
Hidden Markov Model
- IMP:
-
Myo-inositol monophosphatase
- Ins:
-
Myo-inositol
- kDa:
-
Kilo Dalton
- Ips:
-
Ins polyphosphates
- iTOL:
-
Interactive Tree of Life
- MEME:
-
Multiple EM for Motif Elicitation
- MIPS:
-
Myo-inositol-1-phosphate synthase
- NCBI:
-
National Center for Biotechnology
- NJ:
-
Neighbour-joining
- NLS:
-
Nuclear localization signals
- PANNZER:
-
Protein ANNotation with Z-scoRE
- Pfam:
-
Protein Families
- pI:
-
Isoelectric point
- PtdIns:
-
Phospholipid phosphatidylinositol
- PtdInsP:
-
Phosphoinositide phosphates
- qPCR:
-
Quantitative PCR
- RIN:
-
RNA integrity number
- SMART:
-
Simple Modular Architecture Research Tool
- TAIR:
-
The Arabidopsis Information Resource
References
Donahue JL, Alford SR, Torabinejad J, Kerwin RE, Nourbakhsh A, Ray WK, Hernick M, Huang X, Lyons BM, Hein PP. The Arabidopsis thaliana Myo-Inositol 1-Phosphate Synthase1 gene is required for myo-inositol synthesis and suppression of cell death. Plant Cell. 2010;22(3):888–903.
Hazra A, Nandy P. Myo-inositol 1-phosphate synthase–the chosen path of evolution. Biotechnologia. 2016;97(2):95–108.
Hazra A. Computational fishing and structural analysis of MIPS protein from two important plant groups. Int Lett Nat Sci. 2015;42:18–27.
Sharma N, Shree B, Khurana P. Myo-inositol phosphate synthase improves heat stress tolerance by ethylene-mediated modulation of chlorophyll content and photosynthetic efficiency. Protoplasma. 2023;260(4):1097–107.
Karle SB, Kumar K, Srivastava S, Suprasanna P. Cloning, in silico characterization and expression analysis of TIP subfamily from rice (Oryza sativa L.). Gene. 2020;761:145043.
Cui M, Liang D, Ma F. Molecular cloning and characterization of a cDNA encoding kiwifruit L-myo-inositol-1-phosphate synthase, a key gene of inositol formation. Mol Biol Rep. 2013;40:697–705.
Chappell A, Scaboo A, Wu X, Nguyen H, Pantalone V, Bilyeu K. Characterization of the MIPS gene family in Glycine max. Plant Breed. 2006;125(5):493–500.
Kumari S, Jolly M, Krishnan V, Dahuja A, Sachdev A. Spatial and temporal expression analysis of D-myo-inositol 3-phosphate synthase (MIPS) gene family in Glycine max. Afr J Biotechnol. 2012;11(98):16443–54.
Blair MW, Herrera AL, Sandoval TA, Caldas GV, Filleppi M, Sparvoli F. Inheritance of seed phytate and phosphorus levels in common bean (Phaseolus vulgaris L.) and association with newly-mapped candidate genes. Mol Breed. 2012;30:1265–77.
Chun J-A, Jin U-H, Lee J-W, Yi Y-B, Hyung N-I, Kang M-H, Pyee J-H, Suh M, Kang C-W, Seo H-Y. Isolation and characterization of a myo-inositol 1-phosphate synthase cDNA from developing sesame (Sesamum indicum L.) seeds: functional and differential expression, and salt-induced transcription during germination. Planta. 2003;216:874–80.
Geiger JH, Jin X. The structure and mechanism of myo-inositol-1-phosphate synthase. In: Majumder AL, Biswas BB, editors. Biology of inositols and phosphoinositides, subcellular biochemistry, vol. 39. Boston: Springer; 2006. p. 157–80.
Park JK, Cho K, Johnson J, Perez RV. Induction of MIP-1α in Kupffer cell by portal venous transfusion. Transplant Immunol. 2004;13(1):33–8.
Hazra A, Dasgupta N, Sengupta C, Das S. MIPS: functional dynamics in evolutionary pathways of plant kingdom. Genomics. 2019;111(6):1929–45.
Ma R, Song W, Wang F, Cao A, Xie S, Chen X, Jin X, Li H. A cotton (Gossypium hirsutum) Myo-inositol-1-phosphate synthase (GhMIPS1D) gene promotes root cell elongation in Arabidopsis. Int J Mol Sci. 2019;20(5):1224.
Sharma N, Chaudhary C, Khurana P. Wheat Myo-inositol phosphate synthase influences plant growth and stress responses via ethylene mediated signaling. Sci Rep. 2020;10(1):10766.
Conzelmann M, Williams EA, Tunaru S, Randel N, Shahidi R, Asadulina A, Berger J, Offermanns S, Jékely G. Conserved MIP receptor–ligand pair regulates Platynereis larval settlement. Proc Nat Acad Sci. 2013;110(20):8224–9.
Ramos-Figueroa JS, Palmer DR. Phosphonate and α-fluorophosphonate analogues of d-glucose 6-phosphate as active-site probes of 1l-myo-inositol 1-phosphate synthase. Biochemistry. 2022;61(10):868–78.
Luo Y, Qin G, Zhang J, Liang Y, Song Y, Zhao M, Tsuge T, Aoyama T, Liu J, Gu H. D-myo-inositol-3-phosphate affects phosphatidylinositol-mediated endomembrane function in Arabidopsis and is essential for auxin-regulated embryogenesis. Plant Cell. 2011;23(4):1352–72.
Ma L, Li G. FAR1-related sequence (FRS) and FRS-related factor (FRF) family proteins in Arabidopsis growth and development. Front Plant Sci. 2018;9:692.
Latrasse D, Jegu T, Meng P-H, Mazubert C, Hudik E, Delarue M, Charon C, Crespi M, Hirt H, Raynaud C. Dual function of MIPS1 as a metabolic enzyme and transcriptional regulator. Nucleic Acids Res. 2013;41(5):2907–17.
Ma L, Tian T, Lin R, Deng X-W, Wang H, Li G. Arabidopsis FHY3 and FAR1 regulate light-induced myo-inositol biosynthesis and oxidative stress responses by transcriptional activation of MIPS1. Mol Plant. 2016;9(4):541–57.
Fleet C, Yen J, Hill E, Gillaspy G. Co-suppression of AtMIPS demonstrates cooperation of MIPS1, MIPS2 and MIPS3 in maintaining myo-inositol synthesis. Plant Mol Biol. 2018;97:253–63.
Zhou T, Xing Q, Sun J, Wang P, Zhu J, Liu Z. The mechanism of KpMIPS gene significantly improves resistance of Koelreuteria paniculata to heavy metal cadmium in soil. Sci Total Environ. 2023;906:167219.
Karmakar A, Bhattacharya S, Sengupta S, Ali N, Sarkar SN, Datta K, Datta SK. RNAi-mediated silencing of ITPK gene reduces phytic acid content, alters transcripts of phytic acid biosynthetic genes, and modulates mineral distribution in rice seeds. Rice Sci. 2020;27(4):315–28.
Kashem MA, Hossain MZ. Climate-induced droughts and its implications for legume crops. In: Hossain MZ, Md Anawar H, Chaudhary DR, editors. Climate change and legumes: stress mitigation for sustainability and food security. 1st ed. Boca Raton: CRC Press; 2023. p. 12 ISBN 978-1-032-07984-4.
Thoppurathu FJ, Ghorbanzadeh Z, Vala AK, Hamid R, Joshi M. Unravelling the treasure trove of drought-responsive genes in wild-type peanut through transcriptomics and physiological analyses of root. Funct Integr Genomics. 2022;22(2):215–33.
Siddiq M, Uebersax MA, Siddiq F. Global production, trade, processing and nutritional profile of dry beans and other pulses. In: Siddiq M, Uebersax MA, editors. Dry beans and pulses: production, processing, and nutrition. 2nd ed. West Sussex: John Wiley and Sons; 2022. p. 1–28 ISBN:9781119777113.
Ku Y-S, Contador CA, Ng M-S, Yu J, Chung G, Lam H-M. The effects of domestication on secondary metabolite composition in legumes. Front Genet. 2020;11:581357.
Lebrazi S, Fikri-Benbrahim K. Potential of tree legumes in agroforestry systems and soil conservation. In: Meena RS, Kumar S, editors. Advances in legumes for sustainable intensification. Academic Press, Elsevier; 2022. p. 461–482. ISBN:978–0–323–85797–0.
Rathod V, Hamid R, Tomar RS, Padhiyar S, Kheni J, Thirumalaisamy P, Munshi NS. Peanut (Arachis hypogaea) transcriptome revealed the molecular interactions of the defense mechanism in response to early leaf spot fungi (Cercospora arachidicola). Plant Gene. 2020;23:100243.
Goodstein DM, Shu S, Howson R, Neupane R, Hayes RD, Fazo J, Mitros T, Dirks W, Hellsten U, Putnam N. Phytozome: a comparative platform for green plant genomics. Nucleic Acids Res. 2012;40(D1):D1178–86.
Mistry J, Chuguransky S, Williams L, Qureshi M, Salazar GA, Sonnhammer EL, Tosatto SC, Paladin L, Raj S, Richardson LJ. Pfam: The protein families database in 2021. Nucleic Acids Res. 2021;49(D1):D412–9.
Lu S, Wang J, Chitsaz F, Derbyshire MK, Geer RC, Gonzales NR, Gwadz M, Hurwitz DI, Marchler GH, Song JS. CDD/SPARCLE: the conserved domain database in 2020. Nucleic Acids Res. 2020;48(D1):D265–8.
Letunic I, Khedkar S, Bork P. SMART: recent updates, new developments and status in 2020. Nucleic Acids Res. 2021;49(D1):D458–60.
Duvaud S, Gabella C, Lisacek F, Stockinger H, Ioannidis V, Durinx C. Expasy, the Swiss bioinformatics resource portal, as designed by its users. Nucleic Acids Res. 2021;49(W1):W216–27.
Consortium U. UniProt: a worldwide hub of protein knowledge. Nucleic Acids Res. 2019;47(D1):D506–15.
Tamura K, Stecher G, Kumar S. MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol. 2021;38(7):3022–7.
Letunic I, Bork P. Interactive tree of life (iTOL) v5: an online tool for phylogenetic tree display and annotation. Nucleic Acids Res. 2021;49(W1):W293–6.
Bailey TL, Williams N, Misleh C, Li WW. MEME: discovering and analyzing DNA and protein sequence motifs. Nucleic Acids Res. 2006;34(suppl 2):W369–73.
Chen C, Chen H, Zhang Y, Thomas HR, Frank MH, He Y, Xia R. TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant. 2020;13(8):1194–202.
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S. PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res. 2002;30(1):325–7.
Kosugi S, Hasebe M, Tomita M, Yanagawa H. Systematic identification of cell cycle-dependent yeast nucleocytoplasmic shuttling proteins by prediction of composite motifs. Proc Nat Acad Sci. 2009;106(25):10171–6.
Törönen P, Holm L. PANNZER—a practical tool for protein function prediction. Protein Sci. 2022;31(1):118–28.
Horton P, Park K-J, Obayashi T, Fujita N, Harada H, Adams-Collier C, Nakai K. WoLF PSORT: protein localization predictor. Nucleic Acids Res. 2007;35(suppl 2):W585–7.
Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS. MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res. 2009;37(suppl 2):W202–8.
Shen Y, Liu J, Geng H, Zhang J, Liu Y, Zhang H, Xing S, Du J, Ma S, Tian Z. De novo assembly of a Chinese soybean genome. Sci China Life Sci. 2018;61:871–87.
Kumar A, Kumar V, Krishnan V, Hada A, Marathe A, Jolly M, Sachdev A. Seed targeted RNAi-mediated silencing of GmMIPS1 limits phytate accumulation and improves mineral bioavailability in soybean. Sci Rep. 2019; 9(1):7744.
Zhu J, Pan J, Wang X, Huang Y, Qin C, Qiao F, Qin J, Chen L. Alleviation of the adverse effect of dietary carbohydrate by supplementation of myo-inositol to the diet of nile tilapia (Oreochromis niloticus). Animals. 2020;10(11):2190.
Zhu J, Chen L, Huang Y, Zhang F, Pan J, Li E, Qin J, Qin C, Wang X. New insights into the influence of myo-inositol on carbohydrate metabolism during osmoregulation in Nile tilapia (Oreochromis niloticus). Anim Nutr. 2022;10:86–98.
Zhu J, Wang X, Bu X, Wang C, Pan J, Li E, Shi Q, Zhang M, Qin JG, Chen L. Relationship between myo-inositol synthesis and carbohydrate metabolism changes in Mozambique tilapia (Oreochromis mossambicus) under acute hypersaline stress. Aquaculture. 2021;532:736005.
Kurowska MM. TIP aquaporins in plants: Role in abiotic stress tolerance. In: Fahad S, Saud S, Chen Y, Wu C, Wang D, editors. Abiotic Stress in Plants. London: IntechOpen; 2020. p. 423–44. ISBN 978-1-83881-055-9.
Banerjee A, Roychoudhury A. The role of aquaporins during plant abiotic stress responses. In: Tripathi DK, Singh VP, Chauhan DK, Sharma S, Prasad SM, Dubey NK, Ramawat N, editors. Plant Life Under Changing Environment: Responses and Management. Oxford: Academic Press, Elsevier; 2020. p. 643–61. ISBN 978-0-12-818204-8.
Zhu Y-X, Yang L, Liu N, Yang J, Zhou X-K, Xia Y-C, He Y, He Y-Q, Gong H-J, Ma D-F. Genome-wide identification, structure characterization, and expression pattern profiling of aquaporin gene family in cucumber. BMC Plant Biol. 2019;19:345.
Ma A, Cui W, Wang X, Zhang W, Liu Z, Zhang J, Zhao T. Osmoregulation by the myo-inositol biosynthesis pathway in turbot Scophthalmus maximus and its regulation by anabolite and c-Myc. Comp Biochem Physiol A: Mol Integr Physiol. 2020;242:110636.
Park W, Scheffler BE, Bauer PJ, Campbell BT. Identification of the family of aquaporin genes and their expression in upland cotton (Gossypium hirsutum L.). BMC Plant Biol. 2010;10:142.
Saddhe AA, Mosa KA, Kumar K, Prasad M, Dhankher OP. Genome-wide characterization of major intrinsic protein (MIP) gene family in Brachypodium distachyon. Curr Bioinf. 2018;13(5):536–52.
Gangwar H, Kumari P, Gahlaut V, Kumar S, Jaiswal V. Identification and comprehensive analysis of MIPSs in Rosaceae and their expression under abiotic stresses in rose (Rosa chinensis). Front Plant Sci. 2022;13:1021297.
Forrest KL, Bhave M. Major intrinsic proteins (MIPs) in plants: a complex gene family with major impacts on plant phenotype. Funct Integr Genomics. 2007;7:263–89.
Qin D, Toyonaga D, Saneoka H. Characterization of myo-inositol-1-phosphate synthase (MIPS) gene expression and phytic acid accumulation in oat (Avena sativa) during seed development. Cereal Res Commun. 2022;50:379–84.
Dastidar KG, Chatterjee A, Chatterjee A, Majumder AL. Evolutionary divergence of L-myo-inositol 1-phosphate synthase: significance of a “core catalytic structure”. Biology of Inositols and Phosphoinositides: Subcellular Biochemistry. 2006:315–40.
Qiao X, Li Q, Yin H, Qi K, Li L, Wang R, Zhang S, Paterson AH. Gene duplication and evolution in recurring polyploidization–diploidization cycles in plants. Genome Biol. 2019;20:38.
Jithesh M, Shukla PS, Kant P, Joshi J, Critchley AT, Prithiviraj B. Physiological and transcriptomics analyses reveal that Ascophyllum nodosum extracts induce salinity tolerance in Arabidopsis by regulating the expression of stress responsive genes. J Plant Growth Regul. 2019;38:463–78.
Zhou Y, Sun M, Sun P, Gao H, Yang H, Jing Y, Hussain MA, Saxena RK, Carther FI, Wang Q. Tonoplast inositol transporters: Roles in plant abiotic stress response and crosstalk with other signals. J Plant Physiol. 2022;271:153660.
Zhai H, Wang F, Si Z, Huo J, Xing L, An Y, He S, Liu Q. A myo‐inositol‐1‐phosphate synthase gene, IbMIPS1, enhances salt and drought tolerance and stem nematode resistance in transgenic sweet potato. Plant Biotechnol J. 2016;14(2):592–602.
Khurana N, Chauhan H, Khurana P. Expression analysis of a heat-inducible, Myo-inositol-1-phosphate synthase (MIPS) gene from wheat and the alternatively spliced variants of rice and Arabidopsis. Plant Cell Rep. 2012;31:237–51.
Kishor KP, Venkatesh K, Amareshwari P, Kumari HP, Punita D, Kumar AS, Rani RA, Puppala N. Genetic engineering for salt and drought stress tolerance in peanut (Arachis hypogaea L.). Indian J Plant Physiol. 2018;23:647–52.
Smart LB, Moskal WA, Cameron KD, Bennett AB. MIP genes are down-regulated under drought stress in Nicotiana glauca. Plant Cell Physiol. 2001;42(7):686–93.
Alexandersson E, Danielson JÅ, Råde J, Moparthi VK, Fontes M, Kjellbom P, Johanson U. Transcriptional regulation of aquaporins in accessions of Arabidopsis in response to drought stress. Plant J. 2010;61(4):650–60.
Tan J, Wang C, Xiang B, Han R, Guo Z. Hydrogen peroxide and nitric oxide mediated cold-and dehydration-induced myo-inositol phosphate synthase that confers multiple resistances to abiotic stresses. Plant Cell Environ. 2013;36(2):288–99.
Acknowledgements
Authors thank Eng. Neda Hamid for her energetic help with data analysis and preparation of graphs.
Funding
This work is a part of the Ph.D. programme of the first author. The programme was supported by Indian Council of Agricultural Research (ICAR) through Senior Research Fellowship.
Author information
Authors and Affiliations
Contributions
Laboratory procedures, data analysis and writing the first draft of the manuscript were done by FJ and RH. Rest of the authors commented on previous versions of the manuscript. RV was involved in genome analysis. ZGH assisted in data analysis and visualization. LSA performed RT-qPCR. DM conceptualized the experiment, supervised the project, analysed data and finalized the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The experimental studies conducted on plants were in compliance with the relevant institutional, national, and international guidelines and legislation.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Table S1.
Detailed information of primers. Table S2. Legume MIPS proteins and their related information. Table S3. The detailed information of the motifs in MIPS proteins.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Jacob, F., Hamid, R., Ghorbanzadeh, Z. et al. Genome-wide identification, characterization, and expression analysis of MIPS family genes in legume species. BMC Genomics 25, 95 (2024). https://doi.org/10.1186/s12864-023-09937-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12864-023-09937-7