Abstract
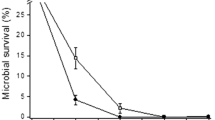
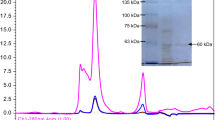
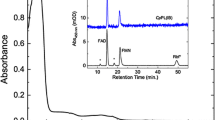
UV-resistant Acinetobacter sp. Ver3 isolated from High-Altitude Andean Lakes (HAAL) in Argentinean Puna, one of the highest UV exposed ecosystems on Earth, showed efficient DNA photorepairing ability, coupled to highly efficient antioxidant enzyme activities in response to UV-B stress. We herein present the cloning, expression, and functional characterization of a cyclobutane pyrimidine dimer (CPD)-class I photolyase (Ver3Phr) from this extremophile to prove its involvement in the previously noted survival capability. Spectroscopy of the overexpressed and purified protein identified flavin adenine dinucleotide (FAD) and 5,10-methenyltetrahydrofolate (MTHF) as chromophore and antenna molecules, respectively. All functional analyses were performed in parallel with the ortholog E. coli photolyase. Whereas the E. coli enzyme showed the FAD chromophore as a mixture of oxidised and reduced states, the Ver3 chromophore always remained partly (including the semiquinone state) or fully reduced under all experimental conditions tested. Functional complementation of Ver3Phr in Phr−-RecA E. coli strains was assessed by traditional UFC counting and measurement of DNA bipyrimidine photoproducts by HPLC coupled with electrospray ionisation-tandem mass spectrometry (ESI-MS/MS) detection. The results identified strong photoreactivation ability in vivo of Ver3Phr while its nonphotoreactivation function, probably related with the stimulation of nucleotide excision repair (NER), was not as manifest as for EcPhr. Whether this is a question of the approach using an exogenous photolyase incorporated in a non-genuine host or a fundamental different behaviour of a novel enzyme from an exotic environment will need further studies.
Similar content being viewed by others
References
M. J. Seufferheld, H. M. Alvarez and M. E. Farias, Role of polyphosphates in microbial adaptation to extreme environments. Appl. Environ. Microbiol., 2008, 74, 5867–5874.
V. H. Albarracín, J. R. Dib, O. F. Ordoñez and M. E. Farias, A Harsh Life to Indigenous Proteobacteria at the Andean Mountains: Microbial Diversity and Resistance Mechanisms Towards Extreme Conditions, in Proteobacteria: Phylogeny, Metabolic Diversity and Ecological Effects, ed. M. L. Sezenna, Nova Science Publishers, 2011, pp. 91–131.
V. Fernandez-Zenoff, F. Sineriz and M. E. Farias, Diverse responses to UV-B radiation and repair mechanisms of bacteria isolated from high-altitude aquatic environments. Appl. Environ. Microbiol., 2006, 72, 7857–7863.
M. R. Flores, O. F. Ordonez, M. J. Maldonado and M. E. Farias, Isolation of UV-B resistant bacteria from two high altitude Andean lakes (4,400 m) with saline and non saline conditions. J. Gen. Appl. Microbiol., 2009, 55, 447–458.
J. Dib, J. Motok, V. F. Zenoff, O. Ordonez and M. E. Farias, Occurrence of resistance to antibiotics, UV-B, and arsenic in bacteria isolated from extreme environments in high-altitude (Above 4400 m) andean wetlands. Curr. Microbiol., 2008, 56, 510–517.
C. Di Capua, A. Bortolotti, M. E. Farias and N. Cortez, UV-resistant Acinetobacter sp isolates from Andean wetlands display high catalase activity. FEMS Microbiol. Lett., 2011, 317, 181–189.
O. F. Ordonez, M. R. Flores, J. R. Dib, A. Paz and M. E. Farias, Extremophile culture collection from Andean lakes: extreme pristine environments that host a wide diversity of microorganisms with tolerance to UV radiation. Microb. Ecol., 2009, 58, 461–473.
M. E. Farias, N. Rascovan, D. M. Toneatti, V. H. Albarracin, M. R. Flores, D. G. Poire, M. M. Collavino, O. M. Aguilar, M. P. Vazquez and L. Polerecky, The discovery of stromatolites developing at 3,570 m above sea level in a High-Altitude volcanic Lake Socompa, Argentinean Andes. PLoS One, 2013, 8.
V. H. Albarracín, G. P. Pathak, T. Douki, J. Cadet, C. D. Borsarelli, W. Gärtner and M. E. Farias, Extremophilic Acinetobacter Strains from High-Altitude Lakes in Argentinean Puna: Remarkable UV-B resistance and efficient DNA damage repair. Orig. Life Evol. Biosph., 2012, 42, 201–221.
S. Weber, Light-driven enzymatic catalysis of DNA repair: a review of recent biophysical studies on photolyase. Biochim. Biophys. Acta, 2005, 1707, 1–23.
S. Akasaka and K. Yamamoto, Construction of Escherichia coli K12 PHR Deletion and insertion mutants by gene replacement. Mutat. Res., 1991, 254, 27–35.
G. P. Pathak, A. Ehrenreich, A. Losi, W. R. Streit, W. Gärtner, Novel blue light-sensitive proteins from a metagenomic approach. Environ. Microbiol., 2009, 11, 2388–2399.
O. Jahn, D. Hesse, M. Reinelt and H. D. Kratzin, Technical innovations for the automated identification of gel-separated proteins by MALDI-TOF mass spectrometry. Anal. Bioanal. Chem., 2006, 386, 92–103.
S. Guindon and O. Gascuel, A simple, fast, and accurate algorithm to estimate large phylogenies by maximum likelihood. Syst. Biol., 2003, 52, 696–704.
D. T. Jones, W. R. Taylor and J. M. Thornton, The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci., 1992, 8, 275–282.
I. Letunic, R. R. Copley, B. Pils, S. Pinkert, J. Schultz and P. Bork, SMART 5: domains in the context of genomes and networks. Nucleic Acids Res., 2006, 34, D257–D260.
N. Ozturk, Y.-T. Kao, C. P. Selby, I. H. Kavakli, C. L. Partch, D. Zhong and A. Sancar, Purification and characterization of a type III photolyase from Caulobacter crescentus. Biochemistry, 2008, 47, 10255–10261.
J. L. Johnson, S. Hammalvarez, G. Payne, G. B. Sancar, K. V. Rajagopalan and A. Sancar, Identification of the 2nd chromophor of Escherichia coli and yeast DNA photolyases as 5,10-Methenyltetrahydrofolate. Proc. Natl. Acad. Sci. U. S. A., 1988, 85, 2046–2050.
S. T. Kim, P. F. Heelis, T. Okamura, Y. Hirata, N. Mataga and A. Sancar, Determination of rates and yields of interchromophore (folate––flavin) energy transfer and intermolecular (flavin––DNA) electron transfer in Escherichia coli photolyase by time-resolved fluorescence and absorption spectroscopy. Biochemistry, 1991, 30, 11262–11270.
T. Douki, M. Court, S. Sauvaigo, F. Odin and J. Cadet, Formation of the main UV-induced thymine dimeric lesions within isolated and cellular DNA as measured by high performance liquid chromatography-tandem mass spectrometry. J. Biol. Chem., 2000, 275, 11678–11685.
S. Mouret, P. Bogdanowicz, M. J. Haure, N. Castex-Rizzi, J. Cadet, A. Favier and T. Douki, Assessment of the photoprotection properties of sunscreens by chromatographic measurement of DNA damage in skin explants. Photochem. Photobiol., 2011, 87, 109–116.
T. Douki and J. Cadet, Individual determination of the yield of the main UV-induced dimeric pyrimidine photoproducts in DNA suggests a high mutagenicity of CC photolesions. Biochemistry, 2001, 40, 2495–2501.
C. P. Selby and A. Sancar, A cryptochrome/photolyase class of enzymes with single-stranded DNA-specific photolyase activity. Proc. Natl. Acad. Sci. U. S. A., 2006, 103, 17696–17700.
Y. Geisselbrecht, S. Fruhwirth, C. Schroeder, A. J. Pierik, G. Klug and L. O. Essen, CryB from Rhodobacter sphaeroides: a unique class of cryptochromes with new cofactors. EMBO Rep., 2012, 13, 223–229.
A. Sancar, Structure and function of DNA photolyase and cryptochrome blue-light photoreceptors. Chem. Rev., 2003, 103, 2203–2237.
J. J. Kim and G. W. Sundin, Construction and analysis of photolyase mutants of Pseudomonas aeruginosa and Pseudomonas syringae: contribution of photoreactivation, nucleotide excision repair, and mutagenic DNA repair to cell survival and mutability following exposure to UV-B radiation. Appl. Environ. Microbiol., 2001, 67, 1405–1411.
H. W. Park, S. T. Kim, A. Sancar and J. Deisenhofer, Crystal structure of E. coli photolyase at 2.3A resolution. Biophys. J., 1995, 68, A324.
Y. T. Kao, C. Saxena, T. F. He, L. J. Guo, L. J. Wang, A. Sancar and D. P. Zhong, Ultrafast dynamics of flavins in five redox states. J. Am. Chem. Soc., 2008, 130, 13132–13139.
L. Valle, F. E. M. Vieyra and C. D. Borsarelli, Hydrogen-bonding modulation of excited-state properties of flavins in a model of aqueous confined environment. Photochem. Photobiol. Sci., 2012, 11, 1051–1061.
D. Chandrasekhar, B. Van Houten, In vivo formation and repair of cyclobutane pyrimidine dimers and 6-4 photoproducts measured at the gene and nucleotide level in Escherichia coli. Mutat. Res., Fundam. Mol. Mech. Mutagen., 2000, 450, 19–40.
G. B. Sancar, Enzymatic photoreactivation: 50 years and counting. Mutat. Res., Fundam. Mol. Mech. Mutagen., 2000, 451, 25–37.
J. van Noort, K. O. van der Werf, B. G. de Grooth and J. Greve, Conformation of individual photolyase dna complexes studied by atomic force microscopy. Biophys. J., 1999, 76, A386–A386.
M. C. DeRosa, A. Sancar and J. K. Barton, Electrically monitoring DNA repair by photolyase. Proc. Natl. Acad. Sci. U. S. A., 2005, 102, 10788–10792.
K. Yamamoto, M. Satake, H. Shinagawa and Y. Fujiwara, Amelioration of the Ultraviolet sensitivity of an Escherichia coli RECA mutant in the dark by photoreactivating enzyme. Mol. Gen. Genet., 1983, 190, 511–515.
A. Sancar, K. A. Franklin and G. B. Sancar, Escherichia coli DNA photolyase stimulates UVR-ABC excision nuclease in vitro. Proc. Natl. Acad. Sci. U. S. A., 1984, 81, 7397–7401.
G. B. Sancar and F. W. Smith, Interactions between yeast photolyase and nucleotide excision repair proteins in Saccharomyces cerevisiae and Escherichia coli. Mol. Cell. Biol., 1989, 9, 4767–4776.
R. P. Sinha and D. P. Hader, UV-induced DNA damage and repair: a review. Photochem. Photobiol. Sci., 2002, 1, 225–236.
Z. Ozer, J. T. Reardon, D. S. Hsu, K. Malhotra and A. Sancar, The other function of DNA photolyase: Stimulation of excision repair of chemical damage to DNA. Biochemistry, 1995, 34, 15886–15889.
G. M. Myles, B. Vanhouten and A. Sancar, Utilization of DNA photolyase, pyrimidine dimer endonucleases, and alkalihydrolysis in the analysis of aberrant ABC excinuclease incisions adjacent to UV-induced DNA photoproducts. Nucleic Acids Res., 1987, 15, 1227–1243.
W. L. Carrier and R. B. Setlow, Excision of Pyrimidine Dimers From Irradiated Deoxyribonucleic Acid in vitro. Biochim. Biophys. Acta, 1966, 129, 318–325.
D. Vlcek, S. Podstavkova and E. Miadokova, Interactions between photolyase and dark repair processes in Chlamydomonas reinhardtii. Mutat. Res.-DNA Repair, 1995, 336, 251–256.
F. Thoma, Light and dark in chromatin repair: repair of UV-induced DNA lesions by photolyase and nucleotide excision repair. EMBO J., 1999, 18, 6585–6598.
W. Yang, Surviving the sun: Repair and bypass of DNA UV lesions. Protein Sci., 2011, 20, 1781–1789.
G. B. Sancar, DNA photolyases: Physical properties, action mechanism, and roles in dark repair. Mutat. Res., 1990, 236, 147–160.
A. Sancar, F. W. Smith and G. B. Sancar, Purification of Escherichia coli DNA photolyase. J. Biol. Chem., 1984, 259, 6028–6032.
S. Bequer Urbano, V. H. Albarracín, O. F. Ordonez, M. E. Farias and H. M. Alvarez, Lipid storage in High-Altitude Andean Lakes extremophiles and its mobilization under stress conditions in Rhodococcus sp. A5, a UV-resistant actinobacterium. Extremophiles, 2013, 17, 217–227.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Electronic supplementary information (ESI) available: Table S2 and Fig. S1, S3 and S4. See DOI: 10.1039/c3pp50399b
Rights and permissions
About this article
Cite this article
Albarracín, V.H., Simon, J., Pathak, G.P. et al. First characterisation of a CPD-class I photolyase from a UV-resistant extremophile isolated from High-Altitude Andean Lakes. Photochem Photobiol Sci 13, 739–751 (2014). https://doi.org/10.1039/c3pp50399b
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1039/c3pp50399b