Abstract
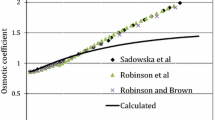
The component solubilities of FeSO4·7H2O–ZnSO4·xH2O–H2O (x = 6, 7) systems at 25 and 40°C were calculated using Pitzer’s ion-interaction model and its Harvie–Weare extension. The calculated results of FeSO4 · 7H2O–ZnSO4 · xH2O–H2O (x = 6, 7) systems are in good agreement with the experimental data both at 25 and 40°C. The model can be extended to a wider range of temperatures for single- or multi-component systems using temperature-dependent binary parameters, providing an alternative approach for the prediction of electrolyte solubilities.


Similar content being viewed by others
REFERENCES
P. M. Kobylin, H. Sippola and P.A. Taskinen, Calphad 35, 499 (2011). https://doi.org/10.1016/j.calphad.2011.08.005
P. M. Kobylin, H. Sippola and P.A. Taskinen, Calphad 38, 185 (2012). https://doi.org/10.1016/j.calphad.2012.06.011
T. Vielma, J. Salminen and U. Lassi, Calphad 60, 126 (2018). https://doi.org/10.1016/j.calphad.2017.12.006
D. P. Tao, Thermochim. Acta 363, 105 (2000). https://doi.org/10.1016/S0040-6031(00)00603-1
K. S. Pitzer, J. Phys. Chem. 4, 249 (1973). https://doi.org/10.1021/j100621a026
K. S. Pitzer, Activity Coefficients in Electrolyte Solutions (CRC Press, Ann Arbor, 1991).
C. E. Harvie and J. H. Weare, Geochim. Cosmochim. Acta 44, 981 (1980). https://doi.org/10.1016/0016-7037(80)90287-2
C. E. Harvie, H. P. Eugste, and J. H. Wear, Geochim. Cosmochim. Acta 46, 1603 (1982). https://doi.org/10.1016/0016-7037(82)90317-9
C. E. Harvie, N. Moller, and J. H. Weare, Geochim. Cosmochim. Acta 48, 723 (1984). https://doi.org/10.1016/0016-7037(84)90098-X
A. S. Malyutin, N. A. Kovalenko, and I. A. Uspenskaya, Russ. J. Inorg. Chem. 65, 781 (2020). https://doi.org/10.1134/S0036023620050149
M. A. Urusova and V. M. Valyashko, Russ. J. Inorg. Chem. 65, 1085 (2020). https://doi.org/10.1134/S003602362006025X
X. P. Zhang, L. R. Zhao, S. Y. Zhou, et al., Russ. J. Inorg. Chem. 65, 2062 (2020). https://doi.org/10.1134/S0036023620140089
Y. Li, P. Song, S. Xia, et al., Calphad 24, 295 (2000). https://doi.org/10.1016/S0364-5916(01)00006-2
K. S. Pitzer and G. Mayorga, J. Solution Chem. 3, 539 (1974). https://doi.org/10.1007/BF00648138
E. J. Reardon and R. D. Beckie, Geochim. Cosmochim. Acta 51, 2355 (1987). https://doi.org/10.1016/0016-7037(87)90290-0
B. Hu, P. S. Song, Y. H. Li, et al., Calphad 31, 541 (2007). https://doi.org/10.1016/j.calphad.2007.03.002
F. Wang and B. Hu, Russ. J. Inorg. Chem. 55, 441 (2010). https://doi.org/10.1134/S0036023610030253
C. Baiarew and V. Karaivanova, Krist. Tech. 10, 1101 (1975). https://doi.org/10.1002/crat.19750101102
V. C. Balarew, Z. Anorg. Allg. Chem. 422, 283 (1976). https://doi.org/10.1002/zaac.19764220313
Funding
This work was financially supported by the National Natural Science Foundation of China (Grant nos. 51804146, 51964029), the National key R & D plan of China (Grant no. 2018YFC1900402), the Applied Basic Research Project of Yunnan Province in China (no. 202001AT070079), and the Analysis Testing Fund of Kunming University of Science and Technology (nos. 2020T20090030, 2020P20181102007).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Yubo Xing, Deng, Z., Yang, F. et al. Solubility Prediction of FeSO4·7H2O–ZnSO4·xH2O–H2O (x = 6, 7) System Using the Pitzer Ion-Interaction Model. Russ. J. Inorg. Chem. 66, 1549–1553 (2021). https://doi.org/10.1134/S0036023621100211
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023621100211