Abstract
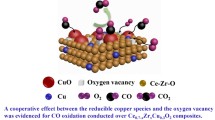
The article reports the synthesis of 5%CuO/Ce1– xZrxO2 catalysts based on CeO2, ZrO2 oxides and Ce1– xZrxO2 solid solutions with х = 0.2, 0.5, and 0.8. It is found that copper oxide is present in the catalysts in a highly dispersed form. In strong interaction with supports, it forms active oxygen, which participates in CO chemisorption and low-temperature oxidation of CO in the presence of hydrogen. In selective CO oxidation, the highest conversion (γmах = 100%) was obtained at temperatures of 120–160°С in the presence of 5%CuO/CeO2. In the modification of CeO2 by zirconium cations, the conversion on 5%Ce0.5Zr0.5O2 decreases to 92% at 160°С because oxygen binding strengthens on copper-containing sites. On the 5%CuO/ZrO2 sample, the maximum conversion is 67% at 180°С. The modification of ZrO2 by cerium cations leads to an increase in the conversion to 87% at 160°С on the 5%CuO/Ce0.2Zr0.8O2 sample as a result of increasing the amount of oxygen vacancies in the support. Taking into account the properties of CO complexes formed on copper-containing oxidation and adsorption sites, and the interaction of these complexes with adsorbed oxygen, their participation in the reaction of low-temperature CO oxidation by oxygen in excess of hydrogen on 5%CuO/CeO2 and 5%CuO/ZrO2 catalysts is considered.








Similar content being viewed by others
REFERENCES
Mishra, A. and Prasad, R., Bull. Chem. React. Eng. Catal., 2011, vol. 6, no. 1, p. 1.
Olah, D., Goeppert, A., and Surya Prokash, G.K., Beyond Oil and Gas: The Methanol Economy, Weinheim: Wiley, 2009.
Avgouropoulos, G. and Ioannides, T., Appl. Catal., A, 2003, vol. 244, p. 155.
Martinez-Arias, A., Gamarra, D., Hungria, A.B., Fernandez-Garcia, M., Munuera, G., Hornes, A., Bera, P., Conesa, J.C., and Camara, A.L., Catalysts, 2013, vol. 3, p. 378.
Firsova, A.A., Il’ichev, A.N., Khomenko, T.I., Gorobinskii, L.V., Maksimov, Yu.V., Suzdalev, I.P., and Korchak, V.N., Kinet. Catal., 2007, vol. 48, no. 2, p. 282.
Balducci, G., Islam, M.S., Kaspar, J., Fornasiero, P., and Graziani, M., Chem. Mater., 2003, vol. 15, p. 3781.
Kaspar, J., Fornasiero, P., and Hickey, N., Catal. Today, 2003, vol. 77, p. 419.
Cao, J.-L., Wang, Y., Zhang, T.-Y., Wu, S.-H., and Yuan, Z.-Y., Appl. Catal., B, 2008, vol. 78, p. 120.
Manzoli, M., Monte, R.D., Boccuzzi, F., Coluccia, S., and Kaspar, J., Appl. Catal., B, 2005, vol. 61, p. 192.
Ratnasamy, P., Strinivas, D., Satyanarayana, C.V.V., Manikandan, P., Kumaran, R.S.S., Sachin, M., and Shetti, V.N., J. Catal., 2004, vol. 221, p. 455.
Martinez-Arias, A., Fernandez-Garcia, M. Galvez, O., Coronado, J.M., Anderson, J.A., Conesa, J.C., Soria, J., and Munuera, G., J. Catal., 2000, vol. 195, p. 207.
Il’ichev, A.N., Firsova, A.A., and Korchak, V.N., Kinet. Catal., 2006, vol. 47, no. 4, p. 585.
Il’ichev, A.N., Bykhovskii, M.Ya., Fattakhova, Z.T., Shashkin, D.P., Matyshak, V.A., and Korchak, V.N., Kinet. Catal., 2018, vol. 59, no. 2, p. 179.
Firsova, A.A, Khomenko, T.I., Il’ichev, A.N., and Korchak, V.N., Kinet. Catal., 2011, vol. 52, no. 3, p. 434.
Sadykov, V.A., Simonov, M.N., Mezentseva, N.V., Pavlova, S.N., Fedorova, Y.E., Bobin, A.S., Bespalko, Y.N., Ishchenko, A.V., Krieger, T.A., Glazneva, T.S., Larina, T.V., Cherepanova, S.V., Kaichev, V.V., Saraev, A.A., Chesalov, Y., Shmakov, A.N., Roger, A.-C., and Adamski, A., Open Chem., 2016, vol. 14, p. 363.
Powder Diffraction Fale. Alphabetical Indoxos. Inorganic phases, JCPDS, Pennsylvania: International Center for Diffraction Data, 1983.
Mirkin, L.I., Spravochnik po rentgenostrukturnomu analizu polikristallov (Polycrystalls X-ray Diffraction Handbook), Moscow: Gos. Izd. Fiz.-Mat. Lit., 1961.
Tret'yakov, I.I., Shub, B.R., and Sklyarov, A.V., Zh. Fiz. Khim., 1970, vol. 44, p. 2112.
Handbuch der Präparativen Anorganischen Chemie. Brauer, G., 1962, Stuttgart: Ferdinand Enke-Verlag.
Rodriguez, J.A. Hanson, J.C. Kim, J.-Y. Liu, G., Iglesias-Juez, A., and Fernandez-Garcia, M., J. Phys. Chem. B, 2003, vol. 107, p. 3535.
Narula, C.K., Haack, L.P., Chun, W., Jen, H.-W., and Graham, G.W., J. Phys. Chem. B, 1999, vol. 103, p. 3634.
Trovarelli, A., Zamar, F., Llorca, J., Leitenburg, C., Dolcetti, G., and Kiss, J.T., J. Catal., 1997, vol. 169, p. 490.
Fornasiero, P., Balducci, G., Monte, R.D., Kaspar, J., Sergo, V., Gubitosa, G., Ferrero, A., and Graziani, M., J. Catal., 1996, vol. 164, p. 173.
Fully, F., Perrichon, V., Vidal, H., Kaspar, J., Blanco, G., Pintado, J.M., Bernal, S., Golon, G., Daturi, M., and Lavalley, J.C., Catal. Today, 2000, vol. 59, p. 373.
Maity, S.K., Rana, M.S., Srinavas, B.N., Bej, S.K., Dhar, G.M., and Prasada Rao, T.S.R., J. Mol. Catal. A: Chem., 2000, vol. 153, p. 121.
Il’ichev, A.N., Matyshak, V.A., and Korchak, V.N., Kinet. Catal., 2015, vol. 56, no. 1, p. 115.
Il’ichev, A.N., Shashkin, D.P., Matyshak, V.A., and Korchak, V.N., Kinet. Catal., 2015, vol. 56, no. 2, p. 197.
Luo, M.F., Ma, J.-M., Lu, J.-Q., Song, Y.-P., and Wang, Y.-J., J. Catal., 2007, vol. 246, p. 52
Gomez-Cortes, A., Marquez, Y., Arenas-Alatorre, J., and Diaz, G., Catal. Today, 2008, vols. 133–135, p. 743.
Polster, C.S., Naier, H., and Baertsch, C.D., J. Catal., 2009, vol. 266, p. 308.
Moretti, E., Storaro, L., Talon, A., Lenarda, M., Riello, P., Frattini, R., Yuso, M.V.M., Jimenez-Lopez, A., Rodriguez-Gastellon, E., Ternero, F., Caballero, A., and Holgado, J.P., Appl. Catal., B, 2011, vol. 102, p. 627.
Arango-Diaz, A., Cecilia, J.A., Moretti, E., Talon, A., Nunez, P., Morrero-Jerez, J., Jimenez-Jimenez, J., Jimenez-Lopez, A., and Rodriguez-Gastellon, E., Int. J. Hydrogen Energy, 2014, vol. 39, p. 4102.
Wang, S.-P., Zheng, X.-C., Wang, X.-Y., Wang, S.-R., Zhang, S.-M., Yu, L.-H., Huang, W.-P., and Wu, S.-H., Catal. Lett., 2005, vol. 105, nos. 3–4, p. 163.
Dow, W.-P. and Huang, T.-J., J. Catal., 1996, vol. 160, p. 171.
Dow, W.-P., Wang, Y.P., and Huang, T.-J., Appl. Catal., A, 2000, vol. 190, p. 25.
Il’ichev, A.N., Shashkin, D.P., Khomenko, T.I., Fattakhova, Z.T., and Korchak, V.N., Kinet. Catal., 2010, vol. 51, no. 5, p. 743.
Dow, W.-P., Wang, Y.P., and Huang, T.-J., J. Catal., 1996, vol. 160, p. 155.
Krylov, O.V., Geterogennyi kataliz (Heterogeneous Catalysis), Moscow: Akademkniga, 2004.
Funding
This work was supported by grant no. 18-03-00627 A from the Russian Foundation for Basic Research and was carried out as part of the state assignment of the FASO Russia (theme V.46. 13, 0082-2014-0007, registration no. AAAA-A18-118020890105-3).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Il’ichev, A.N., Bykhovsky, M.Y., Fattakhova, Z.T. et al. Effect of Zr Content on the Activity of 5%СuO/Ce1– xZrxO2 Catalysts in CO Oxidation by Oxygen in the Excess of Hydrogen. Kinet Catal 60, 661–671 (2019). https://doi.org/10.1134/S002315841905001X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002315841905001X