Abstract
We studied the chemical composition of the phenolic complex and the structure of oligomeric lignans in Siberian spruce (Picea obovata Ledeb.). We used the wood of Siberian spruce collected near the city of Irkutsk. The extractives were isolated from ground wood (particle size: 10–15 mm; humidity: 5.9%) by three-stage acetone extraction. The extract was separated by consecutive treatment with the solvents with increasing polarity: hexane, ethyl acetate and n-butanol. The main amount of phenolic compounds (lignans) was concentrated in the ethyl acetate fraction and it was 0.7% in absolutely dry wood (adw).
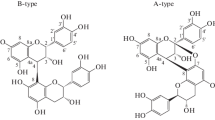
The ethyl acetate fraction of spruce wood extract was separated by silica gel column; a chloroform-acetone mixture was used as the eluent (the concentration of acetone in the mixture was increased from 0 to 100%). Monomeric lignans (~60–65% of the ethyl acetate fraction of spruce wood extract), oligomeric lignans (~20–25%), and polymer lignans (~12–15%) were isolated.
We also obtained 13C nuclear magnetic resonance (NMR) spectra for the main monomeric, oligomeric and polymeric lignan compounds of phenolic complexes. It was found that oligomeric and polymeric fractions contain monomeric lignan units with the butyrolactone cycle, primarily, the fragments with hydroxymatairesinol structure. Oligomeric lignans contain the fragments with a pinoresinol and lariciresinol structure. All the monomeric structural units are characterized by the guaiacyl substitution type of the aromatic cycles.
A preliminary study has been performed on the antiviral and antioxidant activity of the ethyl acetate fraction of acetone extract of Siberian spruce wood. It was found that the lignan complex is active against Coxsackie B4 virus in cell culture and in the pancreatitis model in white mice, reducing the activity of enteroviruses in the cell culture approximately 100 times. The antioxidant activity rate of polyphenol complex of Siberian spruce wood is comparable to that of a known antioxidant dihydroquercetin.
Similar content being viewed by others
References
Babkin, V.A., Khim. Rastit. Syr’ya, 2014, no. 3, pp. 111–119.
Babkin, V.A., Ostroukhova, L.A., and Trofimova, N.N., Biomassa listvennitsy: ot khimicheskogo sostava do innovatsionnykh produktov (Larch Biomass: From the Chemical Composition to Innovative Products), Novosibirsk, 2011.
Neverova, N.A., Levchuk, A.A., Medvedeva, E.N., Ostroukhova, L.A., Onuchina, N.A., Golobokova, G.M., and Babkin, V.A., Russ. J. Bioorg. Chem., 2014, vol. 40, no. 7, pp. 762–770.
Levdanskii, V.A., Butylkina, A.I., Ivanchenko, N.M., and Kuznetsov, B.N., Khim. Rastit. Syr’ya, 2011, no. 1, pp. 93–99.
Eklund, P., Sillanpaa, R., and Sjoholm, R.J., Chem. Soc., Perkin Trans., 2002, no. 16, pp. 1906–1910.
Pietarinen, S.P., Willfor, S.M., Ahotupa, M.O., Hemming, J.E., and Holmbom, B.R., J. Wood Sci., 2006, vol. 52, pp. 436–444.
Modonova, L.D., Tyukavkina, N.A., and Shostakovskii, M.F., Khim. Prirod. Soed., 1969, no. 1, pp. 67–68.
Modonova, L.D., Leont’eva, V.G., and Tyukavkina, N.A., Khim. Prirod. Soed., 1970, no. 4, p. 477.
Medvedeva, S.A., Modonova, L.D., Leont’eva, V.G., Glazkova, V.N., and Tyukavkina, N.A., Khim. Prirod. Soed., 1971, no. 1, pp. 113–114.
Modonova, L.D., Voronov, V.K., Leont’eva, V.G., and Tyukavkina, N.A., Khim. Prirod. Soed., 1972, no. 2, pp. 165–170.
Modonova, L.D. and Tiukavkina, N.A., Khimiia i ispol’zovanie lignina (Chemistry and the Use of Lignin), Riga, 1974, pp. 73–86.
Leont’eva, V.G., Phenolic compounds of wood of some species of the family Pinaceae, Extended Abstract of Cand. Sci. (Chem.) Dissertation, Irkutsk, 1978.
Agrawal, P.K. and Thakur, R.S., Magn. Reson. Chem., 1985, vol. 23, no. 6, pp. 389–418.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.E. Fedorova, S.V. Fedorov, V.A. Babkin, 2015, published in Khimiya Rastitel’nogo Syr’ya, 2015, No. 3, pp. 49–53.
Rights and permissions
About this article
Cite this article
Fedorova, T.E., Fedorov, S.V. & Babkin, V.A. Oligolignans in the wood of Picea obovata Ledeb.. Russ J Bioorg Chem 42, 712–715 (2016). https://doi.org/10.1134/S1068162016070062
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162016070062