Abstract
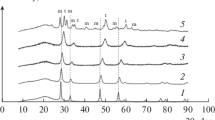
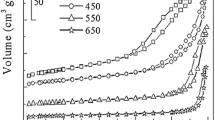
Formaldehyde has been successfully converted into CO2 and H2O at low temperature in the presence of CeO2x–TiO2(100−x) mixed oxides prepared by the sol–gel method. A high selectivity in CO2 was observed with the Ce50Ti50 catalyst. In order to improve the selectivity of the reaction by-products, the Ce50Ti50 was doped with 5 wt% of different transition metals (Ni, Co, Cu, Fe) by impregnation and then activated by reduction under H2. These catalysts have shown excellent catalytic results, which followed the activity order: Co/Ce50Ti50 > Cu/Ce50Ti50 > Ni/Ce50Ti50 > Fe /Ce50Ti50 > Ce50Ti50. Cu/Ce50Ti50 was the most selective solid for the production of CO2. ICP analyses have shown a similarity between the experimental and theoretical values of the metal amount. Physisorption measurements have revealed the mesoporous texture of the solids with large specific surface areas. The XRD analyses exhibited the presence of anatase and cubic phases of Ce50Ti50 mixed oxide. XPS profiles and TEM Images for the best catalyst, Cu/Ce50Ti50, showed that copper particles were identified on the support. From OSC and H2–TPR measurements, the OSC values increase with the Ceria loading on the support increasing the reducibility of the material.












Similar content being viewed by others
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
References
World Health Organization, WHO, air pollution (2016)
E. Uhde, T. Salthammer, Atmos. Environ. 41, 3111 (2007)
IARC classifies Formaldehyde as carcinogenic to humans, Press Release N°153 (International Agency for Research on Cancer, Lyon, 2004)
Directive 001/58/CEE: https://www.ineris.fr/aida
Z.X. Yan, Z.H. Xu, J.G. Yu, M. Jaroniec, Environ. Sci. Technol. 49, 6637 (2015)
H. Huang, Y. Xu, Q. Feng, D.Y.C. Leung, Catal. Sci. Technol. 5, 2649 (2015)
S.C. Jung, Y.K. Park, H.Y. Jung, U.I. Kang, J.W. Nah, S.C. Kim, Res Chem Intermed 42, 185 (2015)
S.C. Kim, C.Y. Park, Res. Chem. Intermed. 28, 441 (2002)
H. Liu, X. Ye, Z. Lian, Y. Wen, W. Shangguan, Res Chem Intermed 32, 9 (2006)
F. Moulis, J. Krýsa, Res Chem Intermed 41, 9233 (2015)
A.J. Jafari, R.R. Kalantari, M. Kermani, M.H. Firooz, Res Chem Intermed 46, 119 (2020)
J.X. Peng, S.D. Wang, Appl. Catal. B 73, 282 (2007)
C.B. Zhang, H. He, Catal. Today 126, 345 (2007)
H.B. Huang, D.Y.C. Leung, J. Catal. 280, 60 (2011)
N. An, W. Zhang, X. Yuan, B. Pan, G. Liu, M. Jia, W. Yan, W. Zhang, (2013) Chem. Eng. J. 215, 1 (2013)
S. Colussi, M. Boaro, L. de Rogatis, A. Pappacena, C. de Leitenburg, J. Llorca, A. Trovarelli, Catal. Today 253, 163 (2015)
L. Yu, R. Peng, L. Chen, M. Fu, J. Wu, D. Ye, Chem. Eng. J. 334, 2480 (2018)
J. Zhang, Y. Jin, C. Li, Y. Shen, L. Han, Z. Hu, X. Di, Z. Liu, App. Catal. B-Environ. 91, 11 (2009)
Y. Zhang, Y. Shen, X. Yang, S. Sheng, T. Wang, M.F. Adebajo, H. Zhu, J. Mol. Catal. A: Chem. 316, 100 (2010)
H. Chen, Z. Rui, X. Wang, H. Ji, Catal. Today 258, 56 (2015)
Z. Qu, S. Shen, D. Chen, Y. Wang, J. Mol. Catal. A: Chem. 356, 171 (2012)
J. Quiroz, J.M. Giraudon, J.F. Lamonier, Catal. Today 176, 277 (2011)
J. Zhang, Y. Li, L. Wang, C. Zhang, H. He, Catal. Sci. Technol. 5, 2305 (2015)
J. Quiroz, J.M. Giraudon, A. Gervasini, C. Dujardin, C. Lancelot, M. Trentesaux, J.F. Lamonier, ACS Catal. 5, 2260 (2015)
L. Bai, F. Wyrwalski, M. Safariamin, R. Bleta, J.F. Lamonier, C. Przybylski, E. Monflier, A. Ponchel, J. Catal. 341, 191 (2016)
J. Xie, M. Meng, Y. Tang, P. Yang, C. Kang, Z. Zhou, S. Huang, Res Chem Intermed 45, 3879 (2019)
Y.S. Xia, H.X. Dai, L. Zhang, J.G. Deng, H. He, C.T. Au, Appl. Catal. B-environ. 100, 229 (2010)
Y. Sekine, Atmos. Environ. 36, 5543 (2002)
T. Hammedi, M. Triki, Z. Ksibi, A. Ghorbel, F. Medina, J. Porous Mat. 22, 939 (2015)
S. Keav, A.E. de Los Monteros, J. Barbier, D. Duprez, App. Catal. B-Environ 150, 402 (2014)
W. Shan, N. Ma, J. Yang, X. Dong, C. Liu, L. Wei, J. Nat. Gas Chem. 19, 86 (2010)
U. Diebold, Surf. Sci. Rep. 48, 53 (2003)
S.L. Swartz, J. Am. Chem. Soc. 124, 12923 (2002)
H.J. Sedjame, C. Fontaine, G. Lafaye, J. Barbier Jr., App. Catal. B- Environ. 144, 233 (2014)
C. Zhang, H. He, K. Tanaka, Appl. Catal. B 65, 37 (2006)
R.J. Gorte, AlChE J. 56, 1126 (2010)
S.S. Lin, D.J. Chang, C.H. Wang, C.C. Chen, Water Res 37, 793 (2003)
C.H. Wang, S.S. Lin, Appl. Catal. A Gen. 268, 227 (2004)
S. Yang, W. Zhu, Z. Jiang, Z. Chen, J. Wang, App. Surf. Sci. 252, 8499 (2006)
M. Al-Amin, S.C. Dey, TUr. Rashid, Md. Ashaduzzaman, SMd. Shamsuddin, IJLRET 2, 14 (2016)
Z. Li, B. Hou, Y. Xu, D. Wu, Y. Sun, W. Hu, F. Deng, J. Solid State Chem. 178, 1395 (2005)
A.A. Ansari, P.R. Solanki, B.D. Malhotra, J. Biotechnol. 142, 179 (2009)
A.S. Dezfuli, M.R. Ganjali, H.R. Naderi, P. Norouzi, RSC Adv 5, 46050 (2015)
M. Thommes, K. Kaneko, A.V. Neimark, J.P. Olivier, F. Rodriguez-Reinoso, J. Rouquerol, K.S.W. Sing, Pure Appl. Chem. 87, 9 (2015)
D. Martin, D. Duprez, J. Phys. Chem. 100, 9429 (1996)
D. Martin, D. Duprez, J. Mol. Catal. Chem. 118, 113 (1997)
G. Lafaye, J. Barbier Jr., D. Duprez, Catal. Today 253, 89 (2015)
G. Ranga Rao, B.G. Mishra, Bull. Catal. Soc. India 2, 122 (2003)
C.B. Zhang, H. He, K.I. Tanaka, Appl. Catal. B-environ 65, 37 (2006)
N.S. de Resende, J.-G. Eon, M. Schmal, J. Catal. 183, 6 (1999)
F.L.S. Carvalho, Y.J.O. Asencios, J.D.A. Bellido, E.M. Assaf, Fuel Process Technol 142, 182 (2016)
S. Akram, Z. Wang, L. Chen, Q. Wang, G. Shen, N. Han, Y. Chen, G. Ge, Catal. Commun. 73, 123 (2016)
P.M. Heynderickx, J.W. Thybaut, H. Poelman, D. Poelman, G.B. Marin, J. Catal. 272, 109 (2010)
S.C. Kim, Y.K. Park, J.W. Nah, Powder Technol. 266, 292 (2014)
Z. Yaakob, A. Bshish, A. Ebshish, S. Masrinda Tasirin, F.H. Alhasan, Materials 6, 2229 (2013)
G. Delahay, A. Guzmán-Vargas, D. Valade, B. Coq, Stud. Surf. Sci. Catal. C. 154, 2501 (2004)
L. Delannoy, G. Thrimurthulu, P.S. Reddy, C. Methivier, J. Nelayah, B.M. Reddy, C. Ricolleaud, C. Louis, Phys. Chem. Chem. Phys 16, 26514 (2014)
J. Li, X. Xu, Z. Hao, Z. Wei, J. Porous Mater 15, 163 (2008)
J. Wang, W. Zhu, S. Yang, W. Wang, Y. Zhou, Appl. Catal. B: Environ. 78, 30 (2008)
E. Bêche, P. Charvin, D. Perarnau, S. Abanades, G. Flamant, Surf. Interface Anal 40, 264 (2008)
P.O. Larsson, A. Andersson, J. Catal. 179, 72 (1998)
Acknowledgments
This work was supported by the European Union (FEDER), the region Nouvelle Aquitaine (France) and the ministry of higher education and scientific research of Tunisia as part of a hosting agreement between laboratories.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest regarding the publication of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bensouilah, R., Olivet, L., Hammedi, T. et al. Effect of the variation of metal and cerium loadings on CeO2x–TiO2(100−x) supports in the complete catalytic oxidation of formaldehyde. Res Chem Intermed 47, 813–834 (2021). https://doi.org/10.1007/s11164-020-04299-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04299-1