Abstract
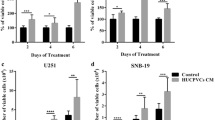
The influence of mesenchymal stem cells (MSC) on the growth of various tumors is ambiguous. MSC derived from different tissues stimulate growth of some tumor types, but have a prominent antitumor affect on other tumors. It has been reported recently that the outcome may be determined by crosstalk between tumor cells and MSC. The aim of this study was to examine the impact of MSC derived from fetal tissues on glioma cell proliferation during prolonged coculturing. We have analyzed the proliferative activity of glioma cells exposed to conditioned medium (CM) from MSC derived from fetal bone marrow (FetMSC), fetal muscle (M-FetMSC), and CM from cocultures of the fetal MSC with U251MG glioma cells. The effect was compared with the influence of CM from adult dermal fibroblasts (DF). Using MTT assay, we found that CM both from the fetal MSC and adult DF (without coculturing with glioma cells) had no effect on U251MG and A172 glioma cell proliferation. However, CM from early cocultures (3–9 days) of U251MG cells with FetMSC or M-FetMSC exerted a stimulatory effect on U251MG cell proliferation while CM taken from the same cocultures later (15–21 days) inhibited the cell proliferation. Adult DF displayed a persistent stimulation of U251MG cell proliferative activity. Immunofluorescence analysis revealed declined expression of cell cycle protein cyclin D1 in U251MG cells after their treatment with CM from 21-day cocultures of U251MG cells with FetMSC or M-FetMSC. In contrast, CM from 21-day cocultures of U251MG cells with DF did not decrease cyclin D1 expression. These results show that the effect of fetal MSC on glioma cell proliferation is ambivalent. Stimulation of proliferative activity was observed in the early period of cocultivation; however, inhibition of glioma cell proliferation was registered after 3 weeks of their coculturing with fetal MSC. This is the first report showing reversion of tumor cell proliferative program during long-term coculturing with MSC. These data suggest that CM obtained at different time points of coculturing may be used in the modeling of prolonged cellular crosstalk.
Similar content being viewed by others
Abbreviations
- DF:
-
dermal fibroblasts
- CM:
-
conditioned medium
- MSC:
-
mesenchymal stem cells
References
Akimoto, K., Kimur, K., Nagano, M., Takano, S., To’a Salazar, G., Yamashita, T., and Ohneda, O., Umbilical cord blood-derived mesenchymal stem cells inhibit, but adipose tissue-derived mesenchymal stem cells promote glioblastoma multiforme proliferation, Stem Cells Dev., 2013, vol. 22, pp. 1370–1386.
Baldin, V., Lukas, J., Marcote, M.J., Pagano, M., and Draetta, G., Cyclin D1 is a nuclear protein required for cell cycle progression in G1, Genes Dev., 1993, vol. 7, pp. 812–821.
Barbash, I.M., Chouraqui, P., Baron, J., Feinberg, M.S., Etzion, S., Tessone, A., Miller, L., Guetta, E., Zipori, D., Kedes, L.H., Kloner, R.A., and Leor, J., Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution, Circulation, 2003, vol. 108, pp. 863–868.
Bitsika, V., Vlahou, A., and Roubelakis, M.G., Fetal mesenchymal stem cells in cancer therapy, Curr. Stem Cell Res. Ther., 2013, vol. 8, pp. 133–143.
Brower, V., Search and destroy: recent research exploits adult stem cells’ attraction to cancer, J. Natl. Cancer Inst., 2005, vol. 97, pp. 414–416.
Burdon, T.J., Paul, A., Noiseux, N., Prakash, S., and Shum-Tim, D., Bone marrow stem cell derived paracrine factors for regenerative medicine: current perspectives and therapeutic potential, Bone Marrow Res., 2011, vol. 207326. doi: 10.1155/2011/207326
Chen, J., Li, Y., Wang, L., Zhang, Z., Lu, D., Lu, M., and Chopp, M., Therapeutic benefit of intravenous administration of bone marrow stromal cells after cerebral ischemia in rats, Stroke, 2001, vol. 32, pp. 1005–1011.
Ciavarella, S., Dominici, M., Dammacco, F., and Silvestris, F., Mesenchymal stem cells: new promise in anticancer therapy, Stem Cells Dev., 2011, vol. 20, pp. 1–10.
Dasari, V.R., Velpula, K.K., Kaur, K., Fassett, D., Klopfenstein, J.D., Dinh, D.H., Gujrati, M., and Rao, J.S., Cord blood stem cell-mediated induction of apoptosis in glioma downregulates X-linked inhibitor of apoptosis protein (XIAP), PLoS One, 2010, vol. 5, p. e11813.
Djouad, F., Plence, P., Bony, C., Tropel, P., Apparailly, F., Sany, J., Noël, D., and Jorgensen, C., Immunosuppressive effect of mesenchymal stem cells favors tumor growth in allogeneic animals, Blood, 2003, vol. 102, pp. 3837–3844.
Doucette, T., Rao, G., Yang, Y., Gumin, J., Shinojima, N., Bekele, B.N., Qiao, W., Zhang, W., and Lang, F.F., Mesenchymal stem cells display tumor-specific tropism in an RCAS/Ntv-a glioma model, Neoplasia, 2011, vol. 13, pp. 716–725.
Eterno, V., Zambelli, A., Pavesi, L., Villani, L., Zanini, V., Petrolo, G., Manera, S., Tuscano, A., and Amato, A., Adipose-derived mesenchymal stem cells (ASCs) may favour breast cancer recurrence via HGF/c-Met signaling, Oncotarget, 2014, vol. 5, pp. 613–633.
Götze, S., Wolter, M., Reifenberger, G., Müller, O., and Sievers, S., Frequent promoter hypermethylation of Wnt pathway inhibitor genes in malignant astrocytic gliomas, Int. J. Cancer., 2010, vol. 126, pp. 2584–2593.
Gregory, C.A., Singh, H., Perry, A.S., and Prockop, D.J., The Wnt signaling inhibitor Dickkopf-1 is required for reentry into the cell cycle of human adult stem cells from bone marrow, J. Biol. Chem., 2003, vol. 278, pp. 28067–28078.
Guo, Y., Harwalkar, J., Stacey, D.W., and Hitomi, M., Destabilization of cyclin D1 message plays a critical role in cell cycle exit upon mitogen withdrawal, Oncogene, 2005, vol. 24, pp. 1032–1042.
Han, X., Meng, X., Yin, Z., Rogers, A., Zhong, J., Rillema, P., Jackson, J.A., Ichim, T.E., Minev, B., Carrier, E., Patel, A.N., Murphy, M.P., Min, W.P., and Riordan, N.H., Inhibition of intracranial glioma growth by endometrial regenerative cells, Cell Cycle, 2009, vol. 8, pp. 606–610.
Hinds, P.W., Dowdy, S.F., Eaton, E.N., Arnold, A., and Weinberg, R.A., Function of a human cyclin gene as an oncogene, Proc. Natl. Acad. Sci. U.S.A., 1994, vol. 91, pp. 709–713.
Ho, I.A., Chan, K.Y., Ng, W.H., Guo, C.M., Hui, K.M., Cheang, P., and Lam, P.Y., Matrix metalloproteinase 1 is necessary for the migration of human bone marrow-derived mesenchymal stem cells toward human glioma, Stem Cells, 2009, vol. 27, pp. 1366–1375.
Hong, X., Miller, C., Savant-Bhonsale, S., and Kalkìanis, S.N., Antitumor treatment using nterleukin-12-secreting marrow stromal cells in an invasive glioma model, Neurosurgery, 2009, vol. 64, pp. 1139–1147.
Hou, L., Wang, X., Zhou, Y., Ma, H., Wang, Z., He, J., Hu, H., Guan, W., and Ma, Y., Inhibitory effect and mechanism of mesenchymal stem cells on liver cancer cells, Tumour Biol., 2014, vol. 35, pp. 1239–1250.
Jiao, H., Guan, F., Yang, B., Li, J., Shan, H., Song, L., Hu, X., and Du, Y., Human umbilical cord blood-derived mesenchymal stem cells inhibit c6 glioma via downregulation of cyclin D1, Neurol. India, 2011, vol. 59, pp. 241–247.
Jiao, H., Guan, F., Yang, B., Li, J., Song, L., Hu, X., and Du, Y., Human amniotic membrane derived-mesenchymal stem cells induce C6 glioma apoptosis in vivo through the Bcl-2/caspase pathways, Mol. Biol. Rep., 2012, vol. 39, pp. 467–473.
Joshi, M., Patil, P., He, Z., Holgersson, J., Olausson, M., and Sumitran-Holgersson, S., Fetal liver-derived mesenchymal stromal cells augment engraftment of transplanted hepatocytes, Cytotherapy, 2012, vol. 14, pp. 657–669.
Kang, S.G., Jeun, S.S., Lim, J.Y., Kim, S.M., Yang, Y.S., Oh, W.I., Huh, P.W., and Park, C.K., Cytotoxicity of human umbilical cord blood-derived mesenchymal stem cells against human malignant glioma cells, Childs Nerv. Syst., 2008, vol. 24, pp. 293–302.
Khakoo, A.Y., Pati, S., Anderson, S.A., Reid, W., Elshal, M.F., Rovira, I.I., Nguyen, A.T., Malide, D., Combs, C.A., Hall, G., Zhang, J., Raffeld, M., Rogers, T.B., Stetler-Stevenson, W., Frank, J.A., Reitz, M., and Finkel, T., Human mesenchymal stem cells exert potent antitumorigenic effects in a model of Kaposi’s sarcoma, J. Exp. Med., 2006, vol. 203, pp. 1235–1247.
Krylova, T.A., Musorina, A.S., Zenin, V.V., Yakovleva, T.K., and Poljanskaya, G.G., Comparative characteristics of mesenchymal stem cell lines derived from bone marrow and muscle of limb of early human embryo, Tsitologiia, 2014, vol. 56, no. 8, pp. 562–573.
Kucerova, L., Matuskova, M., Hlubinova, K., Altanerova, V., and Altaner, C., Tumor cell behaviour modulation by mesenchymal stromal cells, Mol. Cancer, 2010, vol. 9, p. 129. doi: 10.1186/1476-4598-9-129
Li, G., Wang, R., Gao, J., Deng, K., Wei, J., and Wei, Y., RNA interference-mediated silencing of iASPP induces cell proliferation inhibition and G0/G1 cell cycle arrest in U251 human glioblastoma cells, Mol. Cell. Biochem., 2011, vol. 350, pp. 193–200.
Lu, Y.R., Yuan, Y., Wang, X.J., Wei, L.L., Chen, Y.N., Cong, C., Li, S.F., Long, D., Tan, W.D., Mao, Y.Q., Zhang, J., Li, Y.P., and Cheng, J.Q., The growth inhibitory effect of mesenchymal stem cells on tumor cells in vitro and in vivo, Cancer Biol. Ther., 2008, vol. 7, pp. 245–251.
Ma, S., Liang, S., Jiao, H., Chi, L., Shi, X., Tian, Y., Yang, B., and Guan, F., Human umbilical cord mesenchymal stem cells inhibit C6 glioma growth via secretion of Dickkopf-1 (DKK1), Mol. Cell. Biochem., 2014, vol. 385, pp. 277–286.
Menon, L.G., Kelly, K., Yang, H.W., Kim, S.K., Black, P.M., and Carroll, R.S., Human bone marrow-derived mesenchymal stromal cells expressing S-TRAIL as a cellular delivery vehicle for human glioma therapy, Stem Cells, 2009, vol. 27, pp. 2320–2330.
Mishra, P.J., Mishra, P.J., Humeniuk, R., Medina, D.J., Alexe, G., Mesirov, J.P., Ganesan, S., Glod, J.W., and Banerjee, D., Carcinoma-associated fibroblast-like differentiation of human mesenchymal stem cells, Cancer Res., 2008, vol. 68, pp. 4331–4339.
Nakamizo, A., Marini, F., Amano, T., Khan, A., Studeny, M., Gumin, J., Chen, J., Hentschel, S., Vecil, G., Dembinski, J., Andreeff, M., and Lang, F.F., Human bone marrow-derived mesenchymal stem cells in the treatment of gliomas, Cancer Res., 2005, vol. 65, pp. 3307–3318.
Phinney, D.G. and Sensebé, L., Mesenchymal stromal cells: misconceptions and evolving concepts, Cytotherapy, 2013, vol. 15, pp. 140–145.
Qiao, L., Xu, Z.L., Zhao, T.J., Ye, L.H., and Zhang, X.D., Dkk-1 secreted by mesenchymal stem cells inhibits growth of breast cancer cells via depression of Wnt signalling, Cancer Lett., 2008a, vol. 269, pp. 67–77.
Qiao, L., Xu, Z., Zhao, T., Zhao, Z., Shi, M., Zhao, R.C., Ye, L., and Zhang, X., Suppression of tumorigenesis by human mesenchymal stem cells in a hepatoma model, Cell Res., 2008b, vol. 18, pp. 500–507.
Reya, T. and Clevers, H., Wnt signalling in stem cells and cancer, Nature, 2005, vol. 434, pp. 843–850.
Rhodes, L.V., Antoon, J.W., Muir, S.E., Elliott, S., Beckman, B.S., and Burow, M.E., Effects of human mesenchymal stem cells on ER-positive human breast carcinoma cells mediated through ER-SDF-1/CXCR4 crosstalk, Mol. Cancer, 2010, vol. 9, p. 295. doi: 10.1186/1476-4598-9-295
Sallinen, S.L., Sallinen, P.K., Kononen, J.T., Syrjäkoski, K.M., Nupponen, N.N., Rantala, I.S., Helén, P.T., Helin, H.J., and Haapasalo, H.K., Cyclin D1 expression in astrocytomas is associated with cell proliferation activity and patient prognosis, J. Pathol., 1999, vol. 188, pp. 289–293.
Sasportas, L.S., Kasmieh, R., Wakimoto, H., Hingtgen, S., van de Water, J.A., Mohapatra, G., Figueiredo, J.L., Martuza, R.L., Weissleder, R., and Shah, K., Assessment of therapeutic efficacy and fate of engineered human mesenchymal stem cells for cancer therapy, Proc. Natl. Acad. Sci. USA, 2009, vol. 106, pp. 4822–4827.
Surawicz, T.S., Davis, F., Freels, S., Laws, E.R.Jr., and Menck, H.R., Brain tumor survival: results from the national cancer data base, J. Neurooncol., 1998, vol. 40, pp. 151–160.
Takahara, K., Ii, M., Inamoto, T., Komura, K., Ibuki, N., Minami, K., Uehara, H., Hirano, H., Nomi, H., Kiyama, S., Asahi, M., and Azuma, H., Adipose-derived stromal cells inhibit prostate cancer cell proliferation inducing apoptosis, Biochem. Biophys. Res. Commun., 2014, vol. 446, pp. 1102–1107.
Van Meir, E.G., Hadjipanayis, C.G., Norden, A.D., Shu, H.K., Wen, P.Y., and Olson, J.J., Exciting new advances in neuro-oncology: the avenue to a cure for malignant glioma, CA Cancer J. Clin., 2010, vol. 60, pp. 166–193.
Velpula, K.K., Dasari, V.R., Tsung, A.J., Gondi, C.S., Klopfenstein, J.D., Mohanam, S., and Rao, J.S., Regulation of glioblastoma progression by cord blood stem cells is mediated by downregulation of cyclin D1, PLoS One, 2011, vol. 6, p. e18017. doi: 10.1371/journal.pone.0018017
Wang, Y., Zhang, Y., Qian, C., Cai, M., Li, Y., Li, Z., You, Q., Wang, Q., Hu, R., and Guo, Q., GSK3β/β-catenin signaling is correlated with the differentiation of glioma cells induced by wogonin, Toxicol. Lett., 2013, vol. 222, pp. 212–223.
Xu, F., Shi, J., Yu, B., Ni, W., Wu, X., and Gu, Z., Chemokines mediate mesenchymal stem cell migration toward gliomas in vitro, Oncol. Rep., 2010, vol. 23, pp. 1561–1567.
Ye, H., Cheng, J., Tang, Y., Liu, Z., Xu, C., Liu, Y., and Sun, Y., Human bone marrow-derived mesenchymal stem cells produced TGFbeta contributes to progression and metastasis of prostate cancer, Cancer Invest., 2012, vol. 30, pp. 513–518.
Yu, J.M., Jun, E.S., Bae, Y.C., and Jung, J.S., Mesenchymal stem cells derived from human adipose tissues favor tumor cell growth in vivo, Stem Cells Dev., 2008, vol. 17, pp. 463–473.
Zhang, T., Lee, Y.W., Rui, Y.F., Cheng, T.Y., Jiang, X.H., and Li, G., Bone marrow-derived mesenchymal stem cells promote growth and angiogenesis of breast and prostate tumors, Stem Cell Res. Ther., 2013, vol. 4, p. 70.
Zhou, Y., Liu, F., Xu, Q., and Wang, X., Analysis of the expression profile of Dickkopf-1 gene in human glioma and the association with tumor malignancy, Exp. Clin. Cancer Res. 29: 138. doi:, 2010, vol. 101186/1756-9966-29-138.
Zhu, W., Huang, L., Li, Y., Qian, H., Shan, X., Yan, Y., Mao, F., Wu, X., and Xu, W.R., Mesenchymal stem cell-secreted soluble signaling molecules potentiate tumor growth, Cell Cycle, 2011, vol. 10, pp. 3198–3207.
Zimmerlin, L., Park, T.S., Zambidis, E.T., Donnenberg, V.S., and Donnenberg, A.D., Mesenchymal stem cell secretome and regenerative therapy after cancer, Biochimie, 2013, vol. 95, pp. 2235–2245.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.A. Chistyakova, G.G. Poljanskaya, 2014, published in Tsitologiya, 2014, Vol. 56, No. 11, pp. 800–808.
Rights and permissions
About this article
Cite this article
Chistyakova, I.A., Poljanskaya, G.G. The influence of human fetal mesenchymal stem cells on glioma cell proliferation. The consequence of cellular crosstalk. Cell Tiss. Biol. 9, 71–78 (2015). https://doi.org/10.1134/S1990519X15020042
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X15020042