Abstract
The accurate preoperative diagnosis of periprosthetic joint infection (PJI) of total knee arthroplasty (TKA) can be difficult despite the use of a combination of serum and synovial markers. In such inconclusive cases, incisional open biopsy might be considered. This study investigates the usefulness of biopsies in patients with inconclusive diagnostic findings. We retrospectively identified 63 patients who underwent incisional biopsy for chronic PJI in the operation theatre following TKA revision between 2010 and 2018 after inconclusive preoperative diagnostics for PJI. In all cases, 5 independent biopsies were taken. Results from open biopsy for PJI were analyzed for diagnostic accuracy using the intraoperative results from following revision surgery as gold standard. 27 patients (43%) had a positive culture taken during biopsy. 15 cases (24%) met the diagnostic criteria for a chronic PJI. Most common organisms were Coagulase-negative staphylococci (67%) and Cutibacterium acnes (30%). Compared to the findings during revision surgery, biopsies showed a sensitivity of 47% and a specificity of 77% for PJI. Open incisional biopsy following inconclusive serum- and synovial diagnostics for low grade PJI may be considered for identification of microorganisms. Due to its low sensitivity and moderate specificity found in the present cohort, microbiological analysis should be combined with additional diagnostic markers and histological investigation.
Level of Evidence. Retrospective cohort study (Level III).
Similar content being viewed by others
Introduction
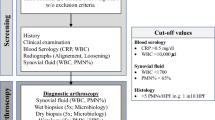
Periprosthetic joint infection (PJI) following total knee arthroplasty (TKA) is a rare but serious complication1,2 that is a leading cause for revision and can be associated with devastating consequences including high mortality of up to 21% after 5 years and repeat revision surgeries3,4,5. An accurate diagnosis or exclusion of PJI is paramount prior to revision surgery, but can be very difficult in low-grade infections in which common clinical signs are often absent1,6. The high prevalence of pain following primary and revision TKA further complicates clinical diagnosis and surgeons must rely on a combination of serum and synovial markers and microbiological findings to diagnose infection7. Currently, the Musculoskeletal Infection Society (MSIS) or International Consensus Meeting (ICM) criteria that incorporate these diagnostic tools in a scoring system are widely used8,9. However, while the sensitivity and specificity of individual tests and a combination of tests is high, there are challenging cases that don’t fulfil the criteria for infection, but present with a high suspicion for PJI8. Despite the high sensitivity and specificity of synovial markers there are cases of a dry tap, clotted specimens or blood contamination of the aspirate, which have been described in up to 36% that greatly impair the quality of synovial analysis10,11. Moreover even if all necessary diagnostic parameters according to the MSIS Criteria have been analyzed an inconclusive score (“possibly infected”) (2–5 points) may occur and further investigation is recommended in these cases8. Furthermore, microbiological cultures from synovial fluid vary greatly in their accuracy in the identification of the causative pathogen in PJI depending on the underlying study. A range of sensitivities from 12 to 94% has been reported by Patridge et al.12, complicating targeted antibiotic treatment.
A potential option for patients in whom PJI is suspected, but preoperative diagnostic workup has been inconclusive, joint biopsy with sampling of periprosthetic and synovial tissue has been proposed. These procedures can be performed arthroscopically, through needle biopsy or as incisional open biopsies with no approach being clearly superior to the others13,14,15,16. Previous studies reported high sensitivity and specificity for routine synovial biopsies in the diagnosis of low-grade chronic PJI13,14,17,18,19. However, there is a lack of studies on open synovial biopsies in cases of inconclusive or intermediate preoperative score results in the diagnostics for chronic PJI in TKA. This study investigated if there is a benefit of open incisional biopsies after inconclusive results of preoperative serum and synovial markers.
Methods
Ethical approval was obtained by the local ethics committee (Ethik Kommission der Ärztekammer Westfalen-Lippe und der Westfälischen Wilhelms-Universität ref. no 2019-728-f-S). The study was conducted according to the principles of the declaration of Helsinki by the World Medical Association.
In a retrospective analysis of revision arthroplasty database (2010–2018), we identified 63 patients with a painful or radiographically loosened TKA (Table 1), that underwent an open incisional biopsy for further evaluation after inconclusive prior diagnostics for PJI. A biopsy was offered prior to revision surgery if patients either had an intermediate ICM score (2–5 points) or had a dry tap or clotted aspirate. Sixteen patients without following revision surgery in our institute were excluded.
All patients underwent analysis of serum inflammatory markers (c-reactive protein (CRP), serum white blood cell count (WBC) and serum interleukin-6 (IL-6)) and a sterile joint aspiration. Synovial fluid leukocyte count as well as percentage of neutrophils was determined, and fluid samples were sent for microbiological analysis (Table 2). Since only TKAs were included in this study a leukocyte count ≥ 1700/microliter or percentage of neutrophils ≥ 65% in the fluid leukocyte differentiation was considered suspicious for PJI20.
Conventional radiographs were checked for signs of implant loosening or osteolysis. Implant loosening was suspected when radiolucent lines in a.-p. and lateral radiographs amounted to ≥ 2 mm applying the criteria by the Knee Society Scoring System21. After patients’ informed consent all open biopsies were performed under strictly sterile conditions in the operation theatre. After standard disinfection using alcohol disinfectant (Codan Schülke & Mayer, Zürich, Switzerland) for a minimum of five minutes and sterile draping an iodine-impregnated incision drape (3 M™ Ioban™ antimicrobial incision drape) was generally applied. No tourniquet was used. Though a suprapatellar mini-arthrotomy of 3-5 cm a minimum of 5 synovial biopsies for microbiological analysis were taken. The open biopsies were generally taken in a strictly standardized manner to gain a most representative result: tissue was obtained individually superior to the patella medial and lateral, inferior to the patella medial and lateral as well as from the patella (Fig. 1).
For every biopsy site, a new forceps was used to prevent contamination. Following the sampling a perioperative intravenous antibiotic prophylaxis with cefuroxime 1.5 g was applied (or clindamycin 600 mg in cases of penicillin allergy) and irrigation with sterile Ringers lactate solution was performed. Each sample was preserved in a sterile tube and immediately sent for culturing. All samples were cultured on chocolate agar, Schaedler agar and Columbia agar for a period of 10–14 days. If a fungal infection was suspected, chromogenic agar and Kimmig agar was used in addition22. All cultures were checked daily for growth. They were declared negative if no growth was detected within 10–14 days of cultivation. Microorganisms were analyzed using standard microbiological procedures for bacterial differentiation and antibiotic resistant patterns.
On basis of the microbiological findings and preoperative score for PJI TKAs were classified as aseptic or PJI according to the ICM Criteria. During revision surgery following the biopsy, a minimum of three to five intraoperative deep tissue samples for additional microbiological analysis were taken and processed as described above. The culture results of incisional biopsies were compared to the culture results of revision surgery. To calculate sensitivity and specificity of incisional biopsy, diagnosis of PJI based on ICM criteria with respect to the findings of revision surgery were used as gold standard.
Statistical analysis
Patients’ data were anonymized prior to analysis. Statistical analysis was performed using SPSS Statistics for Windows Version 25 (IBM Corporation, Armonk, NY, USA). Depending on the distribution of data, medians and 25–75% interquartile ranges (IQR) for non-parametric data and means and ranges for parametric data are presented. Sensitivity and specificity of the open incisional biopsy was calculated using the microbiological findings of patients that received revision surgery of the TKA of any kind. Differences between groups were analyzed via crosstables and the chi-squared-test for categorical variables, while metric data were compared using the Mann–Whitney U-test or students’ t-test depending on the distribution of data. Possible risk factors for positive cultures in the open biopsy or in the revision surgery underwent a univariate analysis using the Cramer-V test for categorical variables.
Results
Microbiological findings from open biopsy showed positive cultures in 27 patients (43%). Based on these findings 19 patients (30%) were diagnosed with PJI.
The most frequent microbiological findings included (CONS) in 18 cases (67%) and Cutibacterium acnes species (ssp.) in 8 of cases (30%). Polymicrobial findings (≥ 2 different species) were present in 7 cases (26%) (Table 3).
With the numbers available, serum CRP ≥ 10 mg/l, , previous history of PJI, elevation of leukocyte count ≥ 1700 µl, or PMN ≥ 65% in the joint aspirate were not correlated with the diagnosis of PJIin the open biopsy (p = 0.16; p = 0.31; p = 0.94; p = 0.84 Cramer V test).
One patient developed a wound healing disorder after open biopsy and resulted in a PJI and subsequent two-stage exchange of the TKA. Further complications could not be observed. The most frequent pathogens at the time of revision were CONS (55%) and Cutibacterium acnes (14%) or Enterococcus species (14%). In contrast to the findings in the open biopsy highly virulent pathogens like methicillin sensitive Staphylococcus aureus (MSSA) and Klebsiella pneumoniae were also found (Table 4).
The median time to revision was 1.8 month (IQR 1.3–2.9 month). In patients that were diagnosed with PJI at the time of open biopsy (n = 19), the diagnosis of PJI was confirmed in 9 cases using the intraoperative samples taken during revision surgery as gold standard.
In contrast, among the 44 patients that were considered to have aseptic failure and underwent single stage revision, unexpected positive cultures were found in 23% (10/44 patients) changing the diagnosis to PJI. On the other hand, in 67% (34/44 patients) the diagnosis of aseptic failure was confirmed.
The calculated sensitivity of open incisional biopsies amounted to 47%, a positive predictive value of 39%, a specificity of 77% and a negative predictive value of 62% for chronic PJI (p = 0.05 Chi Square Test) (Table 5).
Discussion
The correct diagnosis of low-grade PJI in TKA following either dry tap joint aspiration or uncertain results in the preoperative diagnostic workup remains one of the most difficult challenges for the orthopedic surgeon. An undetected low grade infection prior to a partial or complete exchange of the prosthesis may result in repeated revisions, prolonged hospitalization and increased morbidity and mortality23,24. While Microbiological cultures are a useful tool in the detection of a PJI and offer the opportunity to establish a tailored antibiotic treatment, reported incidences of culture negative PJI from 7 to 42% show the urgent need for further improvement of isolation and identification of the respective organism25.
Several studies reported high sensitivity and specificity of routine biopsies in the diagnosis of PJI14,16,17,19. In a prospective evaluation of joint aspiration and biopsy by Meermans et al. using a needle technique in 120 patients with suspected PJI, aspiration showed a sensitivity of 83% and 79% for the biopsy with 100% specificity for both approaches16. In a recent study, Fink et al. presented comparably accurate results using an arthroscopically guided biopsy forceps in routine biopsies (sensitivity 93.8% specificity 97.3%) in cases of implant loosening in a large cohort of 508 patients (277 TKA, 237 THA)19, which emphasized the positive results from the same group assessing unguided synovial biopsies performed for the same indication in 145 TKAs (sensitivity 100%, specificity 98.1%)14. These results lead to the conclusion that a routinely performed biopsy without prior selection of cases with an inconclusive prior diagnostic work-up for PJI in TKA before revision surgery is a reliable diagnostic tool regardless of the chosen device.
In contrast to these findings, a prospective comparison of guided vs. unguided biopsy for histological and microbiological diagnostics in 40 patients with chronic pain after TKA and inconclusive joint aspiration, Scheele et al. reported negative results for PJI in 36 cases with intraoperative positive findings in the following revision in 4 patients. Unfortunately, all four patients with a suspicious biopsy for PJI denied a revision of the TKA making determination of sensibility and specificity impossible13. Nevertheless, this data already alludes to the challenge of organisms isolation and correct diagnostic of low grade PJI following previous inconclusive diagnostics. To our knowledge, the present study is the first to assess specificity and sensitivity of open incisional biopsies following an inconclusive diagnostic workup and comparison with intraoperative tissue samples from the subsequent TKA revision.
The open incisional biopsy technique for microbiological investigation alone shows a poor performance in this particular cohort with a sensitivity of only 47% and a specificity of 77% for a positive result in following revision surgery. As the indication for open biopsy in the present study was inconclusive prior diagnostic work-up and we did not routinely perform synovial biopsies prior to a planned revision of a TKA, our results could be influenced by the fact that patients with conclusive serum and synovial markers that already either ruled out or confirmed a PJI where not included in our cohort.
The findings of this study must be interpreted considering several limiting factors. Due to its retrospective design and small collective of possible low-grade PJI, there might be potential factors that impact these findings that were not accessible for analysis. In addition the underlying gold standard of this study may be impeded by the fact that the amount of perioperative tissue samples differs from three to five samples. Furthermore, while these results were collected over a period of nine years during which the MSIS and ICM criteria for PJI have evolved over time and novel markers and criteria have been added more recently that were not analyzed in the present cohort. Moreoverwhile serum CRP analysis was performed for every case, obligatory analysis of serum IL-6 was later added to our institution’s diagnostic algorithm for PJI and therefore only available in 34 cases (Table 2). Because of its high sensitivity for infection IL-6 could be a useful addition in the diagnostic of low-grade PJI and should further be evaluated26,27. The lack of routinely performed histological analysis of the gained tissue samles presents a further limitation From 2010–2018 it only performed if an osteolysis was visible on the preoperative radiographs for additional investigation for malignancy.
In our institution’s current practice, whenever open biopsy is recommended to confirm or exclude low-grade PJI we conduct collection of microbiology tissue samples as described, as well as histological sampling, a quantitative alpha-defensin analysis and additional cell count and differential by an outside laboratory28. For every revision arthroplasty with complete or partial exchange of the prosthesis sonication is used in our department in addition to synovial aspiration and tissue sampling29.
Conclusion
Open incisional biopsy for microbiological analysis in cases of prior inconclusive serum- and synovial diagnostics for low grade PJI may be considered for isolation of possible microorganisms. If PJI is ruled out by open biopsy, an aseptic approach for revision surgery is reasonable. However, due to its low sensitivity and moderate specificity found in the present cohort, an invasive procedure must be weighed carefully, and microbiological analysis should be combined with additional diagnostic markers and histological investigation.
Data availability
The datasets generated during and/or analysed during the current study are not publicly available due to our institutions regulations of data privacy but are available from the corresponding author on reasonable request.
Abbreviations
- CDC:
-
Centre of disease control
- CONS:
-
Coagulase-negative staphylococci
- CRP:
-
C-reactive protein
- ICM:
-
International consensus meeting
- IDSA:
-
Infectious diseases Society of America
- IL-6:
-
Interleukin-6
- MSIS:
-
Musculoskeletal Infection Society
- OR:
-
Odd ratio
- PJI:
-
Periprosthetic joint infection
- TKA:
-
Total knee arthroplasty
- WBC:
-
White blood cell count
References
Vasso, M. & Schiavone Panni, A. Low-grade periprosthetic knee infection: diagnosis and management. J. Orthopaed. Traumatol. 16, 1–7 (2015).
Blom, A. W. et al. Infection after total knee arthroplasty. J. Bone Jt. Surg. Br. 86, 688–691 (2004).
Koh, C. K. et al. Periprosthetic joint infection is the main cause of failure for modern knee arthroplasty: An analysis of 11,134 knees. Clin. Orthop. Relat. Res. 475, 2194–2201 (2017).
Delanois, R. E. et al. Current epidemiology of revision total knee arthroplasty in the United States. J. Arthroplasty 32, 2663–2668 (2017).
Lum, Z. C. et al. Mortality during total knee periprosthetic joint infection. J. Arthroplasty 33, 3783–3788 (2018).
Balato, G. et al. Diagnostic accuracy of synovial fluid, blood markers, and microbiological testing in chronic knee prosthetic infections. Arch. Orthop. Trauma Surg. 138, 165–171 (2018).
Beswick, A. D., Wylde, V., Gooberman-Hill, R., Blom, A. & Dieppe, P. What proportion of patients report long-term pain after total hip or knee replacement for osteoarthritis? A systematic review of prospective studies in unselected patients. BMJ Open 2, e000435 (2012).
Parvizi, J. et al. The 2018 definition of periprosthetic hip and knee infection: An evidence-based and validated criteria. J. Arthroplasty 33, 1309–1314 (2018).
Goswami, K., Parvizi, J. & Maxwell Courtney, P. Current recommendations for the diagnosis of acute and chronic PJI for hip and knee—cell counts, alpha-defensin, leukocyte esterase, next-generation sequencing. Curr. Rev. Musculoskelet. Med. 11, 428–438 (2018).
Ottink, K. D., Strahm, C., Muller-Kobold, A., Sendi, P. & Wouthuyzen-Bakker, M. Factors to consider when assessing the diagnostic accuracy of synovial leukocyte count in periprosthetic joint infection. J. Bone Jt. Infect. 4, 167–173 (2019).
Zahar, A. et al. How reliable is the cell count analysis in the diagnosis of prosthetic joint infection?. J. Arthroplasty 33, 3257–3262 (2018).
Partridge, D. G., Winnard, C., Townsend, R., Cooper, R. & Stockley, I. Joint aspiration, including culture of reaspirated saline after a “dry tap”, is sensitive and specific for the diagnosis of hip and knee prosthetic joint infection. Bone Jt. J. 100, 749–754 (2018).
Scheele, C. et al. Guided and unguided biopsy in the diagnostic of periprosthetic infections of the knee—evaluation of an evidence-based algorithm. Zeitschrift fur Orthopadie und Unfallchirurgie 157, 684–694 (2019).
Fink, B. et al. The value of synovial biopsy, joint aspiration and C-reactive protein in the diagnosis of late peri-prosthetic infection of total knee replacements. J. Bone Jt. Surg. Br. 90, 874–878 (2008).
Signore, A. et al. Consensus document for the diagnosis of prosthetic joint infections: A joint paper by the EANM, EBJIS, and ESR (with ESCMID endorsement). Eur. J. Nucl. Med. Mol. Imaging 46, 971–988 (2019).
Meermans, G. & Haddad, F. S. Is there a role for tissue biopsy in the diagnosis of periprosthetic infection?. Clin. Orthop. Relat. Res. 468, 1410–1417 (2010).
Fink, B., Gebhard, A., Fuerst, M., Berger, I. & Schäfer, P. High diagnostic value of synovial biopsy in periprosthetic joint infection of the hip. Clin. Orthop. Relat. Res. 471, 956–964 (2013).
Wimmer, M. D. et al. Pre-operative intra-articular deep tissue sampling with novel retrograde forceps improves the diagnostics in periprosthetic joint infection. Int. Orthop. 41, 1355–1359 (2017).
Fink, B., Schuster, P., Braun, R., Tagtalianidou, E. & Schlumberger, M. The diagnostic value of routine preliminary biopsy in diagnosing late prosthetic joint infection after hip and knee arthroplasty. Bone Jt. J. 102, 329–335 (2020).
Trampuz, A. et al. Synovial fluid leukocyte count and differential for the diagnosis of prosthetic knee infection. Am. J. Med. 117, 2 (2004).
Greiner, B., Braun, M. & Becker, W. Value of the plain roentgen image and arthrography in diagnosis of loosening of cemented hip endoprostheses. Z. Orthop. Ihre Grenzgeb. 135, 292–296 (1997).
Theil, C. et al. Fungal prosthetic joint infection in total hip or knee arthroplasty: A retrospective single-centre study of 26 cases. Bone Jt. J. 101, 589–595 (2019).
Boddapati, V. et al. Revision total knee arthroplasty for periprosthetic joint infection is associated with increased postoperative morbidity and mortality relative to noninfectious revisions. J. Arthroplasty 33, 521–526 (2018).
Alp, E., Cevahir, F., Ersoy, S. & Guney, A. Incidence and economic burden of prosthetic joint infections in a university hospital: A report from a middle-income country. J. Infect. Public Health 9, 494–498 (2016).
Reisener, M. & Perka, C. Do culture-negative periprosthetic joint infections have a worse outcome than culture-positive periprosthetic joint infections? A systematic review and meta-analysis. Biomed. Res. Int. 2018, 6278012 (2018).
Majors, I. & Jagadale, V. S. Serum interleukin 6 could be a valuable initial diagnostic tool in prosthetic knee joint infections. Eur. J. Orthopaed. Surg. Traumatol. 29, 1781–1788 (2019).
Ettinger, M. et al. Circulating biomarkers for discrimination between aseptic joint failure, low-grade infection, and high-grade septic failure. Clin. Infect. Dis. 61, 332–341 (2015).
Bonanzinga, T. et al. The role of alpha defensin in prosthetic joint infection (PJI) diagnosis: A literature review. EFORT Open Rev. 4, 10–13 (2019).
Rothenberg, A. C., Wilson, A. E., Hayes, J. P., O’Malley, M. J. & Klatt, B. A. Sonication of arthroplasty implants improves accuracy of periprosthetic joint infection cultures. Clin. Orthop. Relat. Res. 475, 1827–1836 (2017).
Acknowledgements
We acknowledge support from the Open Access Publication Fund of the University of Muenster.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
J.S.: data collection, data analysis, draft of the manuscript, conceptualization, proof reading of the manuscript. B.M.: conceptualization, proof reading of the manuscript, study design. G.G.: conceptualization, proof reading of the manuscript, supervision. T.S.-B.: conceptualization, proof reading of the manuscript, manuscript design. L.L.: data collection, conceptualization. S.K.: data collection, conceptualization. T.A.: data collection, conceptualization, correction of the manuscript. C.T.: data analysis, statistics, draft of the manuscript, conceptualization, proof reading of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Schwarze, J., Moellenbeck, B., Gosheger, G. et al. Poor performance of open incisional biopsy for the microbiological diagnosis of periprosthetic knee joint infection. Sci Rep 11, 10989 (2021). https://doi.org/10.1038/s41598-021-90475-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-90475-1
- Springer Nature Limited