Abstract
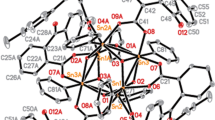
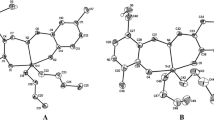
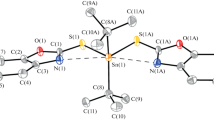
Two new 1D Chain benzyltin complexes {[C6H4(O)C=N–N=C(Me)COO](CH3OH)(C6H5CH2)2Sn}n (1), {[C6H4(O)C–NH–N=C(Me)COO](C6H5CH2)3Sn}n (2) were synthesized by the reactions of 2-oxo-propionic acid benzoyl hydrazone with dibenzyltin dichloride or tribenzyltin chloride, respectively. The two complexes were characterized by FT-IR, 1H, 13C and 119Sn NMR spectra, elemental analysis, X-ray single crystal diffraction and TGA. In vitro antitumor activities of both complexes were evaluated by the 3-(4,5-dimethylthiazoly-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay against three human cancer cell lines (NCI-H460, HepG2, MCF7) and human cell line (HL7702). The 1 exhibited strong antitumor activity than 2, then it was expected after further chemical optimization of candidate compounds as anti-cancer drugs. The interaction between complexes and calf thymus DNA were studied by EB fluorescent probe, the interaction of 1 and 2 with calf thymus DNA were intercalation and electrostatic attraction.










Similar content being viewed by others
References
N.M. Brown, Thesis for the Doctorate of Clemson University (United States, South Carolina, 1972)
R.F. Zhang, J.J. Nie, P.Z. Yan, Q.L. Li, S.L. Zhang, C.L. Ma, J. Organomet. Chem. 819, 11 (2016)
M.A. Salam, M.A. Hussein, I. Ramli, S.M. Islam. J. Organomet. Chem. 813, 71 (2016)
C.E. Carraher Jr., M.R. Roner, J. Organomet. Chem. 751, 67 (2014)
X. Shang, B. Zhao, G. Xiang, M.F.C. Guedes da Silva, A.J.L. Pombeiro, RSC Adv. 5, 45053 (2015)
M. Gajewska, K.V. Luzyanin, M.F.C. Guedes da Silva, Q. Li, J. Cui, A.J.L. Pombeiro, Eur. J. Inorg. Chem. 2009, 3765 (2009)
M. Nath, P.K. Saini, A. Kumar, Appl. Organomet. Chem. 23, 434 (2009)
A. Silva, D. Luís, S. Santos, J. Silva, A.S. Mendo, L. Coito, T.F.S. Silva, M.F.C.G. da Silva, L.M.D.R.S. Martins, A.J.L. Pombeiro, P.M. Borralho, C.M.P. Rodrigues, M.G. Cabral, P.A. Videira, C. Monteiro, A.R. Fernandes, Drug Metab. Drug Interact. 28, 167 (2013)
A. Ray, C. Rizzoli, G. Pilet, C. Desplanches, E. Garribba, E. Rentschler, S. Mitra, Eur. J. Inorg. Chem. 2009, 2915 (2009)
E. Jayanthi, S. Kalaiselvi, V.V. Padma, N.S.P. Bhuvanesh, N. Dharmaraj, Dalton Trans. 45, 1693 (2016)
U. Golla, A. Adhikary, A.K. Mondal, S.R. Tomar, S. Konar, Dalton Trans. 45, 11849 (2016)
H.G. Fu, X.X. Hu, C.R. Li, Y.H. Li, Y.X. Wang, J.D. Jiang, C.W. Bi, S. Tang, X.F. You, D.Q. Song, Eur. J. Med. Chem. 110, 151 (2016)
E. Tatar, İ.D. Küçükgüzel, Daelemans, T.T. Talele, N. Kaushik Basu, E. De Clercq, C. Pannecouque, Arch. Pharm. 346, 140 (2013)
M. Mohanraj, G. Ayyannan, G. Raja, C. Jayabalakrishnan, J. Photoch. Photobio. B. 158, 164 (2016)
S.Y. He, W.K. Cao, J.L. Chen, J.S. Zhao, Q.Z. Shi, R.X. Wang, J. Sun, Chem. J. Chinese Universities 23, 991 (2002)
K. Sisido, Y. Takeda, Z. Kinugawa, J. Am. Chem. Soc. 83, 538 (1961)
W.L.F. Armarego, C.C.L. Lin, Purification of Laboratory Chemicals, 6th edn. (Elsevier, Oxford, 2009)
G.M. Sheldrick, SHELXL-97, A Program for Crystal Structure Refinement. (University of Geöttingen, Germany, 1997)
L.J. Farrugia, WINGX: A Windows Program for Crystal Structure Analysis. (University of Glasgow, Germany, 1988)
S. Sun, J. Yang, Acta Crystallogr. E. 67, m544 (2011)
H.D. Yin, S.W. Chen, L.W. Li, D.Q. Wang, Inorg. Chim. Acta 360, 2215 (2007)
W.J. Jiang, N.F. Yang, D.Z. Kuang, Y.L. Feng, F.X. Zhang, J.Q. Wang, M.Q. Liu, J.X. Yu, Chinese J. Struct. Chem. 30, 1327 (2011)
L. Labib, T.E. Khalil, M.F. Iskander, S.L. Refaat, Polyhedron 15, 3697 (1996)
J.M. Rivera, D. Guzmán, M. Rodriguez, J.F. Lamère, K. Nakatani, R. Santillan, P.G. Lacroix, N. Farfán, J. Organomet. Chem. 691, 1722 (2006)
Y.L. Feng, J.H. Zheng, W.J. Jiang, Y.X. Tan, J.X. Yu, Z.J. Zhang, F.X. Zhang, D.Z. Kuang, Chinese J. Inorg. Chem. 12, 2767 (2014)
F. Wang, H. Yin, J. Cui, Y. Zhang, H. Geng, M. Hong, J. Organomet. Chem. 759, 83 (2014)
J.M. Hu, Y.G. Liu, Z.C. Hao, G.-H. Cui, J. Inorg. Organomet. Polym. 26, 598 (2016)
F. Javed, S. Ali, S. Shahzadi, S.K. Sharma, K. Qanungo, M.N. Tahir, N.A. Shah, M.R. Khan, N. Khalid, J. Inorg. Organomet. Polym. 26, 48 (2016)
G. Deacon, R. Phillips, Coord. Chem. Rev. 33, 250 (1980)
C. Ma, J. Zhang, G. Tian, R. Zhang, J. Organomet. Chem. 690, 519 (2005)
V. Barba, J. Zaragoza, H. Höpfl, N. Farfán, H.I. Beltrán, L.S. Zamudio-Rivera, J. Organomet. Chem. 696, 1949 (2011)
C. Vatsa, V.K. Jain, T.K. Das, E.R.T. Tiekink, J. Organomet. Chem. 396, 9 (1990)
E. Pretsch, P. Buhlmann, B. Martin, Structure Determination of Organic Compounds, Tables of Spectral Data, 4th edn. (Springer-Verlag, Berlin Heidelberg, 2009)
A.G. Davies, Organotin Chemistry, 2nd edn. (Wiley VCH Verlag GmbH & Co. KGaA, Berlin, 2004)
H.C. Clark, V.K. Jain, R.C. Mehrotra, B.P. Singh, G. Srivastava, T. Birchall, J. Organomet. Chem. 279, 385 (1985)
M. Gielen, M. Biesemans, R. Willem, Appl. Organometal. Chem. 19, 440 (2005)
W. Li, D. Du, S. Liu, C. Zhu, A.M. Sakho, D. Zhu, L. Xu, J. Organomet. Chem. 695, 2153 (2010)
M. Khandani, T. Sedaghat, N. Erfani, M.R. Haghshenas, H.R. Khavasi, J. Mol. Struc. 1037, 136 (2013)
Q. Li, M.F.C. Guedes da Silva, Z. Jinghua, A.J.L. Pombeiro, J. Organomet. Chem. 689, 4584 (2004)
G. Ruisi, A. Silvestri, M.T. Lo Giudice, R. Barbieri, G. Atassi, F. Huber, K. Grätz, L. Lamartina, J. Inorg. Biochem. 25, 229 (1985)
X.Y. Wu, J.F. Liu, G.L. Zhao, Chinese J. Inorg. Chem. 28, 1661 (2012)
A. Rambabu, M. Pradeep Kumar, S. Tejaswi, N. Vamsikrishna, Shivaraj, J. Photoch. Photobio. B. 165, 147 (2016)
M. Mohanraj, G. Ayyannan, G. Raja, C. Jayabalakrishnan, Mater. Sci. Eng. C. 69, 1297 (2016)
A. Raza, X. Xu, L. Xia, C. Xia, J. Tang, Z. Ouyang, J. Fluoresc. 26, 2023 (2016)
J.G. Xu, Z.B. Wang, Fluorescence Analysis, 3rd edn. (Science Press, Beijing, 2006)
B. W. Zhang, B. Li, W. S. Xia, L. Ding, Chinese ProcPharm. Sci. 35, 296 (2011)
P. Yang, F. Gao, Principle of Biological Inorganic Chemistry. (Science Press, Beijing, 2002)
M. Hong, G. Chang, R. Li, M. Niu, New J. Chem. 40, 7889 (2016)
H. Yin, H. Liu, M. Hong, J. Organomet. Chem. 713, 11 (2012)
C. Yan, J. Zhang, T. Liang, Q. Li, Biomed. Pharmacother. 71, 119 (2015)
Acknowledgements
The project was supported by the Scientific and Technological Projects of Hunan Province of China (No. 2015JC3060), Hunan Provincial Natural Science Foundation of China (No. 2016JJ4008) and The Innovation Platform Open Fund Project of Hunan Provincial Education Department of China (No. 16K011).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tan, YX., Zhang, ZJ., Feng, YL. et al. Syntheses, Crystal Structures and Biological Activity of the 1D Chain Benzyltin Complexes Based on 2-Oxo-Propionic Acid Benzoyl Hydrazone. J Inorg Organomet Polym 27, 342–352 (2017). https://doi.org/10.1007/s10904-016-0477-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10904-016-0477-5