Abstract
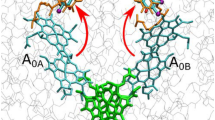
The paper reports on the absorption dynamics of chlorophyll a in a symmetric tetrameric complex of the water-soluble chlorophyll-binding protein BoWSCP. It was measured by a broadband femtosecond laser pump-probe spectroscopy within the range from 400 to 750 nm and with a time resolution of 20 fs-200 ps. When BoWSCP was excited in the region of the Soret band at a wavelength of 430 nm, nonradiative intramolecular conversion S3→S1 was observed with a characteristic time of 83 ± 9 fs. When the complex was excited in the region of the Qy band at 670 nm, relaxation transition between two excitonic states of the chlorophyll dimer was observed in the range of 105 ± 10 fs. Absorption spectra of the excited singlet states S1 and S3 of chlorophyll a were obtained. The delocalization of the excited state between exciton-coupled Chl molecules in BoWSCP tetramer changed in time and depended on the excitation energy. When BoWSCP is excited in the Soret band region, an ultrafast photochemical reaction is observed. This could result from the reduction of tryptophan in the vicinity of chlorophyll.








Similar content being viewed by others
Abbreviations
- BoWSCP:
-
water-soluble chlorophyll-binding protein (WSCP) from Brassica oleracea var. botrytis
- Chl:
-
chlorophyll
References
Charlton, A., and Zachariou, M. (2007) Immobilized metal ion affinity chromatography of histidine-tagged fusion proteins, Methods Mol. Biol., 421, 137-149, https://doi.org/10.1007/978-1-59745-582-4_10.
Cherepanov, D. A., Semenov, A. Y., Mamedov, M. D., Aybush, A. V., Gostev, F. E., Shelaev, I. V., Shuvalov, V. A., and Nadtochenko, V. A. (2022) Current state of the primary charge separation mechanism in photosystem I of cyanobacteria, Biophys. Rev., 14, 805-820, https://doi.org/10.1007/s12551-022-00983-1.
Murata, T., Toda, F., Uchino, K., and Yakushiji, E. (1971) Water-soluble chlorophyll protein of Brassica oleracea var. botrys (cauliflower), Biochim. Biophys. Acta Bioenerg., 245, 208-215, https://doi.org/10.1016/0005-2728(71)90023-5.
Satoh, H., Nakayama, K., and Okada, M. (1998) Molecular cloning and functional expression of a water-soluble chlorophyll protein, a putative carrier of chlorophyll molecules in cauliflower, J. Biol. Chem., 273, 30568-30575, https://doi.org/10.1074/jbc.273.46.30568.
Takahashi, S., Yanai, H., Nakamaru, Y., Uchida, A., Nakayama, K., and Satoh, H. (2012) Molecular cloning, characterization and analysis of the intracellular localization of a water-soluble chl-binding protein from brussels sprouts (Brassica oleracea var. gemmifera), Plant Cell Physiol., 53, 879-891, https://doi.org/10.1093/pcp/pcs031.
Takahashi, S., Yanai, H., Oka-Takayama, Y., Zanma-Sohtome, A., Fujiyama, K., Uchida, A., Nakayama, K., and Satoh, H. (2013) Molecular cloning, characterization and analysis of the intracellular localization of a water-soluble chlorophyll-binding protein (WSCP) from Virginia pepperweed (Lepidium virginicum), a unique WSCP that preferentially binds chlorophyll b in vitro, Planta, 238, 1065-1080, https://doi.org/10.1007/s00425-013-1952-7.
Satoh, H., Uchida, A., Nakayama, K., and Okada, M. (2001) Water-soluble chlorophyll protein in Brassicaceae plants is a stress-induced chlorophyll-binding protein, Plant Cell Physiol., 42, 906-911, https://doi.org/10.1093/pcp/pce117.
Horigome, D., Satoh, H., Itoh, N., Mitsunaga, K., Oonishi, I., Nakagawa, A., and Uchida, A. (2007) Structural mechanism and photoprotective function of water-soluble chlorophyll-binding protein, J. Biol. Chem., 282, 6525-6531, https://doi.org/10.1074/jbc.M609458200.
Maleeva, Y. V., Neverov, K. V., Obukhov, Y. N., and Kritsky, M. S. (2019) Water soluble chlorophyll-binding proteins of plants: structure, properties and functions, Mol. Biol. (Mosk), 53, 998-1011, https://doi.org/10.1134/S0026898419060120.
Schmitt, F. J., Trostmann, I., Theíss, C., Pieper, J., Renger, T., Fuesers, J., Hubrich, E. H., Paulsen, H., Eichler, H. J., and Renger, G. (2008) Excited state dynamics in recombinant water-soluble chlorophyll proteins (WSCP) from cauliflower investigated by transient fluorescence spectroscopy, J. Phys. Chem. B, 112, 13951-13961, https://doi.org/10.1021/jp8024057.
Renger, G., Pieper, J., Theiss, C., Trostmann, I., Paulsen, H., Renger, T., Eichler, H. J., and Schmitt, F. J. (2011) Water soluble chlorophyll binding protein of higher plants: A most suitable model system for basic analyses of pigment–pigment and pigment–protein interactions in chlorophyll protein complexes, J. Plant Physiol., 168, 1462-1472, https://doi.org/10.1016/j.jplph.2010.12.005.
Alster, J., Lokstein, H., Dostál, J., Uchida, A., and Zigmantas, D. (2014) 2D spectroscopy study of water-soluble chlorophyll-binding protein from lepidium virginicum, J. Phys. Chem. B, 118, 3524-3531, https://doi.org/10.1021/jp411174t.
Fresch, E., Meneghin, E., Agostini, A., Paulsen, H., Carbonera, D., and Collini, E. (2020) How the protein environment can tune the energy, the coupling, and the ultrafast dynamics of interacting chlorophylls: the example of the water-soluble chlorophyll protein, J. Phys. Chem. Lett., 11, 1059-1067, https://doi.org/10.1021/acs.jpclett.9b03628.
Theiss, C., Trostmann, I., Andree, S., Schmitt, F. J., Renger, T., Eichler, H. J., Paulsen, H., and Renger, G. (2007) Pigment–pigment and pigment–protein interactions in recombinant water-soluble chlorophyll proteins (WSCP) from cauliflower, J. Phys. Chem. B, 111, 13325-13335, https://doi.org/10.1021/jp0723968.
Renger, T., Trostmann, I., Theiss, C., Madjet, M. E., Richter, M., Paulsen, H., Eichler, H. J., Knorr, A., and Renger, G. (2007) Refinement of a structural model of a pigment–protein complex by accurate optical line shape theory and experiments, J. Phys. Chem. B, 111, 10487-10501, https://doi.org/10.1021/jp0717241.
Friedl, C., Fedorov, D. G., and Renger, T. (2022) Towards a quantitative description of excitonic couplings in photosynthetic pigment-protein complexes: Quantum chemistry driven multiscale approaches, Phys. Chem. Chem. Phys., 24, 5014-5038, https://doi.org/10.1039/d1cp03566e.
Lahav, Y., Noy, D., and Schapiro, I. (2021) Spectral tuning of chlorophylls in proteins – electrostatics vs. ring deformation, Phys. Chem. Chem. Phys., 23, 6544-6551, https://doi.org/10.1039/d0cp06582j.
Agostini, A., Meneghin, E., Gewehr, L., Pedron, D., Palm, D. M., Carbonera, D., Paulsen, H., Jaenicke, E., and Collini, E. (2019) How water-mediated hydrogen bonds affect chlorophyll a/b selectivity in water-soluble chlorophyll protein, Sci. Rep., 9, 18255, https://doi.org/10.1038/s41598-019-54520-4.
Bednarczyk, D., Dym, O., Prabahar, V., Peleg, Y., Pike, D. H., and Noy, D. (2016) Fine tuning of chlorophyll spectra by protein-induced ring deformation, Angew. Chemie Int. Ed., 55, 6901-6905, https://doi.org/10.1002/anie.201512001.
Hughes, J. L., Razeghifard, R., Logue, M., Oakley, A., Wydrzynski, T., and Krausz, E. (2006) Magneto-optic spectroscopy of a protein tetramer binding two exciton-coupled chlorophylls, J. Am. Chem. Soc., 128, 3649-3658, https://doi.org/10.1021/ja056576b.
Obukhov, Y. N., Neverov, K. V., Maleeva, Y. V., and Kritsky, M. S. (2023) Chlorophyll a dimers bound in the water-soluble protein BoWSCP photosensitize the reduction of cytochrome c, Dokl. Biochem. Biophys., 509, 60-64, https://doi.org/10.1134/S1607672923700126.
Takahashi, S., Uchida, A., Nakayama, K., and Satoh, H. (2014) Three-step photoconversion of only three subunits of the water-soluble chlorophyll-binding protein tetramer from Chenopodium album, Protein J., 33, 337-343, https://doi.org/10.1007/s10930-014-9565-y.
Kelly, S. M., Jess, T. J., and Price, N. C. (2005) How to study proteins by circular dichroism, Biochim. Biophys. Acta Proteins Proteomics, 1751, 119-139, https://doi.org/10.1016/j.bbapap.2005.06.005.
Bradford, M. M. (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding, Anal. Biochem., 72, 248-254, https://doi.org/10.1016/0003-2697(76)90527-3.
Cherepanov, D. A., Shelaev, I. V., Gostev, F. E., Mamedov, M. D., Petrova, A. A., Aybush, A. V., Shuvalov, V. A., Semenov, A. Y., and Nadtochenko, V. A. (2017) Mechanism of adiabatic primary electron transfer in photosystem I: Femtosecond spectroscopy upon excitation of reaction center in the far-red edge of the Q Y band, Biochim. Biophys. Acta Bioenerg., 1858, 895-905, https://doi.org/10.1016/j.bbabio.2017.08.008.
Dobryakov, A. L., Pérez Lustres, J. L., Kovalenko, S. A., and Ernsting, N. P. (2008) Femtosecond transient absorption with chirped pump and supercontinuum probe: Perturbative calculation of transient spectra with general lineshape functions, and simplifications, Chem. Phys., 347, 127-138, https://doi.org/10.1016/j.chemphys.2007.11.003.
Golubeva, E. N., Zubanova, E. M., Melnikov, M. Y., Gostev, F. E., Shelaev, I. V., and Nadtochenko, V. A. (2014) Femtosecond spectroscopy and TD-DFT calculations of CuCl42− excited states, Dalt. Trans., 43, 17820-17827, https://doi.org/10.1039/C4DT01409J.
Dobryakov, A. L., Kovalenko, S. A., Weigel, A., Ṕrez-Lustres, J. L., Lange, J., Müller, A., and Ernsting, N. P. (2010) Femtosecond pump/supercontinuum-probe spectroscopy: Optimized setup and signal analysis for single-shot spectral referencing, Rev. Sci. Instrum., 81, 113106, https://doi.org/10.1063/1.3492897.
Kovalenko, S. A., Dobryakov, A. L., Ruthmann, J., and Ernsting, N. P. (1999) Femtosecond spectroscopy of condensed phases with chirped supercontinuum probing, Phys. Rev. A At. Mol. Opt. Phys., 59, 2369-2384, https://doi.org/10.1103/PhysRevA.59.2369.
Šimůnek, J. and Hopmans, J.W. (2002). 1.7 Parameter Optimization and Nonlinear Fitting, in Methods of Soil Analysis (eds Dane, J. H. and Clarke Topp, G.), pp. 139-157, https://doi.org/10.2136/sssabookser5.4.c7.
Clementson, L. A., and Wojtasiewicz, B. (2019) Dataset on the absorption characteristics of extracted phytoplankton pigments, Data Br., 24, 103875, https://doi.org/10.1016/j.dib.2019.103875.
Sirohiwal, A., Berraud-Pache, R., Neese, F., Izsák, R., and Pantazis, D. A. (2020) Accurate computation of the absorption spectrum of chlorophyll a with pair natural orbital coupled cluster methods, J. Phys. Chem. B, 124, 8761-8771, https://doi.org/10.1021/acs.jpcb.0c05761.
Gouterman, M. (1961) Spectra of porphyrins, J. Mol. Spectrosc., 6, 138-163, https://doi.org/10.1016/0022-2852(61)90236-3.
Bricker, W. P., Shenai, P. M., Ghosh, A., Liu, Z., Enriquez, M. G. M., Lambrev, P. H., Tan, H. S., Lo, C. S., Tretiak, S., Fernandez-Alberti, S., and Zhao, Y. (2015) Non-radiative relaxation of photoexcited chlorophylls: theoretical and experimental study, Sci. Rep., 5, 13625, https://doi.org/10.1038/srep13625.
Götze, J. P., Anders, F., Petry, S., Witte, J. F., and Lokstein, H. (2022) Spectral characterization of the main pigments in the plant photosynthetic apparatus by theory and experiment, Chem. Phys., 559, 111517, https://doi.org/10.1016/j.chemphys.2022.111517.
Umetsu, M., Wang, Z. Y., Kobayashi, M., and Nozawa, T. (1999) Interaction of photosynthetic pigments with various organic solvents. Magnetic circular dichroism approach and application to chlorosomes, Biochim. Biophys. Acta Bioenerg., 1410, 19-31, https://doi.org/10.1016/S0005-2728(98)00170-4.
Shipman, L. L., Cotton, T. M., Norris, J. R., and Katz, J. J. (1976) An analysis of the visible absorption spectrum of chlorophyll a monomer, dimer,and oligomers in solution, J. Am. Chem. Soc., 98, 8222-8230, https://doi.org/10.1021/ja00441a056.
Cherepanov, D. A., Milanovsky, G. E., Aybush, A. V., and Nadtochenko, V. A. (2023) Dipole moment of the S0-S1 chlorophyll a transition in solvents with varied refraction index, Russ. J. Phys. Chem. B, 17, 584-593, https://doi.org/10.1134/S1990793123030181.
Berera, R., van Grondelle, R., and Kennis, J. T. M. (2009) Ultrafast transient absorption spectroscopy: principles and application to photosynthetic systems, Photosynth. Res., 101, 105-118, https://doi.org/10.1007/s11120-009-9454-y.
Cherepanov, D. A., Gostev, F. E., Shelaev, I. V., Aibush, A. V., Mamedov, M. D., Shuvalov, V. A., Semenov, A. Y., and Nadtochenko, V. A. (2020) Visible and near infrared absorption spectrum of the excited singlet state of chlorophyll a, High Energy Chem., 54, 145-147, https://doi.org/10.1134/S0018143920020058.
Cherepanov, D. A., Petrova, A. A., Mamedov, M. D., Vishnevskaya, A. I., Gostev, F. E., Shelaev, I. V., Aybush, A. V., and Nadtochenko, V. A. (2022) Comparative absorption dynamics of the singlet excited states of chlorophylls a and d, Biochem., 87, 1179-1186, https://doi.org/10.1134/S000629792210011X.
Davydov, A. S. (1962) The Theory Of Molecular Excitons, McGraw-Hill, New York, NY.
Kasha, M., Rawls, H. R., and El-Bayoumi, M. A. (1965) The exciton model in molecular spectroscopy, Pure Appl. Chem., 11, 371-392, https://doi.org/10.1351/pac196511030371.
Scholes, G. D., and Ghiggino, K. P. (1994) Rate expressions for excitation transfer I. Radiationless transition theory perspective, J. Chem. Phys., 101, 1251-1261, https://doi.org/10.1063/1.467817.
Renger, T., and Marcus, R. A. (2002) On the relation of protein dynamics and exciton relaxation in pigment-protein complexes: an estimation of the spectral density and a theory for the calculation of optical spectra, J. Chem. Phys., 116, 9997-10019, https://doi.org/10.1063/1.1470200.
Pieper, J., Rätsep, M., Trostmann, I., Schmitt, F. J., Theiss, C., Paulsen, H., Eichler, H. J., Freiberg, A., and Renger, G. (2011) Excitonic energy level structure and pigment-protein interactions in the recombinant water-soluble chlorophyll protein. II. Spectral hole-burning experiments, J. Phys. Chem. B, 115, 4053-4065, https://doi.org/10.1021/jp111457t.
Kell, A., Bednarczyk, D., Acharya, K., Chen, J., Noy, D., and Jankowiak, R. (2016) New insight into the water-soluble chlorophyll-binding protein from Lepidium virginicum, Photochem. Photobiol., 92, 428-435, https://doi.org/10.1111/php.12581.
Adolphs, J., Berrer, M., and Renger, T. (2016) Hole-burning spectroscopy on excitonically coupled pigments in proteins: theory meets experiment, J. Am. Chem. Soc., 138, 2993-3001, https://doi.org/10.1021/jacs.5b08246.
Gillie, J. K., Lyle, P. A., Small, G. J., and Golbeck, J. H. (1989) Spectral hole burning of the primary electron donor state of Photosystem I, Photosynth. Res., 22, 233-246, https://doi.org/10.1007/BF00048302.
Pieper, J., Voigt, J., and Small, G. J. (1999) Chlorophyll a Franck–Condon factors and excitation energy transfer, J. Phys. Chem. B, 103, 2321-2322, https://doi.org/10.1021/jp984460e.
Peternian, E. J. G., Pullerits, T., van Grondelle, R., and van Amerongen, H. (1997) Electron-phonon coupling and vibronic fine structure of light-harvesting complex II of green plants: temperature dependent absorption and high-resolution fluorescence spectroscopy, J. Phys. Chem. B, 101, 4448-4457, https://doi.org/10.1021/jp962338e.
Förster, T. (1948) Intermolecular energy migration and fluorescence [in German], Ann. Phys., 437, 55-75, https://doi.org/10.1002/andp.19484370105.
Cherepanov, D. A., Shelaev, I. V., Gostev, F. E., Aybush, A. V., Mamedov, M. D., Shuvalov, V. A., Semenov, A. Y., and Nadtochenko, V. A. (2020) Generation of ion-radical chlorophyll states in the light-harvesting antenna and the reaction center of cyanobacterial photosystem I, Photosynth. Res., 146, 55-73, https://doi.org/10.1007/s11120-020-00731-0.
Cherepanov, D. A., Shelaev, I. V., Gostev, F. E., Aybush, A. V., Mamedov, M. D., Shen, G., Nadtochenko, V. A., Bryant, D. A., Semenov, A. Y., and Golbeck, J. H. (2020) Evidence that chlorophyll f functions solely as an antenna pigment in far-red-light photosystem I from Fischerella thermalis PCC 7521, Biochim. Biophys. Acta Bioenerg., 1861, 148184, https://doi.org/10.1016/j.bbabio.2020.148184.
Shelaev, I. V., Gostev, F. E., Mamedov, M. D., Sarkisov, O. M., Nadtochenko, V. A., Shuvalov, V. A., and Semenov, A. Y. (2010) Femtosecond primary charge separation in Synechocystis sp. PCC 6803 photosystem I, Biochim. Biophys. Acta Bioenerg., 1797, 1410-1420, https://doi.org/10.1016/j.bbabio.2010.02.026.
Van Stokkum, I. H. M., Müller, M. G., Weißenborn, J., Weigand, S., Snellenburg, J. J., and Holzwarth, A. R. (2023) Energy transfer and trapping in photosystem I with and without chlorophyll-f, IScience, 26, 107650, https://doi.org/10.1016/J.ISCI.2023.107650.
Pålsson, L.-O. O., Flemming, C., Gobets, B., van Grondelle, R., Dekker, J. P., and Schlodder, E. (1998) Energy transfer and charge separation in photosystem I: P700 oxidation upon selective excitation of the long-wavelength antenna chlorophylls of Synechococcus elongatus, Biophys. J., 74, 2611-2622, https://doi.org/10.1016/S0006-3495(98)77967-6.
Gobets, B., Kennis, J. T. M., Ihalainen, J. A., Brazzoli, M., Croce, R., Stokkum, I. H. M. van, Bassi, R., Dekker, J. P., van Amerongen, H., Fleming, G. R., and van Grondelle, R. (2001) Excitation energy transfer in dimeric light harvesting complex I: a combined streak-camera/fluorescence upconversion study, J. Phys. Chem. B, 105, 10132-10139, https://doi.org/10.1021/jp011901c.
Riley, K. J., Reinot, T., Jankowiak, R., Fromme, P., and Zazubovich, V. (2007) Red antenna states of photosystem I from cyanobacteria Synechocystis PCC 6803 and Thermosynechococcus elongatus: single-complex spectroscopy and spectral hole-burning study, J. Phys. Chem. B, 111, 286-292, https://doi.org/10.1021/jp062664m.
Gisriel, C., Shen, G., Kurashov, V., Ho, M. Y., Zhang, S., Williams, D., Golbeck, J. H., Fromme, P., and Bryant, D. A. (2020) The structure of Photosystem I acclimated to far-red light illuminates an ecologically important acclimation process in photosynthesis, Sci. Adv., 6, eaay6415, https://doi.org/10.1126/sciadv.aay6415.
Young, R. M., and Wasielewski, M. R. (2020) Mixed electronic states in molecular dimers: Connecting singlet fission, excimer formation, and symmetry-breaking charge transfer, Acc. Chem. Res., 53, 1957-1968, https://doi.org/10.1021/acs.accounts.0c00397.
Casanova, D. (2018) Theoretical modeling of singlet fission, Chem. Rev., 118, 7164-7207, https://doi.org/10.1021/acs.chemrev.7b00601.
Petrova, A. A., Casazza, A. P., Shelaev, I. V., Gostev, F. E., Aybush, A. V., Nadtochenko, V. A., Semenov, A. Y., Santabarbara, S., and Cherepanov, D. A. (2023) Role of pheophytin a in the primary charge separation of photosystem I from Acaryochloris marina: femtosecond optical studies of excitation energy and electron transfer reactions, Biochim. Biophys. Acta Bioenerg., 1864, 148984, https://doi.org/10.1016/j.bbabio.2023.148984.
Akhtar, P., and Lambrev, P. H. (2020) On the spectral properties and excitation dynamics of long-wavelength chlorophylls in higher-plant photosystem I, Biochim. Biophys. Acta Bioenerg., 1861, 148274, https://doi.org/10.1016/J.BBABIO.2020.148274.
Karapetyan, N. V., Bolychevtseva, Y. V., Yurina, N. P., Terekhova, I. V., Shubin, V. V., and Brecht, M. (2014) Long-wavelength chlorophylls in photosystem I of cyanobacteria: origin, localization, and functions, Biochemistry (Moscow), 79, 213-220, https://doi.org/10.1134/S0006297914030067.
Acknowledgments
The authors thank A. L. Dobryakov for assistance in developing the coherent artifact approximation method and setting the femtosecond device. In this paper the equipment (the femtosecond device) provided by the Center for Collective Use of N. N. Semenov Federal Research Center for Chemical Physics, Russian Academy of Sciences “Analysis of Chemical and Biological Systems and Natural Materials: Mass Spectral Microscopy and Femtosecond Laser Microscopy and Spectroscopy” (registration number: 506694). The expression vector pET-24b with the embedded BoWSCP gene was kindly provided by Prof. Dr. Harald Paulsen (Institute of Molecular Physiology, Johannes Gutenberg University Mainz, Germany)
Funding
This research was funded by the Russian Science Foundation (grant no. 21-74-20155).
Author information
Authors and Affiliations
Contributions
D.A.Ch. analysis of spectral measurement results; K.V.N., Yu.N.O., and Yu.V.M. production, recovery, and biochemical characterization of BoWSCP preparations; F.E.G., I.V.Sh., and A.V.A. development of a mathematical apparatus, methods and performing femtosecond measurements; M.S.K. and V.A.N. concept development. All authors made a considerable contribution to the manuscript.
Corresponding authors
Ethics declarations
The authors declare no conflict of interests in financial or any other sphere. This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Cherepanov, D.A., Neverov, K.V., Obukhov, Y.N. et al. Femtosecond Dynamics of Excited States of Chlorophyll Tetramer in Water-Soluble Chlorophyll-Binding Protein BoWSCP. Biochemistry Moscow 88, 1580–1595 (2023). https://doi.org/10.1134/S0006297923100139
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0006297923100139