Abstract
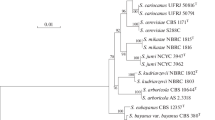
The molecular polymorphism of SUC genes that encode β-fructosidase has been investigated in the yeast genus Saccharomyces. We have determined the nucleotide sequences of subtelomeric SUC3, SUC5, SUC7, SUC8, SUC9, and SUC10 genes of S. cerevisiae and the SUCa gene of S. arboricola. Comparisons of the nucleotide sequences of all known SUC genes revealed the predominance of C → T transitions in the third codon position, which were silent. The amino acid sequences of β-fructosidases studied have identity of 88–100%. SUCa (S. arboricola) and SUCb (S. bayanus) proteins, which had amino acid identity with other SUC proteins of less than 92%, were the most divergent. It was determined that accumulation of the polymeric SUC genes takes place in industrial populations of S. cerevisiae, while the other Saccharomyces species (S. arboricola, S. bayanus, S. cariocanus, S. kudriavzevii, S. mikatae, and S. paradoxus) each harbor only one SUC gene. Subtelomeric repeats of β-fructosidase SUC genes could appear in the genome of S. cerevisiae under the effect of selection in the course of their domestication.
Similar content being viewed by others
References
Goffeau A., Barrell B.G., Barnett J.A. 1981. The utilization of disaccharides and some other sugars by yeasts. Adv. Carbhydr. Chem. Biochem. 39, 347–404.
Badotti F., Dário M.G., Alves S.L., Jr., Cordioli M.L., Miletti L.C., de Araujo P.S., Stambuk B.U. 2008. Switching the mode of sucrose utilization by Saccharomyces cerevisiae. Microb. Cell Fact. 7, 4.
Naumov G.I., Naumoff D.G. 2012. Molecular genetic differentiation of yeast α-glucosidases: maltase and isomaltase. Microbiology (Moscow). 81, 276–280.
Carlson M., Botstein D. 1982. Two differentially regulated mRNAs with different 5′ ends encode secreted and intracellular forms of yeast invertase. Cell. 28, 145–154.
Carlson M., Botstein D. 1983. Organization of the SUC gene family in Saccharomyces. Mol. Cell. Biol. 3, 351–359.
Carlson M., Celenza J.L., Eng F.J. 1985. Evolution of the dispersed SUC gene family of Saccharomyces by rearrangements of chromosome telomeres. Mol. Cell. Biol. 5, 2894–2902.
Sarokin L., Carlson M. 1985. Comparison of two yeast invertase genes: Conservation of the upstream regulatory region. Nucleic Acids Res. 13, 6089–6103.
Sarokin L., Carlson M. 1986. Short repeated elements in the upstream regulatory region of the SUC2 gene of Saccharomyces cerevisiae. Mol. Cell. Biol. 6, 2324–2333.
Mortimer R.K., Contopoulou C.R., King J.S. 1992. Genetic and physical maps of Saccharomyces cerevisiae. Yeast. 8, 817–902.
Naumov G.I., Naumova E.S. 2010. Comparative genetics of yeasts. A novel β-fructosidase gene SUC8 in Saccharomyces cerevisiae. Russ. J. Genet. 46, 323–330.
Naumov G.I., Naumova E.S. 2010. Polygenic control for fermentation of β-fructosides in the yeast Saccharomyces cerevisiae: New genes SUC9 and SUC10. Microbiology (Moscow). 79, 160–166.
Naumov G.I., Naumova E.S., Lantto R.A., Louis E.J., Korhola M. 1992. Genetic homology between Saccharomyces cerevisiae and its sibling species S. paradoxus and S. bayanus: Electrophoretic karyotypes. Yeast. 8, 599–612.
Naumov G.I., Naumova E.S., Sancho E.D., Korhola M.P. 1996. Polymeric SUC genes in natural populations of Saccharomyces cerevisiae. FEMS Microbiol. Lett. 135, 31–35.
Naumova E.S., Korshunova I.V., Jespersen L., Naumov G.I. 2003. Molecular genetic identification of Saccharomyces sensu stricto strains from African sorghum beer. FEMS Yeast Res. 3, 177–184.
Ness F., Aigle M. 1995. RTM1: A member of a new family of telomeric repeated genes in yeast. Genetics. 140, 945–956.
Denayrolles M., de Villechenon E.P., Lonvaud-Funel A., Aigle M. 1997. Incidence of SUC-RTM telomeric repeated genes in brewing and wild wine strains of Saccharomyces. Curr. Genet. 31, 457–461.
Gozalbo D., Hohmann S. 1989. The naturally occurring silent invertase structural gene SUC2 0 contains an amber stop codon that is occasionally read through. Mol. Gen. Genet. 216, 511–516.
Naumov G.I., James S.A., Naumova E.S., Louis E.J., Roberts I.N. 2000. Three new species in the Saccharomyces sensu stricto complex: Saccharomyces cariocanus, Saccharomyces kudriavzevii, and Saccharomyces mikatae. Int. J. Syst. Evol. Microbiol. 50, 1931–1942.
Kurtzman C.P. 2003. Phylogenetic circumscription of Saccharomyces, Kluyveromyces, and other members of the Saccharomycetaceae, and the proposal of the new genera Lachancea, Nakaseomyces, Naumovia, Vanderwaltozyma, and Zygotorulaspora. FEMS Yeast Res. 4, 233–245.
Vaughan-Martini A., Martini A. 2011. Saccharomyces Meyen ex Reess (1870). In: The Yeasts: A Taxonomic Study. Eds. Kurtzman C.P., Fell J.W., Boekhout T. Amsterdam: Elsevier, pp. 733–746.
Goffeau A., Barrell B.G., Bussey H., et al. 1996. Life with 6000 genes. Science. 274, 546–567.
Kellis M., Patterson N., Endrizzi M., Birren B., Lander E.S. 2003. Sequencing and comparison of yeast species to identify genes and regulatory elements. Nature. 423, 241–254.
Liti G., Nguyen Ba A.N., Blythe M., Müller C.A., Bergström A., Cubillos F.A., Dafhnis-Calas F., Khoshraftar S., Malla S., Mehta N., Siow C.C., Warringer J., Moses A.M., Louis E.J., Nieduszynski C.A. 2013. High quality de novo sequencing and assembly of the Saccharomyces arboricolus genome. BMC Genomics. 14, 69.
Korshunova I.V., Naumova E.S., Naumov G.I. 2005. Comparative molecular-genetic analysis of the betafructosidases of yeasts Saccharomyces. Mol. Biol. (Moscow). 39, 366–371.
Wang S.A., Bai F.Y. 2008. Saccharomyces arboricolus sp. nov., a yeast species from tree bark. Int. J. Syst. Evol. Microbiol. 58, 510–514.
Naumov G.I., Lee C.-F., Naumova E.S. 2013. Molecular genetic diversity of the Saccharomyces yeasts in Taiwan: S. arboricola, S. cerevisiae, and S. kudriavzevii. Antonie van Leeuwenhoek. 103, 217–228.
Naumova E.S., Sadykova A. Zh., Martynenko N.N., Naumov G.I. 2013. Molecular genetic characteristics of Saccharomyces cerevisiae distillers’ yeasts. Microbiology (Moscow). 82, 175–185.
Taussing R., Carlson M. 1983. Nucleotide sequence of the yeast SUC2 gene for invertase. Nucleic Acids Res. 11, 1943–1954.
Hohmann S., Gozalbo D. 1988. Structural analysis of the 5’ regions of yeast SUC genes revealed analogous palindromes in SUC, MAL and GAL. Mol. Gen. Genet. 211, 446–454.
Tamura K., Peterson D., Peterson N., Stecher G., Nei M., Kumar S. 2011. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739.
Fischer G., James S.A., Roberts I.N., Oliver S.G., Louis E.S. 2000. Chromosomal evolution in Saccharomyces. Nature. 405, 451–454.
Delorme M.O., Hénaut A., Vigier P. 1988. Mutations in the NAM2 genes of Saccharomyces cerevisiae and S. douglasii are clustered non-randomly as a result of the nucleic acid sequence and not on the protein. Mol. Gen. Genet. 213, 310–314.
Herbert C.J., Dujardin G., Labouesse M., Slonimski P.P. 1988. Divergence of the mitochondrial leucyl tRNA synthetase genes in two closely related yeasts, Saccharomyces cerevisiae and Saccharomyces douglasii: A paradigm of incipient evolution. Mol. Gen. Genet. 213, 297–309.
Herbert C.J., Macadre C., Bécan A-M., Lazowska J., Slonimski P.P. 1992. The MRS1 gene of S. douglasii: Co-evolution of mitochondrial introns and specific splicing proteins encoded by nuclear genes. Gene Expr. 2, 203–214.
Adjiri A., Chanet R., Mezard C., Fabre F. 1994. Sequence comparison of the ARG4 chromosomal regions from the two related yeasts, Saccharomyces cerevisiae and Saccharomyces douglasii. Yeast. 10, 309–317.
Kurtzman C.P., Robnett C.J. 2003. Phylogenetic relationships among yeasts of the “Saccharomyces complex” determined from multigene sequence analyses. FEMS Yeast Res. 3, 417–432.
Oda Yu., Micumo D., Leo F., Urashima T. 2010. Discrimination of Saccharomyces cerevisiae and Saccharomyces paradoxus strains by the SUC2 gene sequences. J. Gen. Appl. Microbiol. 56, 355–358.
Hohmann S., Gozalbo D. 1989. Comparison of the nucleotide sequences of a yeast gene family: 1. Distribution and spectrum of spontaneous base substitutions. Mutation Res. 215, 79–87.
Naumov G.I., Naumova E.S., Masneuf-Pomarède I. 2010. Genetic identification of new biological species Saccharomyces arboricolus Wang et Bai. Antonie van Leeuwenhoek. 98, 1–7.
Zakian V.A. 1996. Structure, function, and replication of Saccharomyces cerevisiae telomeres. Ann. Rev. Genet. 30, 141–172.
Louis E.J., Haber J.E. 1992. The structure and evolution of subtelomeric Y’ repeats in Saccharomyces cerevisiae. Genetics. 131, 559–574.
Louis E.J., Naumova E.S., Lee A., Naumov G., Haber J.E. 1994.The chromosome end in yeast: Its mosaic nature and influence on recombinational dynamics. Genetics. 136, 789–802.
Naumov G.I., Naumova E.S. 2011. Comparative genetics of yeast Saccharomyces cerevisiae: Chromosomal translocations carrying the SUC2 marker. Russ. J. Genet. 47, 147–152.
Codón A.C., Benítez T., Korhola M. 1997. Chromosomal reorganization during meiosis of Saccharomyces cerevisiae baker’s yeasts. Curr. Genet. 32, 247–259.
Gozalbo D., del Castillo Agudo L. 1994. Differential expression of SUC genes: A question of bases. FEMS Microbiol. Rev. 15, 1–7.
Hohmann S., Zimmermann F.K. 1986. Cloning and expression on a multicopy vector of five invertase genes of Saccharomyces. Curr. Genet. 11, 217–225.
Hohmann S. 1987. A region in the yeast genome which favours multiple integration of DNA via homologous recombination. Curr. Genet. 12, 519–526.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © E.S. Naumova, A.Zh. Sadykova, N.N. Martynenko, G.I. Naumov, 2014, published in Molekulyarnaya Biologiya, 2014, Vol. 48, No. 4, pp. 658–668.
Rights and permissions
About this article
Cite this article
Naumova, E.S., Sadykova, A.Z., Martynenko, N.N. et al. Molecular polymorphism of β-fructosidase SUC genes in the Saccharomyces yeasts. Mol Biol 48, 573–582 (2014). https://doi.org/10.1134/S0026893314040086
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893314040086