Abstract
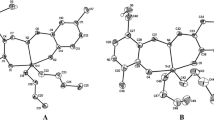
Dinuclear dibutyltin complexes {[2-O-C6H4(O)C=N-N=C(O)CPh2O](n-Bu2Sn)2}n (C1) and {[2-(3-O-C8H6)(O)C=N-N=C-2-(3-O-C8H6)]2(n-Bu2Sn)2}n (C2) have been synthesized and characterized by IR, 1H, 13C, and 119Sn NMR spectra, HRMS, elemental analysis, and thermal stability analysis. Their crystal structures have been determined by X-ray diffraction. In vitro antitumor activities of both complexes have been evaluated by the CCK8 method against three human cancer cell lines (MCF-7, NCI-H460, and HepG2), and the complexes have exhibited high activity against MCF-7 and HepG2.




Similar content being viewed by others
REFERENCES
Sharma, S., Agnihotri, N., Kumar, K., Sihag, S., Randhawa, V., Kaur, R., Singh, R. and Kaur, V., Appl. Organomet. Chem., 2022, vol. 36, p. e6521. https://doi.org/10.1002/aoc.6521
Ullah, H., Previtali, V., Mihigo, H.B., Twamley, B., Rauf, M.K., Javed, F., Waseem, A., Baker, R.J., and Rozas, I., Eur. J. Med. Chem., 2019, vol. 181, p. 111544. https://doi.org/10.1016/j.ejmech.2019.07.047
Villegas-Villalobos, S., Díaz, L.E., Vilariño-Feltrer, G., Vallés-Lluch, A., Gómez-Tejedor, J.A., and Valero, M.F., J. Mater. Res., 2018, vol. 33, p. 2598. https://doi.org/10.1557/jmr.2018.286
Minke, R., Freireich, S. and Zilkha, A., Isr. J. Chem., 1975, vol. 13, p. 212. https://doi.org/10.1002/ijch.197500031
Karpel, S., Pigm. Resin Technol., 1988, vol. 17, p. 13. https://doi.org/10.1108/eb042446
King, S., Pigm. Resin Technol., 1980, vol. 9, p. 8. https://doi.org/10.1108/eb041630
Aguilar-Santelises, M., Castillo-Vera, J., GonzalezMolina, R., Garcia del Valle, A., Cruz Millan, M., and Aguilar-Santelises, L., Folia Microbiol., 2020, vol. 65, p. 87. https://doi.org/10.1007/s12223-019-00707-1
Hu, L., Wang, H., Xia, T., Fang, B., Shen, Y., Zhang, Q., Tian, X., Zhou, H., Wu, J., and Tian, Y., Inorg. Chem., 2018, vol. 57, p. 6340. https://doi.org/10.1021/acs.inorgchem.8b00413
Jain, M., Maanju, S., and Singh, R.V., Appl. Organomet. Chem., 2004, vol. 18, p. 471. https://doi.org/10.1002/aoc.711
Ahmad, N.W., Mohd, S.-A., Balabaskaran, S., and Das, V.G.K., Appl. Organomet. Chem., 1993, vol. 7, p. 583. https://doi.org/10.1002/aoc.590070719
Zarafu, I., Badea, M., Ioniţă, G., Chifiriuc, M.C., Bleotu, C., Popa, M., Ioniţă, P., Tatibouët, A., and Olar, R., J. Therm. Anal. Calorim., 2019, vol. 136, p. 1977. https://doi.org/10.1007/s10973-018-7853-z
Adeyemi, J.O., Onwudiwe, D.C., Ekennia, A.C., Anokwuru, C.P., Nundkumar, N., Singh, M., and Hosten, E.C., Inorg. Chim. Acta, 2019, vol. 485, p. 64. https://doi.org/10.1016/j.ica.2018.09.085
Adeyemi, J.O. and Onwudiwe, D.C., Molecules, 2018, vol. 23, p. 2571. https://doi.org/10.3390/molecules23102571
Hussain, S., Ali, S., Shahzadi, S., Sharma, S.K., Qanungo, K., and Shahid, M., Bioinorg. Chem. Appl., 2014, vol. 2014, p. 959203. https://doi.org/10.1155/2014/959203
Khan, S., Tariq, M., Ashraf, M., Abdullah, S., Al-Rashida, M., Khalid, M., Taslimi, P., Fatima, M., Zafar, R., and Shafiq, Z., Bioorg. Chem., 2020, vol. 102, p. 104082. https://doi.org/10.1016/j.bioorg.2020.104082
Filipović, N.R., Elshaflu, H., Grubišić, S., Jovanović, L.S., Rodić, M., Novaković, I., Malešević, A., Djordjević, I.S., Li, H., Šojić, N., Marinković, A., and Todorović, T.R., Dalton Trans., 2017, vol. 46, p. 2910. https://doi.org/10.1039/c6dt04785h
Ayyannan, G., Mohanraj, M., Raja, G., Bhuvanesh, N., Nandhakumar, R., and Jayabalakrishnan, C., J. Photochem. Photobiol., B, 2016, vol. 163, p. 1. https://doi.org/10.1016/j.jphotobiol.2016.08.003
Omidi, S. and Kakanejadifard, A., RSC Adv., 2020, vol. 10, p. 30186. https://doi.org/10.1039/d0ra05720g
de Oliveira Carneiro Brum, J., França, C.C.T., LaPlante, R.S., and Villar, D.F.J., Mini-Rev. Med. Chem., 2020, vol. 20, p. 342. https://doi.org/10.2174/1389557519666191014142448
Rollas, S. and Küçükgüzel, S. G., Molecules, 2007, vol. 12, p. 1910. https://doi.org/10.3390/12081910
Tan, Y.X., Zhang, Z.J., Liu, Y., Yu, J.X., Zhu, X.M., Kuang, D.Z., and Jiang, W.J., J. Mol. Struct., 2017, vol. 1149, p. 874. https://doi.org/10.1016/j.molstruc.2017.08.058
Tan, Y.-X., Zhang, Z.-J., Feng, Y.-L., Yu, J.-X., Zhu, X.-M., Zhang, F.-X., Kuang, D.-Z., and Jiang, W.-J., J. Inorg. Organomet. P., 2017, vol. 27, p. 342. https://doi.org/10.1007/s10904-016-0477-5
El-bendary, M. M. and Etaiw, S. E.-d.H., Appl. Organomet. Chem., 2018, vol. 32, p. e4152. https://doi.org/10.1002/aoc.4152
Pretsch, E., Bühlmann, P., and Badertscher, M., Structure Determination of Organic Compounds: Tables of Spectral Data, Germany Berlin: Springer-Verlag, 2009
Basu Baul, T.S., Addepalli, M.R., Lyčka, A., van Terwingen, S., and Englert, U., J. Organomet. Chem., 2020, vol. 927, p. 121522. https://doi.org/10.1016/j.jorganchem.2020.121522
Nath, M., Mridula, and Kumari, R., J. Photochem. Photobiol. B, 2017, vol. 174, p. 182. https://doi.org/10.1016/j.jphotobiol.2017.07.017
Arjmand, F. and Yousuf, I., J. Organomet. Chem., 2013, vol. 743, p. 55. https://doi.org/10.1016/j.jorganchem.2013.06.018
Sheldrick, G.M., SHELXL-97, A Program for Crystal Structure Refinement. Germany Geöttingen: University of Geöttingen, 1997.
Funding
This work was supported by the 22nd College students Extracurricular Academic Science and Technology Works Competition Project of Hengyang Normal University (no. 13).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
No conflict of interest was declared by the authors.
Rights and permissions
About this article
Cite this article
Li, A., Wang, T., Feng, Y. et al. Synthesis, Crystal Structure, and Anticancer Activity of the Dinuclear Dibutyltin Complexes. Russ J Gen Chem 92, 908–913 (2022). https://doi.org/10.1134/S1070363222050218
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222050218