Abstract
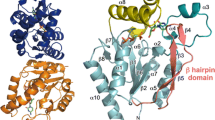
The structure of bacterial uridine phosphorylase (UPh) belonging to the NP-I family in complex with 6-methyluracil was determined for the first time at 1.17 Å resolution. The structural features of bacterial UPh from the bacterium Vibrio cholerae (VchUPh) responsible for selectivity toward 6-methyluracil acting as a pseudosubstrate were revealed. The repulsion between the hydrophilic hydroxyl group of the active-site residue Thr93 of VchUPh and the hydrophobic methyl group of 6-methyluracil prevents the oxygen atom O4' of the ribose moiety and the phosphate oxygen atom O3P of ribose 1-phosphate from forming hydrogen bonds with OG1_Thr93, which are essential for the enzymatic reaction. This, apparently, makes VchUPh inactive in the enzymatic synthesis of 6-methyluridine from 6-methyluracil. Hence, Thr93 is the residue, the modification of which will allow VchUPh to catalyze the biotechnologically important synthesis of 6-methyluridine from 6-methyluracil. Taking into account high structural homology of the functionally significant regions of bacterial UPhs, this conclusion is also true for other bacterial UPhs. It was demonstrated that bacterial thymidine phosphorylases of the NP-II family cannot bind 6-methyluracil in a proper conformation required for the catalysis because of a close contact between the 6-methyl group and Phe210.
Similar content being viewed by others
References
A. P. Chadaev and A. D. Klimiashvili, Khirurgiia (Mosk), No. 1, 54 (2003).
J. el-On and L. Weinrauch, Int. J. Dermatol. 29 (3), 232 (1990).
V. V. Ganzhii, Klin. Khir., No. 11–12, 18 (2002).
M. Gerasimenko, E. V. Vasil’eva, E. V. Kuvshinov, et al., Vopr. Kurortol. Fizioter. Lech. Fiz. Kult., No. 3, 12 (2002).
K. A. Petrov, L. O. Yagodina, G. R. Valeeva, et al., Br. J. Pharmacol. 163 (4), 732 (2011).
Iu. P. Taran and L. N. Shishkina, Radiobiologiia 33 (2), 285 (1993).
K. Felczak, A. K. Drabikowska, J. A. Vilpo, et al., J. Med. Chem. 39 (8), 1720 (1996).
J. C. Leer, K. Hammer-Jespersen, and M. Schwartz, Eur. J. Biochem. 75 (1), 217 (1977).
O. H. Temmink, M. de Bruin, A. W. Turksma, et al., Int. J. Biochem. Cell. Biol. 39 (3), 565 (2007).
S. Watanabe and T. Uchida, Biochem. Biophys. Res. Commun. 216 (1), 265 (1995).
P. W. Woodman, A. M. Sarrif, and C. Heidelberger, Cancer. Res. 40 (3), 507 (1980).
E. Krajewska and D. Shugar, Biochem. Pharmacol. 31 (6), 1097 (1982).
A. A. Lashkov, A. G. Gabdulkhakov, I. I. Prokofev, et al., Acta Crystallogr. F. 68 (11), 1394 (2012).
Prokofev, A. A. Lashkov, A. G. Gabdulkhakov, et al., Acta Crystallogr. F. 70 (1), 60 (2014).
M. Zolotukhina, I. Ovcharova, S. Eremina, et al., Res. Microbiol. 154 (7), 510 (2003).
I. I. Lashkov, A. A. Prokof’ev, A. G. Gabdulkhakov, et al., Crystallogr. Rep. 61 (6), 954 (2016).
W. Kabsch, Integration, Scaling, Space-Group Assignment and Post Refinement. XDS, in International Tables for Crystallography, Ed. by M. G. Rossmann and E. Arnold, (Kluwer, Dordrecht, 2001).
A. Vagin and A. Teplyakov, J. Appl. Crystallogr. 30 (6), 1022 (1997).
G. N. Murshudov, A. A. Vagin, and E. J. Dodson, Acta Crystallogr. D 53 (3), 240 (1997).
P. D. Adams, P. V. Afonine, G. Bunkoczi, et al., Acta Crystallogr. D 66 (2), 213 (2010).
P. V. Afonine, R. W. Grosse-Kunstleve, N. Echols, et al., Acta Crystallogr. D 68 (4), 352 (2012).
P. Emsley and K. Cowtan, Acta Crystallogr. D60 (12), Part 1, 2126 (2004).
P. Emsley, B. Lohkamp, W. G. Scott, et al., Acta Crystallogr. D 66 (4), 486 (2010).
I. W. Davis, A. Leaver-Fay, V. B. Chen, et al., Nucl. Acids Res. 35 (Web Server issue), W375 (2007).
M. A. Larkin, G. Blackshields, N. P. Brown, et al., Bioinformatics 23 (21), 2947 (2007).
X. Robert and P. Gouet, Nucl. Acids Res. 42 (Web Server issue), W230 (2014).
W. L. Delano The PyMOL Molecular Graphics System (2002). http://www.pymol.orgciteulike-article-id:2816763.
A. C. Wallace, R. A. Laskowski, and J. M. Thornton, Protein Sci. 5 (6), 1001 (1996).
Maestro, in Book Maestro (Schrödinger, LLC, New York, 2009).
A. W. Schuttelkopf and D. M. van Aalten, Acta Crystallogr. D 60 (8), 1355 (2004).
R. A. Friesner, J. L. Banks, R. B. Murphy, et al., J. Med. Chem. 47 (7), 1739 (2004).
D. Van Der Spoel, E. Lindahl, B. Hess, et al., J. Comput. Chem. 26 (16), 1701 (2005).
T. Schlesier and G. Diezemann, J. Phys. Chem. B 117 (6), 1862 (2013).
W. G. Touw, C. Baakman, J. Black, et al., Nucl. Acids Res. 43 (Database issue), D364 (2015).
M. G. Rossmann and P. Argos, Annu Rev. Biochem. 50, 497 (1981).
M. J. Pugmire, W. J. Cook, A. Jasanoff, et al., J. Mol. Biol. 281 (2), 285 (1998).
M. J. Pugmire and S. E. Ealick, Biochem. J. 361 (1), 1 (2002).
T. T. Caradoc-Davies, S. M. Cutfield, I. L. Lamont, et al., J. Mol. Biol. 337 (2), 337 (2004).
A. A. Lashkov, N. E. Zhukhlistova, A. H. Gabdoulkhakov, et al., Acta Crystallogr. D 66 (1), 51 (2010).
E. Krissinel and K. Henrick, J. Mol. Biol. 372 (3), 774 (2007).
V. L. Tsuprun, I. V. Tagunova, E. V. Lin’kova, et al., Biokhimiia 56 (5), 930 (1991).
M. V. Dontsova, A. G. Gabdoulkhakov, O. K. Molchan, et al., Acta Crystallogr. F 61 (4), 337 (2005).
A. M. Mikhailov, E. A. Smirnova, V. L. Tsuprun, et al., Biochem. Int. 26 (4), 607 (1992).
W. Bu, E. C. Settembre, M. H. el Kouni, et al., Acta Crystallogr. D 61 (7), 863 (2005).
M. V. Dontsova, A. G. Gabdoulkhakov, O. K. Molchan, et al., Acta Crystallogr. F 61 (4), 337 (2005).
T. H. Tran, S. Christoffersen, P. W. Allan, et al., Biochemistry 50 (30), 6549 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.I. Prokofev, A.A. Lashkov, A.G. Gabdulkhakov, V.V. Balaev, A.S. Mironov, C. Betzel, A.M. Mikhailov, 2018, published in Kristallografiya, 2018, Vol. 63, No. 3, pp. 423–432.
† Deceased.
Rights and permissions
About this article
Cite this article
Prokofev, I.I., Lashkov, A.A., Gabdulkhakov, A.G. et al. Structural and Functional Analysis of Pyrimidine Nucleoside Phosphorylases of the NP-I and NP-II Families in Complexes with 6-Methyluracil. Crystallogr. Rep. 63, 418–427 (2018). https://doi.org/10.1134/S1063774518030239
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063774518030239