Abstract
In the recent past, non-thermal food processing methods have been promoted due to the limitations associated with conventional thermal processing methods such as poor nutrient quality, rheological properties, and sensory characteristics of food products. The microbial and enzyme inactivation in food products subjected to non-thermal processes occurs without the application of heat that in turn results in products of superior quality. Non-thermal food processing for health-promoting beverages is gaining popularity because of the various advantages like processing at ambient temperature resulting in minimal or no changes in the texture, sensory attributes, composition of nutrients, bioactive compounds (antioxidants, anthocyanin, β-carotene and flavonoids), and organic acids. Due to the continuous rise in the research in this field, it is very important to synthesize relevant literature to supplement existing information to benefit all researchers and industrialists in the food processing sectors. This review aims to critically discuss various non-thermal processing technologies like ultrasound, pulsed light, high hydrostatic pressure, supercritical carbon dioxide, cold plasma, membrane technology, and pulsed magnetic field for processing health-promoting beverages. The working principle, effect of non-thermal processing technologies on the nutritional quality, sensory attributes, and elimination of microbial load of health-promoting beverages are also discussed. Most of the studies are performed on a laboratory scale which exhibits the need for the development of industrial-scale trials. Non-thermal processes are potential alternatives to thermal processing methods due to the retention of superior product quality and lower energy requirements. It is concluded that the adaptability of the combination of non-thermal processes along with aseptic packaging and cold temperature storage will result in superior product quality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Foods are derived from either plant or animal origin and contain various nutrients like carbohydrates, proteins, lipids, vitamins, minerals, and other bioactive components. They are prone to spoilage due to different physical, chemical, and microbial agents. Food preservation is defined as processes by which the shelf life of food products can be increased without affecting the product quality in terms of nutrient retention and sensorial attributes [1]. Beverages provide energy, water, and nutrients like vitamins, minerals, proteins, lipids, and carbohydrates. Beverages can be classified into water, unsweetened tea or coffee, milk, sweetened with non-nutritive sweeteners, energy as well as nutrient-rich, and only energy-yielding sweetened beverages [2,3,4]. The demand for health-promoting beverages in the global market is continuously growing due to consumers' continuous health awareness, and they prefer foods of nutritional value that can reduce the risk of diseases [5, 6]. Conventionally, fruit or vegetable-based beverages are processed using thermal processing methods [7, 8]. However, thermal processing can cause severe physical and chemical changes, affecting the organoleptic properties and nutrient bioavailability [9, 10].
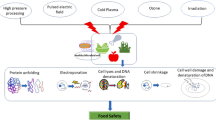
Non-thermal processing techniques like ultrasonication, cold plasma technology, supercritical carbon dioxide (SC-CO2), pulsed light, high hydrostatic pressure (HHP), membrane technology, and pulsed magnetic field can be used for processing health-promoting beverages (Fig. 1). These techniques can be used to prevent the aforementioned drawbacks of conventional thermal processing methods. The non-thermal processing methods can result in a preservation effect similar to thermal processing methods while minimizing the deleterious effects on nutrients, color, texture, and sensorial characteristics of food products [11,12,13,14,15]. Thermal processing requires huge energy requirements and also results in products of poor quality; hence, it can be replaced with environmentally friendly non-thermal processing techniques. During non-thermal treatment, the microbes are inactivated for a shorter duration due to the alteration in the membrane structure of bacteria and the unfolding of DNA structure [16,17,18]. It also affects the non-covalent bonds like hydrogen and ionic bonds to denature the protein, gelatinize the starch, and destroy the pathogenic or food spoilage-causing microorganisms [19,20,21,22]. The thermal treatment affects both covalent and non-covalent bonds, resulting in product quality deterioration [23]. In this review article, the principle and effect of different non-thermal processing technologies on the quality of health-promoting beverages are described.
2 Ultrasonication
In ultrasonication, the treatment of food products is carried out at lower temperatures whereas, in thermosonication, food products are subjected to both ultrasound and moderate temperature [24, 25]. Manosonication is the treatment in which food materials are exposed to ultrasound and pressure (100–300 kPa). In manothermosonication, food products are treated with ultrasound in combination with pressure and temperature [26, 27]. The thermophilic microorganisms can be inactivated by manothermosonication because both the temperature and pressure provide a synergistic effect leading to the collapse of created cavities.
2.1 Working principle
Ultrasound is a sound wave with a frequency higher than 20 kHz which is more than normal human hearing frequency. Ultrasound waves oscillate through the medium resulting in the creation of numerous cycles of expansion and compression [28]. The small cavities are formed due to the presence of air and the cavities expand to a definite size followed by collapse. This results in the generation of huge energy and local hot spots while increasing heat and mass transfer rates [29]. The direct heating of the product takes place during ultrasound treatment due to the release of high energy and intense agitation. Ultrasonication can be classified into low (20–100 kHz), medium (100 kHz–1 MHz), and high (1–100 MHz) range frequencies. Low frequency can produce a large shear force, while medium produces radical species and high frequency produces less shear force. The medium-range frequency of ultrasound waves is more suitable for various processes because that can cause the oxidation of lipids and proteins during food processing. It is carried out by immersion of the health-promoting beverages at a suitable frequency in an ultrasonic bath resulting in the desirable changes in the subjected food material [30]. The diagrammatic representation of ultrasonication is provided in Fig. 2.
Schematic representation of ultrasonication mechanism [99]
2.2 Application of ultrasound in the processing of health-promoting beverages
Ultrasonication can also be used for degassing carbonated beverages by which microbial load reduction can also be attained. A continuous flow high-intensity ultrasound was used for the inactivation of various enzymes during the processing of milk [25]. The synergistic effect of heating (61–75.5 °C) along with ultrasound resulted in the denaturation of enzymes (alkaline phosphatase, gamma-glutamyltranspeptidase, and lactoperoxidase) and proteins (alpha-lactalbumin and beta-lactoglobulin). There was no change in the casein structure but fat globule size was reduced up to 81.5%. The particles were evenly distributed at ~ 70 °C compared to processing at lower temperatures. Ultrasound treatment improves the homogeneity of the solution due to the reduction in particle size and results in superior product quality.
The synergistic effect of carbonation and ultrasonication on nutrient quality and the microbial load reduction in guava juice was investigated by Cheng et al. [31]. Carbonation facilitates the formation of numerous nuclei for cavitation which in turn removes dissolved oxygen. This treatment resulted in the juice with higher ascorbic acid retention but microbial load reduction was low. Sonication of strawberry juice samples at 40–100% amplitude levels at 20 kHz for 2–10 min and pulse durations of 5 s ON and 5 s OFF was reported [32]. The retention of anthocyanin and ascorbic acid content after 10 min of ultrasound treatment was 96.8 and 89%, respectively. The shelf life of sonicated orange juice (0.3–0.81 W/mL for 2–10 min) stored at 10 °C is 27–33 days while thermal pasteurized juice had 19 days of shelf life [33]. The sonicated orange juice retained higher ascorbic acid compared to thermally pasteurized samples. Anthocyanin retention of blackberry juice during ultrasound treatment at 100% amplitude for 10 min is more than 94% with negligible color change [34]. Sonicated passion fruit juice (20 kHz, 263 W, and 89.25 μm) retained the color, pH, and ascorbic acid content higher than thermal preservation while microbial load remained constant for 10 days upon storage at 4 ℃ [35].
Ultrasound treatment of Gulupa juice at 40 kHz for 30 min improved the shelf life to 20 days with minor color changes and a slight decrease in ascorbic acid level [36]. Extraction of noni juice using ultrasound improved the phenolic, and flavonoid content while the nutritional quality and suspension stability of the juice remained the same [37]. The color, pH, soluble solids, flavonoid, and phenolic content of kaji lemon juice are similar to that of thermally processed samples [38]. Ultrasound treatment of strawberry juice for 12 min retained phenolic, flavonoids, and ascorbic acid content similar to that of the unprocessed sample [39]. Conventional pasteurization of pear juice at 95 °C and ultrasound treatment at 65 °C inactivated the microbial load whereas ultrasound treatment resulted in the superior retention of phenolic compounds [40]. The sonicated barberry juice retained higher anthocyanin and phenolic compounds compared to thermally treated samples [41]. Ultrasonication of mulberry juice at 24 kHz for 30 min retained higher anthocyanin (95%) compared to thermally treated samples [42]. Ultrasound treatment of mango juice at 600 W for 40 min resulted in a complete reduction of microbial load and superior retention of nutrients [43]. The quality profile of matured coconut water exposed to ultrasound at 60% amplitude for 10 min was not affected while the microbial load reduction was 4.79 log CFU/mL [44].
The ultrasound treatment of apple juice at 20 °C and 25 kHz for 30–90 min did not alter the pH, color, soluble solids, and titratable acidity while the microbial load was significantly reduced. Ultrasound-treated apple juice also retained a higher amount of ascorbic acid, phenolics, and antioxidant activity [45]. A similar level of inactivation of aerobic mesophiles, yeast, and molds in prebiotic whey beverages using high-intensity ultrasound (600 W) and high-temperature short-time treatment (75 °C for 15 s) was reported by Guimaraes et al. [46]. The phase separation in ultrasound-treated beverages was prevented due to the decrease in particle size, and denaturation of proteins and polysaccharides (inulin and gellan gum). Ultrasound treatment of prebiotic-rich strawberry juice was carried out for 15–30 min at 40 kHz and 180 W [47]. The microbial load was reduced to 1 log CFU/ml and phenolics were increased by 25% compared to untreated samples. The sensorial attributes and ascorbic acid content of strawberry juice were not affected after ultrasound treatment. The various reports on the processing of health-promoting beverages using ultrasound are discussed in Table 1.
3 Cold plasma
Based on the generation of plasma, it is classified into thermal (fully ionized) and cold (partly ionized) plasma. Thermal plasma is composed of thermodynamically balanced ions, electrons, and gas molecules. In cold plasma, local thermodynamic equilibrium is maintained between electrons and gas molecules which in turn leads to the low temperature of the whole system. Cold plasma is more suitable for the processing of beverages because it is carried out under ambient temperature and does not use heat energy to inactivate microbial load, hence the thermolabile compounds are not degraded [12, 48].
3.1 Working principle
Cold plasma is defined as the fourth state of matter and contains a high amount of internal energy that converts the matter from solid-state to liquid then to gas and finally to ionized gaseous state i.e., plasma [49, 50]. The formation of cold plasma occurs when gas is subjected to continuous high electrical energy that in turn facilitates the collision of particles leading to the generation of particles of smaller sizes (Fig. 3). It contains free radicals, electrons, photons, positive ions, negative ions, and excited or non-excited molecules [51, 52]. Atmospheric pressure and energy in the form of electrons are utilized for the formation of cold plasma. The molecules are maintained at ambient temperature; hence this is termed as cold plasma which can be effectively used for processing thermolabile food products [53]. The reactive species generated during cold plasma interact with lipids, proteins, and fatty acids leading to structural variations followed by microbial cell damage. Various factors that affect the efficiency of cold plasma processing are the degree of ionization, nature of gas, temperature source, treatment duration, nature of microbes, and food properties [54].
Schematic diagram of cold plasma equipment. Adapted from [100]
3.2 Application of cold plasma in processing health-promoting beverages
The exposure of orange juice to cold plasma at a voltage of 70 V and pressure of 50 Hz for 1–6 min resulted in the reduction of volatile compounds. The reduction in limonene, y-terpinene, linalool, α-terpineol, terpinene-4-ol, and p-cymene was observed after cold plasma treatment [55]. The effect of cold plasma on the processing of pomegranate juice for 3–7 min at 4 W increased the phenolic content (33%), especially ferulic acid, ellagic acid, and catechin. This treatment also lowered the content of caffeic acid, and punicalagin [56]. Processing of apple juice at a dielectric barrier discharge power of 50 W for 30 s resulted in a 4.3 log reduction of E. coli [57]. Blueberry juice treated with an atmospheric jet plasma power of 11 kV for 6 min resulted in the 7.2 log reduction of Bacillus and enhanced phenolic content [58]. The exposure of tomato juice to the frequency of 50 Hz and voltage of 230 V for 5 min in liquid plasma has not changed the compounds responsible for aroma especially trans-2-hexenal and n-hexanal [59]. The treatment of different juices at an atmospheric jet plasma power of 650 W at the gas flow of 50 L/min for 30–120 s resulted in microbial load reduction as follows: orange juice (1.59 log), tomato juice (1.43 log), apple juice (4 log), and sour cherry nectar (3.34 log). This treatment enhanced the phenolic content (10–15%) and significant changes in color and pH were not observed [60]. Treatment of tender coconut water with high voltage atmospheric cold plasma in the air at 90 kV for 120 s resulted in a 1.3 log reduction of Salmonella enterica. [61]. The formation of reactive gas species like ozone and hydrogen peroxide is responsible for microbial inactivation. Higher microbial cell leakage was further facilitated by the presence of citric acid (400 ppm), resulting in a 5-log reduction of Salmonella enterica. Exposure of white grape juice to dielectric barrier discharge for 4 min at 80 kV resulted in a 7.4 log reduction of Saccharomyces cerevisiae without affecting pH and flavonoid content [62]. Processing of orange juice in dielectric barrier discharge at 90 kV for 2 min resulted in a 4.7 log reduction of Salmonella enterica without significant changes in pH, flavonoid, and phenolic content [63]. A 5-log reduction of Zygosaccharomyces rouxii in apple juice was attained by exposure to dielectric barrier discharge at 90 W for 140 s while oxidative enzymes were not affected [64]. The treatment of tomato juice in gliding discharge plasma at 3.8 kV and 40 W for 5 min resulted in a 5-log reduction of aerobic mesophiles and a 3.7-log reduction of Candida albicans without affecting the lycopene and ascorbic acid content [65]. The changes in the color, ascorbic acid, and vitamin B content of siriguela juice were not observed after glow discharge plasma treatment at 50 Hz for 5–15 min while the polyphenol oxidase activity was reduced to 20% [66].
Treatment of carrot juice with dielectric barrier discharge plasma at 70 kV for 4 min has not affected the content of pigments, sugars, and minerals [67]. The complete reduction of E. coli in sour cherry juice using atmospheric cold plasma at 25, 35, and 50 kV/cm was attained by processing for 1, 5, and 9 min, respectively. The phenolic content in sour cherry juice was not affected while the reduction in anthocyanin and ascorbic acid was 4 and 21%, respectively [68]. Cold atmospheric plasma treatment of apple juice at 30 kV for 7 s resulted in the 4.14 log reduction of Alicyclobacillus acidoterrestris while the complete reduction was attained in 240 s. The pH and color of juice were not affected but a slight reduction in phenolic content was observed [69]. The cold plasma treatment of Jujube juice at 10–30 kV for 2 min resulted in the degradation of mycotoxins like alternariol (1.3–56%) and alternariol monomethyl ether (8.1–62.8%) [70]. Atmospheric cold plasma inactivated acid and non-acid adapted E.coli strains in 120 and 90 s, respectively, without affecting the pH and titratable acidity [71]. Dielectric barrier cold plasma treatment (500 Hz and 20 kV for 10–20 min) imparts a fresh and fruity flavor to the pineapple juice by demethylation of aromatic compounds [72]. Cold plasma treatment of milk at 70 and 80 V for 120 s retained the various physicochemical properties like pH, color, and viscosity while 5 log reduction of Escherichia coli, Listeria monocytogenes, and Staphylococcus aureus was reported [73]. The treatment of cow milk at 70 V for 120 s resulted in a 3.27 log reduction of Cronobacter sakazakii [74]. Treatment of whey protein solution in cold plasma at 70 kV for 30 and 60 min improved the foaming and emulsifying capacity [75]. Exposure of coconut water to cold plasma at 90 kV for 2 min resulted in a 5-log reduction of Salmonella enterica and increased the shelf life up to 45 days [76]. The various reports on the processing of health-promoting beverages using cold plasma are discussed in Table 2.
4 Supercritical carbon dioxide
The application of SC-CO2 improves the shelf life of food products by reducing the microbial load and inactivating lipase that otherwise leads to the formation of free fatty acids, rancidity, and off-flavor development. It reduces the generation of toxic residues and minimizes nutrient loss [77]. The SC-CO2-based processes result in better retention of color, texture, and bioactive compounds of milk, fruit, and vegetable juices [78]. The exposure of food products for a few seconds can eliminate the various spoilage and pathogenic microorganisms but the higher equipment and processing cost limits the application in industries [79, 80].
4.1 Working principle
Carbon dioxide has the properties of liquid and gas when maintained above the critical point generally at high pressure (7.38 MPa) and temperature (31.1 °C). This can inactivate oxidative enzymes and cause cell rupture in the microorganisms [81]. The negligible surface tension and lower viscosity of carbon dioxide facilitate the penetration in the complex microbial cell structure leading to cell rupture [82].
4.2 Application in the processing of health-promoting beverages
The effect of SC-CO2 on the enhancement of the shelf life of orange juice after processing at 36 °C and 13 Mpa with a concentration of 0.385 g CO2/g juice was studied by Fabroni et al. [83]. The product quality with acceptable microbial limit, sensory characteristics, and nutrient content similar to fresh juice was maintained for up to 20 days at refrigerated storage conditions. The treatment of mulberry juice at 15 MPa and 55 °C for 10 min followed by refrigerated storage resulted in the superior retention of bioactive compounds and the shelf life was improved to 21 days [84]. Grape-whey juice treatment at 35 °C and 14–18 MPa for 10 min improved the inhibitory activity of the Angiotensin-converting enzyme due to the interaction of whey protein with the phytochemicals present in the grape juice [85]. About 80% of polyphenol oxidase activity in apple juice was inactivated when treated for 30 min at 25 MPa and 40 °C [86]. The exposure of pomegranate juice to 600 MPa for 3 min enhanced the phenolic content (22%), color, and antioxidant activity at the end of storage for 28 days [87]. The safety and quality of the health-promoting beverages are mainly affected by the oxidative enzymes and microorganisms. The exposure of beverages to SC-CO2 at a pressure range of 10–60 MPa and temperature range of 35–55 °C can improve the shelf life for an additional 20 days with product quality similar to that of fresh juices [77].
The combined treatment of SC-CO2 with ultrasound for 3–4.6 min at 100 bar and 31.5 °C resulted in the complete elimination of microbial load with minimal loss in ascorbic acid, pH, and soluble solids in pineapple juice [88]. The stability of pineapple juice was maintained for up to 4 weeks with a lower reduction in ascorbic acid (8%) and an acceptable microbial limit. Pomegranate juice subjected to SC-CO2 and combined SC-CO2 with ultrasound treatment resulted in the decrease of limonene, 1-hexanol, and camphene content. The treated juice can be differentiated from fresh juice in terms of odor and concentration of volatile compounds (increase in aldehydes and decrease in alcohols, esters, ketones, and terpenes). The flavor of treated juices was not significantly different from fresh juice. The aqueous extract of Pfaffia glomerata roots containing higher fructooligosaccharides (1-kestose, nystose, and fructofuranosylnystose) was added to apple juice [77]. The pH and soluble solid content were not affected by the SC-CO2 treatment while the fructooligosaccharides content remained unchanged indicating SC-CO2 can maintain the prebiotic functionality of beverages. The treatment of strawberry juice at 60 MPa and 45 °C for 30 min resulted in a 3.7 log reduction of aerobic mesophiles [89]. The exposure of apple juice to 6–18 MPa and 20–45 °C resulted in higher inactivation of polyphenol oxidase and complete inactivation of lactic acid bacteria, yeasts, and molds was attained at 12 MPa and 35 °C [90]. The treatment of elderberry juice at 18 MPa and 45 °C for 90 min extended the shelf life up to 2 weeks without any changes in sensory characteristics with complete inactivation of microbes like aerobic mesophiles, yeast, and mold [91]. Orange juice at 74–351 MPa and 33–67 °C for 20–70 min resulted in the prevention of cloud formation by inactivating pectin methyl esterase [92]. Treating blackcurrant juice at 30–60 MPa and 45 °C for 10 min resulted in superior retention of anthocyanin and ascorbic acid compared to thermal pasteurization at 85 °C for 10 min [93]. The pomegranate juice treated with SC-CO2 (12.7 MPa, 45 °C for 15 min) resulted in the reduction of yeast and mold to less than 2 log CFU/ml while aerobic mesophiles was reduced below 2.4 log CFU/ml [94]. The volatile components, pH, and soluble solids of treated juice were not affected during storage for 28 days at 4 °C. These studies indicate that SC-CO2 can be effectively used for developing novel health-promoting beverages with better nutrient quality and better shelf life (Table 3).
5 High hydrostatic pressure
High hydrostatic pressure (HHP) is a non-thermal processing technique wherein food products are subjected to a pressure of 100–1000 MPa at low temperatures to achieve pasteurization, enzyme inactivation, and protein denaturation [95, 96]. The efficiency of this process is dependent on temperature, treatment duration, pressure range, and characteristics of food products [97].
5.1 Working principle
The food product is immersed in a liquid medium and then exposed to high pressure for about 10–20 min in a closed chamber to provide uniform exposure of products to pressure irrespective of their size and shape [88]. Le Chatlier’s principle states that applied force can cause any change in the temperature, pressure, and volume of an equilibrium system leading to the development of a new equilibrium. Adiabatic heating is caused due to the exposure of food products to high pressure that in turn increases the temperature of the liquid medium by 3 °C for a pressure increase of 100 MPa [98].
5.2 Applications of high hydrostatic pressure for processing of health-promoting beverages
The processing of beverages using HHP can prevent the Maillard reaction and caramelization. The texture, color, flavor, and bioactive compounds of food products can be retained after HHP-based processing [99]. The HHP-based processing induces chemical reactions that can prevent microbial growth and also cause cell death by affecting the membranes [100]. The HHP-based treatment of soy smoothie at a pressure of 650 MPa and 20 °C for 3 min resulted in ascorbic acid retention of 55% and no changes in color and bioactive compounds [101]. It has also improved the antioxidant activity due to the increase in the content of β-carotene and lycopene. The sensory characteristics of treated smoothies (450 MPa) and fresh smoothies were similar. The HHP treatment of cucumber juice at 500 MPa for 5 min improved the sensory characteristics compared to high-temperature short-time treated juices [102]. The treatment of watermelon juice at 50 °C and 30 MPa for 30 min reduced polyphenol oxidase activity to 4% [103]. Mulberry juice exposed to the pressure of 500 MPa for 10 min resulted in the inactivation of aerobic mesophiles from 4 log CFU/ml to an undetectable level followed by the complete elimination of yeast and molds [104]. This has also increased flavonoids, phenolics, antioxidant capacity, resveratrol, and volatile compounds like aldehydes, alcohol, and ketones. The HHP-based treatment of strawberry puree at a pressure of 400–600 MPa resulted in higher retention of anthocyanin, phenolics, and ascorbic acid [105]. Aronia berry puree processed at 400 and 600 MPa for 5 min decreased yeast and mold to 1 log CFU/g and aerobic bacteria to < 2 log CFU/g without altering the phenolic content as well as antioxidant activity [106].
The HHP-based process (30–90 MPa, 10–25 °C for 1–7 cycles) reduced the methanol content in sorghum spirits while the sensorial characteristics were improved due to the formation of aroma compounds like methyl acetate [107]. The HHP-based processing of fruit and vegetable smoothies at 630 MPa and 20 °C for 6 min inactivated the peroxidase, polyphenol oxidase, and pectin methylesterase by 31.4, 9.7%, and 83.9%, respectively [108]. The pH and soluble solid content of smoothies were not affected. The exposure of reconstituted cow milk powder containing E. coli, P. fluorescens, and Enterobacter aerogenes to 500–600 MPa and 25 °C for 3–5 min resulted in the inhibition of these gram-negative bacteria [109].
Milk treated at a pressure of 600 MPa for 3 min resulted in the 5 log reduction of E. coli, Salmonella, and L. monocytogenes. This treatment also enhanced the shelf life to 7 days which is comparable to commercial pasteurized milk. The casein content and particle size of fat molecules were similar between HHP-treated and raw milk [110]. Milk processed using HHP for yogurt production resulted in the improvement of product firmness and controlled syneresis [111]. Multifruit-soymilk smoothies processed at 450–650 MPa for 3 min at 20 °C have not modified the color and bioactive compounds (lycopene, β-carotene, and antioxidants). The stability of these compounds was also maintained during refrigerated storage.
The HHP treatment (200–600 MPa for 5–15 min at < 40 °C) of goat milk resulted in the 7-log reduction of Staphylococcus aureus [112]. The viscosity and soluble solid content remained unaltered after HHP treatment and shelf life was improved to 10 days at refrigerated conditions. The treatment of orange juice at 400–600 MPa and 20 °C for 15 min improved the lightness and redness without altering the content of cyanidin-3-glucoside [113]. Treatment of citrus fruits in HHP retained the color and enhanced the shelf life [114]. Raw milk from Bordaleira, as well as Churra Mondegueira da Serra da Estrela sheep breeds treated using HHP of 121 MPa for 30 min, resulted in better curd yield (> 9%) and retained the microbiota beneficial for cheese preparation [115]. Shiikuwasha juice processed at a pressure of 600 MPa for 150 s resulted in > 5 log reduction of E. coli and provided shelf life stability for 28 days without affecting the color, aroma, phenolics, and flavonoids [116]. Carrot orange juice blend treated using HHP of 200–400 MPa for 300 s resulted in the extension of shelf life up to 28 days without altering the phenolic and carotenoid content [117].
Sea buckthorn juice treated at 500 MPa for 5 min retained aroma and flavor similar to fresh juice while the carotenoid and phenol content is comparatively higher than the thermally processed sample [118]. Microorganisms in HHP-treated aronia juice (200–600 MPa for 10 min) after storage for 80 days at 4 °C were below the detection limit and about 8–12% loss in phenolic content was observed [119]. The HHP treatment of apple juice (300 MPa for 10 min) resulted in a 3.7 log reduction of A. acidoterrestris without significantly affecting the pH and color of the juice [120].
The exposure of beetroot juice to HHP treatment at 500 MPa for 10 min resulted in the degradation of betacyanin (12.2%) and betaxanthin (8.9%) while the lactic acid bacteria was reduced up to 4.5 log [121]. Treatment of tomato juice in HHP at a pressure of 500 MPa for 5 min resulted in the superior retention of lycopene content and volatile compounds compared to the thermal pasteurization process. Treatment of kiwi juice in HHP at a pressure of 500 MPa for 10 min resulted in a 2-log reduction of aerobic mesophiles and enhanced the shelf life to 42 days [122]. The exposure of kiwi juice at 200 MPa for 3 cycles in HHP resulted in the extension of shelf life up to 48 days and 7 days in cold storage conditions and room temperature, respectively [123]. The HHP treatment of peanut sprout juice at a pressure of 600 MPa for 5 min retained aroma, color, and pH while the shelf life was extended up to 90 days [124].
The HHP treatment (250 MPa for 4 min) of grape juice resulted in the inhibition of B. cinerea without altering the sensory properties [125]. The HHP treatment of orange juice at a pressure of 600 MPa for 10 min resulted in the reduction of A. acidoterrestris spores to 3.5 log with the extension of shelf life up to 60 days [126]. About 22% of pectin methyl esterase was inactivated while a high reduction of molds (1.85–3.72 log) and yeast (3.19 log) in raspberry juice was attained at HHP treatment of 600 MPa for 10 min [127]. The shelf life of tender coconut water increased up to 120 days after HHP treatment at a pressure of 593 MPa for 3 min without any changes in aroma, pH, color, and taste [128]. Cloudy hawthorn berry juice treated at 300–600 MPa for 30 min at 65 °C inhibited the aerobic mesophiles, lactic acid bacteria, yeast, and mold which in turn improved the shelf life to > 150 days [129]. It has not affected the pH, or titratable acidity but enhanced the sugar content and aroma compounds leading to better flavor and taste. The various reports on the processing of health-promoting beverages using HHP are discussed in Table 4.
6 Pulsed light treatment
The pulsed light treatment is one of the non-thermal processing technologies which is recently been used for processing food products to enhance the shelf life of foods by inactivation of food spoilage and food-born pathogenic microorganisms [129,130,131]. This is due to the inactivation of double-bonded carbon atoms in proteins and nucleic acids leading to the disruption of structure, transcription, and translation processes [132]. The high energy intensity of pulsed light flashes also contributes significantly to microbial inactivation. The effect of microbial inactivation by pulsed light is higher in clear liquids compared to opaque liquids because of the decrease in light intensity due to the absorption and scattering of the light [96].
6.1 Working principle
Pulsed light technology emits high-power pulses either in single or continuous modes of wavelength ranging between 170 and 1000 nm with a gradual increase in energy by using inert gas flash lamps like xenon [133]. The electromagnetic radiation is stored in the capacitor and light flashes can be emitted using inert gas flash lamps to inactivate microorganisms [134, 135]. The process efficiency depends on treatment duration, nature of the food product, packaging material, nature, and number of microorganisms present in the sample [11, 136, 137].
6.2 Application of pulsed light in the processing of health-promoting beverages
The E. coli in apple and orange juices treated with a pulsed light dose of 4 J/cm2 was reduced by 2.9 and 4 logs, respectively [138]. Pulsed light treatment of whey protein isolate at 4–16 J/cm2 improved the formation of carbonyl and free sulfhydryl groups that in turn induced dissociation and partial unfolding of proteins [139]. These changes enhanced the different functional properties like foaming and solution stability. The lactic acid fermented mulberry juice exposed to 2–8 s at a pulse range of 14 J/cm2 resulted in the reduction of microbial load to 1 log CFU/ml [140]. There were no significant changes in the pH, titratable acidity, and soluble solids between pulsed light-treated juice and fresh juice. A slight increase in the phenolic content was observed in the mulberry juice after pulsed light treatment. A decrease in anthocyanin concentration was observed which can be attributed to the adverse photochemical reactions. A slight decrease in the lightness of juice was observed after pulsed light treatment because of the changes in the pigment concentration. The concentration of volatile compounds and odor activity values were enhanced during pulsed light treatment.
The reduction of E. coli in tender coconut water, orange, and pineapple juice using pulsed light treatment (95.2 J/cm2) for 15 s was 4, 4.5, and 5.33 log, respectively [141]. The pulsed light treatment of apricot juice at two different intensities (7 and 14 kV/cm) for 500 µs has not significantly affected the pH, °Brix, and color compared to unprocessed samples. The pulsed light treatment resulted in increased content of phenolic compounds, flavonoids, and antioxidant activity [142]. Apple juice was exposed to pulsed light of different cycles like 4, 6, and 8 with a total number of pulses in the range of 200–400 pulses [143]. The changes in bioactive compounds, ascorbic acid, and polyphenol content were not observed in pulsed light-treated juice even after 72 h of refrigeration. Pulsed light treatment of pineapple juice at 13 kV/cm resulted in higher microbial load reduction compared to treatment at 9 and 11 kV/cm [144]. The total phenolics, flavonoids, ascorbic acid, and β-carotene content was not affected. The pulsed light treatment (31.2 kV/cm, 20 pulse widths at 100 Hz frequency) retained higher antioxidant activity and sensorial properties of ready-to-serve beverage containing tender coconut water and nannari extract in comparison to conventional pasteurization (96 °C for 360 s) [145]. Raspberry juice exposed to pulsed light treatment at 200 Hz retained higher phenolic content (426.4–567.2 mg GAE/100 g) and around 19% of pectin methyl esterase enzyme was inactivated [127]. Complete inactivation of peroxidase and polyphenol oxidase in amla juice was attained when exposed to pulsed light at 3.32 kV/cm for 3 min [146]. The exposure of mixed fruit beverage containing amla, pineapple, and coconut water to pulsed light at 3.3 kV/cm for 3 min resulted in 5 log reduction of aerobic mesophiles, yeast, and mold counts while the oxidative enzymes were completely inactivated [147]. The pulsed light treatment of bael fruit juice at 3.3 kV/cm for 4.5 min inactivated the oxidative enzymes like polyphenol oxidase, and peroxidase (73.5–90%) better than the thermal process [148]. The 5 log reduction of Listeria monocytogenes and E. coli in sweet lime juice was attained after pulsed light treatment at 3.3 kV/cm for 4.5 min without any changes in pH, color, or taste of the juice [149]. The continuous flow pulsed light treatment of coconut water, pineapple, and orange juice for 43, 44, and 45 s resulted in 5-log reduction of aerobic mesophiles, yeast, and mold without altering their physicochemical properties [150]. Processing tender coconut water with pulsed light at 2.5 kV for 2.5 min resulted in a 5 log reduction of B. cereus and L. monocytogenes [151]. Pomegranate juice treatment with pulsed light at 2.7 kV/cm for 90 s resulted in a 5-log reduction of aerobic mesophiles, yeast & molds and retained a higher amount of phenolics (97%), antioxidants (94%), and ascorbic acid (83%) [152]. Pineapple juice treated with pulsed light at 2.4 kV/cm for 3.5 s resulted in the reduction of aerobic mesophiles, yeast, and molds without affecting the sensory attributes [153]. The treatment of mulberry juice using combined pulsed light (1.2 kV/cm for 3.5 s) and ultrasonication treatment (15 min at a frequency of 28 kHz) retained phenolic and antioxidant activities at higher levels compared with the thermal process [154]. Exposure of verjuice to pulsed light at 6.12 J/cm2 for 3.5 s resulted in a 5.1 log reduction of S. cerevisiae and the shelf life of the juice extended up to 6 weeks without any changes in fresh-like characteristics (color, aroma, taste) and sensory attributes [155]. The pulsed light treatment of grape juice at 66 J/cm2 for 3.5 s resulted in a 2.6 log reduction of E. coli [156]. The shelf life of pulsed light-treated (2.9 kJ/cm2 for 3.5 s) pomegranate juice extended up to 41 days with higher retention of phenolics and antioxidant activity compared to thermal-treated samples [157]. The various reports on the processing of health-promoting beverages using pulsed light are discussed in Table 5.
7 Membrane technology
The application of membrane technologies is suitable for the processing of health-promoting beverages due to several advantages like lesser energy consumption, mild processing conditions, and superior product quality compared to conventional thermal processes.
7.1 Working principle
The pressure-driven membrane operations such as microfiltration and ultrafiltration are used for the clarification and concentration of beverages [158]. These processes involve a semipermeable barrier (membrane) for selective separation of compounds from feed solution and pressure gradient acts as the driving force for mass transfer through the membrane. The feed solution is separated into permeate (molecules permeated through the membrane) and retentate (molecules rejected by the membrane). The degree of rejection depends on the pore size, charge, and surface properties of the membrane [159,160,161]. The performance of the pressure-driven membrane process is assessed by permeate flux and degree of separation [162].
7.2 Application of membrane technology in the processing of health-promoting beverages
The pore size of microfiltration and ultrafiltration membranes is 0.1–10 µm and 0.001 to < 0.1 µm, respectively [163]. These membranes can reject the molecules larger than the pore size without causing physicochemical changes in the feed solution. The pathogenic microorganisms can be removed from the beverages by proper selection of membranes of suitable pore size. Ultrafiltration can be used to remove macromolecules like proteins and polysaccharides that in turn cause turbidity in health-promoting beverages. This can also be used for removing all microorganisms. Thereby ultrafiltration can be simultaneously used for clarification and cold sterilization of beverages. The processing of apple juice at the pilot scale level by ultrafiltration membranes composed of ceramic and organic polymer resulted in a 9 and 5-log reduction of bacteria, respectively [164]. The application of an ultrafiltration membrane of 0.03 µm pore size for the processing of white birch sap reduced the microbes from 1.6 × 104 to 2.0 × 102 CFU/mL. The combination of ultraviolet with ultrafiltration resulted in the complete elimination of microorganisms during 40 days of storage at 4 or 25 °C [165]. Banana juice processed using ultrafiltration enhanced the shelf life to 30 days with superior product quality [166]. The quality of bottle gourd juice processed by ultrafiltration was not adversely affected even after 8 weeks of storage [167]. The processing of jamun juice using polysulfone hollow fiber of 50 kDa pore size eliminated the microorganisms [168]. A flat membrane module composed of polyethersulfone (pore size < 0.01 µm) also reduced the microbial load to an undetectable level in coconut water [169].
Microfiltration can be used as a pretreatment prior to ultrafiltration in order to avoid the fouling problem of the membrane by removing the microorganisms and particles of larger size. Coconut water containing 140 CFU/mL of aerobic mesophiles was processed sequentially using 0.8 µm and 0.45 µm of cellulose nitrate membrane. The growth of microbes was not observed even after 180 days of storage and a slight increase in total soluble solids was observed due to the hydrolysis of carbohydrates [170]. The membrane of 0.3 µm pore size resulted in the reduction of microorganisms in pineapple juice to the desired level [171]. The processing of watermelon juice using an ultrafiltration membrane of polyethersulfone (50 kDa molecular weight cut off) resulted in the reduction of soluble solids (2.8%) and ascorbic acid (11.2%) [172]. Dragon fruit juice treated using ultrafiltration membrane of polyethersulfone of different pore sizes (5, 10, and 20 kDa molecular weight cut off) resulted in the retention of betacyanin (30.6%) at a higher level compared to phenolic compounds (11.3%) level [173]. Processing of broccoli juice in an ultrafiltration membrane of polyethersulfone with 5, 10, and 50 kDa molecular weight cut off decreased the phenolic content by 2.7, 9.6, and 5.5%, respectively [174]. Orange juice processed in ultrafiltration membranes composed of polyethersulfone retained better clarity compared to polysulfone membranes [175]. Lemon juice treated with cellulose acetate membrane (100 kDa) retained higher antioxidant activity with a significant amount of eriocitrin and hesperidin [176]. The treatment of cashew apple juice in an ultrafiltration membrane improved the shelf life by 12 weeks [177]. Cloudy apple juice processed in an ultrafiltration membrane of polyethersulfone (10 kDa molecular weight cut off) enhanced the shelf life to 4 weeks without any changes in the quality [178]. The processing of sugarcane juice by ultrafiltration (polysulfone membrane of 30 kDa) extended the shelf life up to 4 weeks by inactivating the bacteria (Streptomyces, Staphylococcus, Lactobacillus) [179]. Apple beetroot and apple juice processed in the ceramic membrane (15 kDa) retained 68 and 71% of soluble solids, respectively [180]. Mulberry juice treated in an ultrafiltration membrane (100 kDa) retained better color and phenolic compounds [181]. Ultrafiltration of watermelon juice in 1 kDa membrane improved the enrichment efficiency which in turn increased the concentration and purity of lycopene [182]. The various reports on the processing of health-promoting beverages using membrane technology are discussed in Table 6.
8 Pulsed magnetic field
The pulsed magnetic field is one of the emerging non-thermal techniques in recent years for enzyme and microbial inactivation in health-promoting beverages. The major parameters that affect the pulsed magnetic field process are magnetic field intensity and number of pulses.
8.1 Working principle
The electric current flow in a coiled wire or solenoid leads to the generation of magnetic current across the coil. The pulsed magnetic field is generated by the generator in the form of short energy pulses which are stored in the capacitor bank. Superconducting coils can be used to generate magnetic field intensities in the range of 5–50 T. Microorganisms and enzymes contain charged particles that undergo drifting under the influence of Lorentz force in the magnetic field. This results in the formation of induced currents followed by the inactivation of microbes and enzymes. Magnetoporation induced by a pulsed magnetic field leads to microbial death due to the loss of cytosol and nucleic acid together with the increase in intracellular Ca2+ content. The pulsed magnetic field affects the enzyme activity by the unfolding of tertiary structure. The microorganisms respond to the domain of a pulsed magnetic field by stimulation which is known as the “window effect” [183].
8.2 Application of pulsed magnetic field in the processing of health-promoting beverages
The pulsed magnetic field processing of orange juice at 7 T and 20 pulses resulted in the 0.61 log reduction of aerobic mesophiles [184]. The pulsed magnetic field range used in this study was not effective in microbial inactivation indicating the possible window effect. Pulsed magnetic field treatment of 8 T with a pulse number of 60 in cucumber juice resulted in a 1.36 log reduction of E. coli [185]. The increase in intensity to three times reduced the bacteria to an undetectable level even after 4 days of storage at 4 °C. The inactivation of bacteria in vegetable juices (cucumber, carrot, spinach, and bitter gourd) was further facilitated by the combination of a pulsed magnetic field (3 times 8 T with 60 pulses) with Litseacubeba essential oil at a concentration of 1.5 mg/ml [186]. The addition of essential oil slightly masked the natural flavor of the juice but the sensorial attributes were not significantly affected. The cell membrane rupture and the inhibition of intracellular enzymes are the major mechanisms responsible for cell death. The higher bacterial load reduction (99.9%) in bovine colostrum was attained during pulsed magnetic field treatment of 2.53 T with a pulse number of 20 with minimal loss of IgG and sensorial characteristics [187]. Maximum inactivation of E. coli, mold, and yeast in milk was attained at 16.02 T with a pulse number of 6–8 [188]. The pulsed magnetic field processing of apple juice at 6 T with 15 pulses inactivated various oxidative enzymes such as polyphenol oxidase, peroxidase, and pectin methyl esterase [189]. The various reports on the processing of health-promoting beverages using a pulsed magnetic field are discussed in Table 7.
9 Current challenges and future prospective
At present, most of the studies are performed on a laboratory scale and very few pilot-scale studies are reported. Hence more scale-up studies have to be performed and economic feasibility studies have to be undertaken to ensure sustainable utilization and implementation. Additionally, more studies focused on the combined or sequential approach of various non-thermal processing methods have to be applied to food products for microbial decontamination, and inactivation of oxidative enzymes. Ultrasonication can be applied for food preservation in combination with other methods to further improve enzyme inactivation and microbial load reduction. The synergistic effect of the ratio of beverage and CO2 in the SC-CO2 process can be studied to improve the process reliability to meet the global demand. In HHP treatment, heat generation leads to process instability and non-uniformity. The other challenges include a lack of data on the synergistic effect between pressure and packaging materials. Dielectric barrier discharge is a type of cold plasma that can be efficiently used for in-pack sterilization of beverages. Further optimization studies of processing variables in pulsed light treatment are needed to obtain products of superior quality. Studies should also focus on the structural integrity, bioavailability, and flow behavior of processed beverages [190]. Ultrafiltration membranes of tiny pore size can result in fouling and poor permeability of nutrients with smaller molecular weight. The proper selection of ultrafiltration membrane of suitable pore size should be selected for the removal of viruses and improved process efficiency. Microfiltration can be used as a pretreatment to remove larger microorganisms to minimize the fouling problem. The application of coagulation agents is also effective in controlling the fouling of membranes [191]. Studies on pulsed magnetic field for microbial load reduction and enzyme inactivation in beverages are very limited. Thus, more studies are required to understand the effect of pulsed magnetic field on the nutritional quality and sensorial attributes of food products [183].
In the future, novel processing techniques can be developed by combining different non-thermal processes to improve product quality and process efficiency. There is also scope for process equipment design and process economics for non-thermal processes to make them more accessible and cost-effective. There is an urgent need for the establishment of streamlined regulatory bodies concerning the utilization of these technologies to make the approval process faster and less expensive. Customized equipment can be developed to process health-promoting beverages to meet consumer demands. Algorithms, artificial intelligence, and data analytics can be incorporated with these advanced techniques for the real-time monitoring and control of the process. For the widespread adoption of non-thermal processing in the beverage industry, it is pivotal to overcome the current challenges as well as to work with future opportunities in this field.
10 Conclusion
Non-thermal food processing techniques are a promising alternative for food processors and provide a glimpse of future innovations in food technology. Conventional thermal treatments are commonly used for the pasteurization of liquid food products but they can affect the product quality in terms of nutrient retention and changes in sensorial attributes. The application of novel green technologies like non-thermal processing methods is environmentally friendly, rapid, and effective in microbial inactivation without affecting product quality. The action mechanisms of various non-thermal food processing techniques driven by different energy sources have been explored in the present review paper. The non-thermal processing methods come under a sustainable approach for microbial inactivation that in turn can improve the shelf life of food products. The non-thermal processes can be combined with preservatives to attain complete sterilization of food products. The low-temperature storage and distribution of non-thermal food products are essential for maintaining their sensorial properties. The HHP treatment can be applied to packaged food products and it has minimal effect on nutrients and sensorial attributes. The pulsed electric field is suitable for processing only liquid food products and reduces the degradation of heat-labile compounds. Ultrasound and membrane technology are economically feasible and also meet the requirements for scale-up. The HHP and SC-CO2 are suitable for both microbial and enzyme inactivation in the temperature range of 35–55 °C. In conclusion, this literature can provide a paramount reference to help the food processors planning to execute these non-thermal techniques for processing several food products.
Data availability
There are no datasets associated with this review article.
References
Rahman MS. Handbook of food preservation. Boca Raton: CRC Press; 2007. https://doi.org/10.1201/9781420017373.
Friede W, Mieke F. Beverage intake: nutritional role, challenges, and opportunities for developing Countries. Nutr Beverages. 2019;1(1):143–73. https://doi.org/10.1016/B978-0-12-816842-4.00005-8.
Nazhand A, Souto EB, Lucarini M, Souto SB, Durazzo A, Santini A. Ready to use therapeutically beverages: focus on functional beverages containing probiotics, prebiotics and synbiotics. Beverages. 2020;6(2):26. https://doi.org/10.3390/beverages6020026.
Dini I. An overview of functional beverages. Funct Med Beverages. 2019;1:1–40. https://doi.org/10.1016/B978-0-12-816397-9.00001-7.
Marsh AJ, Hill C, Ross RP, Cotter PD. Fermented beverages with health-promoting potential: past and future perspectives. Trends Food Sci. 2014;38(2):113–24. https://doi.org/10.1016/j.tifs.2014.05.002.
Yilmaz-Akyuz E, Ustun-Aytekin O, Bayram B, Tutar Y. Nutrients, bioactive compounds, and health benefits of functional and medicinal beverages. In: Grumezescu Alexandru Mihai, Holban Alina Maria, editors. Nutrients in beverages. Cambridge: Academic Press; 2019. p. 175–235. https://doi.org/10.1016/B978-0-12-816842-4.00006-X.
Pereira RN, Vicente AA. Environmental impact of novel thermal and nonthermal technologies in food processing. Food Res Int. 2010;43(1):1936–43. https://doi.org/10.1016/j.foodres.2009.09.013.
Iqbal A, Murtaza A, Hu W, et al. Activation and inactivation mechanisms of polyphenol oxidase during thermal and nonthermal methods of food processing. Food Bioprod Process. 2019;117(1):170–82. https://doi.org/10.1016/j.fbp.2019.07.006.
Petruzzi L, Campaniello D, Speranza B, et al. Thermal treatments for fruit and vegetable juices and beverages: a literature overview. Compr Rev Food Sci Food Saf. 2017;16(4):668–91. https://doi.org/10.1111/1541-4337.12270.
Lodhi S, Vadnere GP. Health-promoting ingredients in beverages. Value Added Ingred Enrich Beverages. 2019;20(1):37–61.
Thirumdas R, Sarangapani C, Annapure US. Cold plasma: a novel nonthermal technology for food processing. Food Biophys. 2014;10(1):1–11. https://doi.org/10.1007/s11483-014-9382-z.
Jadhav HB, Annapure US, Deshmukh RR. Non-thermal technologies for food processing. Front Nutr. 2021;8(1):6570–90. https://doi.org/10.3389/fnut.2021.657090.
Choudhary R, Bandla S. Ultraviolet pasteurization for food industry. Int J Food Sci Nutr. 2012;2(1):12–5. https://doi.org/10.5923/j.food.20120201.03.
Ramaswamy HS, Zaman SU, Smith JP. High pressure destruction kinetics of E. coli (O157: H7) and listeria monocytogenes (Scott A) in a fish slurry. J Food Eng. 2008;87(1):99–106. https://doi.org/10.1016/j.jfoodeng.2007.11.017.
Roobab U, Aadil RM, Madni GM, El-Din BA. The impact of nonthermal technologies on the microbiological quality of juices: a review. Compr Rev Food Sci Food Saf. 2018;17(2):437–57. https://doi.org/10.1111/1541-4337.12336.
Ohlsson T, Bengtsson N. Minimal processing of foods with non-thermal methods. In: Bengtsson N, editor. Minimal processing technologies in the food industries. Cambridge: Woodhead Publishing Ltd.; 2002.
Rifna EJ, Singh SK, Chakraborty S, Dwivedi M. Effect of thermal and non-thermal techniques for microbial safety in food powder: recent advances. Food Res Int. 2019;126(1):108654. https://doi.org/10.1016/j.foodres.2019.108654.
Chen P, Deng S, Cheng Y, et al. Non-thermal food pasteurization processes: an introduction. In: Doona CJ, Kustin K, Feeherry FE, editors., et al., Case studies in novel food processing technologies. Cambridge: Woodhead Publishing Ltd; 2010.
Brunner G. Supercritical fluids: technology and application to food processing. J Food Eng. 2005;67(1–2):21–33. https://doi.org/10.1016/j.jfoodeng.2004.05.060.
Hati S, Patel M, Yadav D. Food bioprocessing by nonthermal plasma technology. Curr Opin Food Sci. 2018;19(1):85–91. https://doi.org/10.1016/j.cofs.2018.03.011.
Mazzutti S, Pedrosa RC, Salvador Ferreira SR. Green processes in foodomics. In: Salvador Ferreira SR, editor. Supercritical fluid extraction of bioactives. Reference module in food science. Cambridge: Elsevier; 2020.
Terefe NS, Janakievski F, Glagovskaia O, Stockmann R. Forward osmosis: an emerging non-thermal concentration technology for liquid foods. In: Stockmann R, editor. Reference module in food science. Cambridge: Elsevier; 2019.
Liu J, Bi J, McClements DJ, Liu X, et al. Impacts of thermal and non-thermal processing on structure and functionality of pectin in fruit-and vegetable-based products: a review. Carbohydr Polym. 2020;250(1):1168–90. https://doi.org/10.1016/j.carbpol.2020.116890.
Zheng L, Sun DW. Innovative applications of power ultrasound during food freezing processes—a review. Trends Food Sci. 2006;17(1):16–23. https://doi.org/10.1016/j.tifs.2005.08.010.
Villamiel M, de Jong P. Influence of high-intensity thermosonication and heat treatment in continuous flow on fat, proteins, and native enzymes of milk. J Agric Food Chem. 2000;48(1):472–8. https://doi.org/10.1021/jf990181s.
Ercan SS, Soysal C. Use of ultrasound in food preservation. Nat Sci. 2013;5(8):5–13. https://doi.org/10.4236/ns.2013.58A2002.
Chemat F, Khan MK. Applications of ultrasound in food technology: processing, preservation and extraction. Ultrason Sonochem. 2011;18(4):813–35. https://doi.org/10.1016/j.ultsonch.2010.11.023.
Mason T. Sonochemistry and the environment—providing a “green” link between chemistry, physics and engineering. Ultrason Sonochem. 2007;14(1):476–83. https://doi.org/10.1016/j.ultsonch.2006.10.008.
Bhangu SK, Ashokkumar M. Theory of sonochemistry. Top Curr Chem. 2016;374(4):56. https://doi.org/10.1007/s41061-016-0054-y.
Lopez-Malo A, Palou E, Jimenez-Fernandez M, Alzamora SM, Guerrero S. Multifactorial fungal inactivation combining thermosonication and antimicrobials. J Food Eng. 2005;67(1–2):87–93. https://doi.org/10.1016/j.jfoodeng.2004.05.072.
Cheng LH, Soh CY, Liew SC, Teh FF. Effects of sonication and carbonation on guava juice quality. Food Chem. 2007;104(4):1396–401. https://doi.org/10.1016/j.foodchem.2007.02.001.
Tiwari BK, O’Donnell CP, Patras A, Cullen PJ. Anthocyanin and ascorbic acid degradation in sonicated strawberry juice. J Agric Food Chem. 2008;56(21):10071–7. https://doi.org/10.1021/jf801824v.
Tiwari BK, Donnell CPO, Muthukumarappan K, Cullenn PJ. Ascorbic acid degradation kinetics of sonicated orange juice during storage and comparison with thermally pasteurized juice. LWT Food Sci Techno. 2009;42(3):700–4. https://doi.org/10.1016/j.lwt.2008.10.009.
Tiwari BK, ODonnell CP, Cullen PJ. Effect of sonication on retention of anthocyanins in blackberry juice. J Food Eng. 2009;93(1):166–71. https://doi.org/10.1016/j.jfoodeng.2009.01.027.
GómezLópez VM, Buitrago ME, Tapia MS, MartínezYépez A. Effect of ultrasonication on microbial quality, colour, and ascorbic acid content of passion-fruit juice during storage. Acta Aliment. 2017;46:470–80. https://doi.org/10.1556/066.2017.46.4.10.
Calderón-Martínez V, Delgado-Ospina J, Ramírez-Navas JS, Flórez-López E, Valdés-Restrepo MP, Grande-Tovar CD, Chaves-López C. Effect of pretreatment with low-frequency ultrasound on quality parameters in gulupa (Passiflora edulis Sims) pulp. Appl Sci. 2021;11(4):1734. https://doi.org/10.3390/app11041734.
Wang S, Liu Z, Zhao S, Zhang L, Li C, Liu S. Effect of combined ultrasonic and enzymatic extraction technique on the quality of noni (Morinda citrifolia L.) juice. Ultrason Sonochem. 2023;92(1):106231. https://doi.org/10.1016/j.ultsonch.2022.106231.
Kesavan RK, Nayak PK, Mili P. Quality assessment of Kaji Lemon (Citrus limon) juice under ultrasound and pasteurization processing conditions. InIntelligent Techniques and Applications in Science and Technology: Proceedings of the First International Conference on Innovations in Modern Science and Technology. 2020; 1(1) :16-23
Wang J, Wang J, Ye J, et al. Influence of high-intensity ultrasound on bioactive compounds of strawberry juice: profiles of ascorbic acid, phenolics, antioxidant activity and microstructure. Food Control. 2019;96:128–36. https://doi.org/10.1016/j.foodcont.2018.09.007.
Muhammad Saeeduddin, Muhammad Abid, Saqib Jabbar, et al. Quality assessment of pear juice under ultrasound and commercial pasteurization processing conditions. LWT. 2015;64(1):452–8. https://doi.org/10.1016/j.lwt.2015.05.005.
Muhammd Farhadi Chitgar, Mehran Aalami, Yahya Maghsoudlou, Elnaz Milani. Comparative study on the effect of heat treatment and sonication on the quality of barberry (Berberis Vulgaris) juice. J Food Process Preserv. 2016;41(3):e12956. https://doi.org/10.1111/jfpp.12956.
Engmann Felix N, Yongkun Ma, William Tchabo, Hui Ma. Ultrasonication treatment effect on anthocyanins, color, microorganisms and enzyme inactivation of mulberry (Moraceae nigra) juice. J Food Process Preserv. 2014;39(6):854–62. https://doi.org/10.1111/jfpp.12296.
Wang J, Liu Q, Xie B, Sun Z. Effect of ultrasound combined with ultraviolet treatment on microbial inactivation and quality properties of mango juice. Ultrason Sonochem. 2020;64(1):105000. https://doi.org/10.1016/j.ultsonch.2020.105000.
Jacob A, Sudagar IP, Pandiselvam R, Rajkumar P, Rajavel M. Optimization of ultrasound processing parameters for preservation of matured coconut water using a central composite design. Qual Assur Saf Crop. 2022;14(1):33–41. https://doi.org/10.1558/qas.v14iSP1.1145.
Abid M, Jabbar S, Wu T, et al. Effect of ultrasound on different quality parameters of apple juice. Ultrasonics Sonochem. 2013;20(5):1182–7. https://doi.org/10.1016/j.ultsonch.2013.02.010.
Guimaraes JT, Silva EK, Alvarenga VO, Costa ALR, et al. Physicochemical changes and microbial inactivation after high-intensity ultrasound processing of prebiotic whey beverage applying different ultrasonic power levels. Ultrason Sonochem. 2018;44(1):251–60. https://doi.org/10.1016/j.ultsonch.2018.02.012.
Cassani L, Tomadoni B, Moreira R. Green ultrasound-assisted processing for extending the shelf-life of prebiotic-rich strawberry juices. J Sci Food Agric. 2020;100(15):5518–26. https://doi.org/10.1002/jsfa.10604.
Phan KTK, Phan HT, Brennan CS, Phimolsiripol Y. Nonthermal plasma for pesticide and microbial elimination on fruits and vegetables: an overview. Int J Food Sci Technol. 2017;52(1):2127–37. https://doi.org/10.1111/ijfs.13509.
Irving L. Oscillations in ionized gases. Proc Natl Acad Sci USA. 1928;14(8):627–37. https://doi.org/10.1073/2Fpnas.14.8.627.
Luo QZ, DAngelo N, Merlino RL. Shock formation in a negative ion plasma. Phys Plasmas. 1998;5(8):2868–70. https://doi.org/10.1063/1.873007.
Hertwig C, Meneses N, Mathys A. Cold atmospheric pressure plasma and low energy electron beam as alternative nonthermal decontamination technologies for dry food surfaces: a review. Trends Food Sci. 2018;77(1):131–42. https://doi.org/10.1016/j.tifs.2018.05.011.
Mandal R, Singh A, Pratap-Singh A. Recent developments in cold plasma decontamination technology in the food industry. Trends Food Sci. 2018;80(1):93–103. https://doi.org/10.1016/j.tifs.2018.07.014.
Stoica M, Alexe P, Mihalcea L. Atmospheric cold plasma as new strategy for foods processing-an overview. Innov Rom Food Biotechnol. 2014;15(1):1–8.
Roth JR, Nourgostar S, Bonds TA. The one atmosphere uniform glow discharge plasma (OAUGDP)—a platform technology for the 21st century. IEEE Trans Plasma Sci. 2007;35:233–50. https://doi.org/10.1109/TPS.2007.892711.
Alves Filho E, Rodrigues T, Fernandes F, et al. An untargeted chemometric evaluation of plasma and ozone processing effect on volatile compounds in orange juice. IFSET. 2019;53:63–9. https://doi.org/10.1016/j.ifset.2017.10.001.
Herceg Z, Kovacevic DB, Kljusurić JG, et al. Gas phase plasma impact on phenolic compounds in pomegranate juice. Food Chem. 2016;190(1):665–72. https://doi.org/10.1016/j.foodchem.2015.05.135.
Liao X, Liu D, Xiang Q, et al. Inactivation mechanisms of non-thermal plasma on microbes: a review. Food Control. 2017;75(1):83–91. https://doi.org/10.1016/j.foodcont.2016.12.021.
Hou Y, Wang R, Gan Z, et al. Effect of cold plasma on blueberry juice quality. Food Chem. 2019;290(1):79–86. https://doi.org/10.1016/j.foodchem.2019.03.123.
Ma TJ, Lan WS. Effects of non-thermal plasma sterilization on volatile components of tomato juice. IJEST. 2015;12(12):3767–72. https://doi.org/10.1007/s13762-015-0796-z.
Dasan BG, Boyaci IH. Effect of cold atmospheric plasma on inactivation of Escherichia coli and physicochemical properties of apple, orange, tomato juices, and sour cherry nectar. Food Bioproc Tech. 2018;11(2):334–43. https://doi.org/10.1007/s11947-017-2014-0.
Mahnot NK, Mahanta CL, Farkas BE, Keener KM, Misra NN. Atmospheric cold plasma inactivation of Escherichia coli and listeria monocytogenes in tender coconut water: Inoculation and accelerated shelf-life studies. Food Control. 2019;106(1):1066–78. https://doi.org/10.1016/j.foodcont.2019.06.004.
Pankaj SK, Wan Z, Colonna W, Keener KM. Effect of high voltage atmospheric cold plasma on white grape juice quality. JSFA. 2017;97(12):4016–21. https://doi.org/10.1002/jsfa.8268.
Xu L, Garner AL, Tao B, Keener KM. Microbial inactivation and quality changes in orange juice treated by high voltage atmospheric cold plasma. Food Bioproc Tech. 2017;10(10):1778–91. https://doi.org/10.1007/s11947-017-1947-7.
Xiang Q, Liu X, Li J, Liu S, Zhang H, Bai Y. Effects of dielectric barrier discharge plasma on the inactivation of zygosaccharomyces rouxii and quality of apple juice. Food Chem. 2018;254:201–7. https://doi.org/10.1016/j.foodchem.2018.02.008567.
Starek A, Pawłat J, Chudzik B, et al. Evaluation of selected microbial and physicochemical parameters of fresh tomato juice after cold atmospheric pressure plasma treatment during refrigerated storage. Sci Rep. 2019;9(1):1–11. https://doi.org/10.1038/s41598-019-44946-1.
Paixão LMN, Fonteles TV, Oliveira VS, et al. Cold plasma effects on functional compounds of siriguela juice. Food Bioproc Tech. 2019;12:110–21. https://doi.org/10.1007/s11947-018-2197-z.
Umair M, Jabbar S, Senan AM, Sultana T, Nasiru MM, Shah AA, Zhuang H, Jianhao Z. Influence of combined effect of ultra-sonication and high-voltage cold plasma treatment on quality parameters of carrot juice. Foods. 2019;8(11):593. https://doi.org/10.3390/foods8110593.
Hosseini SM, Rostami S, Hosseinzadeh Samani B, Lorigooini Z. The effect of atmospheric pressure cold plasma on the inactivation of Escherichia coli in sour cherry juice and its qualitative properties. Food Sci Nutr. 2020;8(2):870–83. https://doi.org/10.1002/2Ffsn3.1364.
Ding H, Wang T, Sun Y, Zhang Y, Wei J, Cai R, Guo C, Yuan Y, Yue T. Role and mechanism of cold plasma in inactivating Alicyclobacillus acidoterrestris in apple juice. Foods. 2023;12(7):1531. https://doi.org/10.3390/foods12071531.
Wang X, Han Y, Niu H, Zhang LiHua, Xiang Q, Zong W. Alternaria mycotoxin degradation and quality evaluation of jujube juice by cold plasma treatment. Food Control. 2022;137:108926. https://doi.org/10.1016/j.foodcont.2022.108926.
Ozen Emine, Kumar Govindaraj Dev, Mishra Abhinav, Rakesh K. Singh, inactivation of Escherichia coli in apple cider using atmospheric cold plasma. Int J Food Microbiol. 2022;382:109913. https://doi.org/10.1016/j.ijfoodmicro.2022.109913.
Porto ECM, de Brito ES, Rodrigues S, Fernandes FAN. Effect of atmospheric cold plasma on the aroma of pineapple juice: improving fresh and fruity notes and reducing undesired pungent and sulfurous aromas. Processes. 2023;11(8):2303. https://doi.org/10.3390/pr11082303.
Wu X, Luo Y, Zhao F, SM M, Mu G. Influence of dielectric barrier discharge cold plasma on physicochemical property of milk for sterilization. Plasma Process Polym. 2021;18(1):1900219. https://doi.org/10.1002/ppap.201900219.
Chen D, Peng P, Zhou N, Cheng Y, Min M, Ma Y, et al. Evaluation of cronobacter sakazakii inactivation and physicochemical property changes of non-fat dry milk powder by cold atmospheric plasma. Food Chem. 2019;290:270–6. https://doi.org/10.1016/j.foodchem.2019.03.149.
Segat A, Misra NN, Cullen PJ, Innocente N. Atmospheric pressure cold plasma (ACP) treatment of whey protein isolate model solution. IFSET. 2015;29:247–54. https://doi.org/10.1016/j.ifset.2015.03.014.
Mahnot NK, Mahanta CL, Keener KM, et al. Strategy to achieve a 5-log salmonella inactivation in tender coconut water using high voltage atmospheric cold plasma (HVACP). Food Chem. 2019;284:303–11. https://doi.org/10.1016/j.foodchem.2019.01.084.
Silva EK, Meireles MAA, Saldana MD. Supercritical carbon dioxide technology: a promising technique for the non-thermal processing of freshly fruit and vegetable juices. Trends Food Sci. 2020;97(1):381–90. https://doi.org/10.1016/j.tifs.2020.01.025.
Smigic N, Djekic I, Tomic N, Udovicki B, Rajkovic A. The potential of foods treated with supercritical carbon dioxide (sc-CO2) as novel foods. Br Food J. 2019;121(3):815–34. https://doi.org/10.1108/BFJ-03-2018-0168.
Deotale SM, Dutta S, Moses JA, Anandharamakrishnan C. Advances in supercritical carbon dioxide assisted sterilization of biological matrices. In: Knoerzer K, Juliano P, Smithers G, editors. Innov food process technol. Cambridge: Elsevier; 2021.
Lefebvre T, Destandau E, Lesellier E. Sequential extraction of carnosic acid, rosmarinic acid and pigments (carotenoids and chlorophylls) from rosemary by online supercritical fluid extraction supercritical fluid chromatography. J Chromatogr A. 2020;16(39):461709. https://doi.org/10.1016/j.chroma.2020.461709.
Verma DK, Srivastav PP. Science and technology of aroma, flavor, and fragrance in rice. New York: Apple Academic Press; 2018. https://doi.org/10.1201/b22468.
Zhou L, Bi X, Xu Z, Yang Y, Liao X. Effects of high-pressure CO2 processing on flavor, texture, and color of foods. Crit Rev Food Sci Nutr. 2018;55(6):750–68. https://doi.org/10.1080/10408398.2012.677871.
Fabroni S, Amenta M, Timpanaro N, Rapisarda P. Supercritical carbon dioxide-treated blood orange juice as a new product in the fresh fruit juice market. Innov Food Sci Emerg. 2016;11(1):477–84. https://doi.org/10.1016/j.ifset.2010.02.004.
Zou H, Lin T, Bi X, Zhao L, Wang Y, Liao X. Comparison of high hydrostatic pressure, high-pressure carbon dioxide and high-temperature short-time processing on quality of mulberry juice. Food Bioproc Tech. 2016;9(2):217–31. https://doi.org/10.1007/s11947-015-1606-9.
Amaral GV, Silva EK, Cavalcanti RN, Martins CP, et al. Whey-grape juice drink processed by supercritical carbon dioxide technology: physicochemical characteristics, bioactive compounds and volatile profile. Food Chem. 2018;239(1):697–703. https://doi.org/10.1016/j.foodchem.2017.07.003.
Murtaza A, Iqbal A, Linhu Z, et al. Effect of high pressure carbon dioxide on the aggregation and conformational changes of polyphenol oxidase from apple (Malus domestica) juice. Innov Food Sci Emerg. 2019;54(1):43–50. https://doi.org/10.1016/j.ifset.2019.03.001.
Bertolini FM, Morbiato G, Facco P, et al. Optimization of the supercritical CO2 pasteurization process for the preservation of high nutritional value of pomegranate juice. J Supercrit Fluids. 2020;164(S2):104914. https://doi.org/10.1016/j.supflu.2020.104914.
PaniaguaMartínez I, Mulet A, García-Alvarado MA, Benedito J. Inactivation of the microbiota and effect on the quality attributes of pineapple juice using a continuous flow ultrasound-assisted supercritical carbon dioxide system. Food Sci Technol Int. 2018;24(7):547–54. https://doi.org/10.1177/1082013218774694.
Marszałek K, Skąpska S, Woźniak Ł, Sokołowska B. Application of supercritical carbon dioxide for the preservation of strawberry juice: microbial and physicochemical quality, enzymatic activity and the degradation kinetics of anthocyanins during storage. IFSET. 2015;32(1):101–9. https://doi.org/10.1016/j.ifset.2015.10.005.
Manzocco L, Plazzotta S, Spilimbergo S, Nicoli MC. Impact of highpressure carbon dioxide on polyphenoloxidase activity and stability of fresh apple juice. LWT. 2017;85(1):363–71. https://doi.org/10.1016/j.lwt.2016.11.052.
Torabian G, Bahramian B, Zambon A, Spilimbergo S, Adil Q, Schindeler A, et al. A hybrid process for increasing the shelf life of elderberry juice. J Supercrit Fluids. 2018;140(1):406–14. https://doi.org/10.1016/j.supflu.2018.07.023.
Sultani TM, Francisco ACP, Dacanal GC, Oliveira AL, Petrus RR. The combined effect of supercritical carbon dioxide and mild temperatures on pectin methylesterase inactivation in orange juice. Food Sci Technol Int. 2023. https://doi.org/10.1177/10820132231172363.
Trych U, Buniowska M, Skąpska S, Kapusta I, Marszałek K. Bioaccessibility of antioxidants in blackcurrant juice after treatment using supercritical carbon dioxide. Molecules. 2022;27(3):1036. https://doi.org/10.3390/molecules27031036.
Mosca AC, Menghi L, Aprea E, Mazzucotelli M, Benedito J, Zambon A, Spilimbergo S, Gasperi F. Effect of CO2 preservation treatments on the sensory quality of pomegranate juice. Molecules. 2020;25(23):5598. https://doi.org/10.3390/molecules25235598.
Van Loey IA, Smout C, Hendrickx M. High hydrostatic pressure technology in food preservation. In: Zeuthen P, Bogh-Sorensen L, editors. Food preservation techniques. Cambridge: Woodhead Publishing; 2003.
Huang Y, Ye M, Cao X, Chen H. Pulsed light inactivation of murine norovirus, tulane virus, Escherichia coli O157: H7 and salmonella in suspension and on berry surfaces. Food Microbiol. 2017;61(1):1–4. https://doi.org/10.1016/j.fm.2016.08.001.
RoselloSoto E, Poojary MM, Barba FJ, et al. Thermal and non-thermal preservation techniques of tiger nuts’ beverage “horchata de chufa”. Implications for food safety, nutritional and quality properties. Food Res Int. 2018;105(1):945–51. https://doi.org/10.1016/j.foodres.2017.12.014.
Moralesde la Pena M, WeltiChanes J, Martín-Belloso O. Novel technologies to improve food safety and quality. Curr Opin Food Sci. 2019;30(1):1–7. https://doi.org/10.1016/j.cofs.2018.10.009.
Huang HW, Hsu CP, Wang CY. Healthy expectations of high hydrostatic pressure treatment in food processing industry. J Food Drug Anal. 2020;28(1):1–13. https://doi.org/10.1016/j.jfda.2019.10.002.
Santhirasegaram V, Razali Z, Somasundram C. Safety improvement of fruit juices by novel thermal and nonthermal processing. In: Kotzekidou Parthena, editor. Food hygiene and toxicology in ready-to-eat foods. Cambridge: Academic Press; 2016.
Andres V, Mateo-Vivaracho L, Guillamon E, et al. High hydrostatic pressure treatment and storage of soy-smoothies: colour, bioactive compounds and antioxidant capacity. LWT Food Sci. 2016;69(1):123–30. https://doi.org/10.1016/j.lwt.2016.01.033.
Liu F, Zhang X, Zhao L, Wang Y, Liao X. Potential of high-pressure processing and high-temperature/short-time thermal processing on microbial, physicochemical and sensory assurance of clear cucumber juice. Innov Food Sci Emerg. 2016;34:51–8. https://doi.org/10.1016/j.ifset.2015.12.030.
Liu Y, Zhao XY, Zou L, et al. Effect of high hydrostatic pressure on overall quality parameters of watermelon juice. Food Sci Technol Int. 2013;19(3):197–207. https://doi.org/10.1177/1082013212442194.
Wang F, Du BL, Cui ZW, et al. Effects of high hydrostatic pressure and thermal processing on bioactive compounds, antioxidant activity, and volatile profile of mulberry juice. Food Sci Technol Int. 2017;23(2):119–27. https://doi.org/10.1177/1082013216659610.
Khan MK, Ahmad K, Hassan S, et al. Effect of novel technologies on polyphenols during food processing. Innov Food Sci Emerg. 2018;45(1):361–81. https://doi.org/10.1016/j.ifset.2017.12.006.
Yuan B, Danao MC, Lu M, Weier SA, Stratton JE, Weller CL. High pressure processing (HHP) of aronia berry puree: pilot scale processing and a shelf-life study. Innov Food Sci Emerg Technol. 2018;47:241–8. https://doi.org/10.1016/j.ifset.2018.03.006.
Chang C, Ko W, Chen Y, et al. Evaluation of using high-pressure homogenization technology in enhancing the aroma synthesis of sorghum spirits. LWT. 2019;105(1):314–20. https://doi.org/10.1016/j.lwt.2019.01.053.
Fernandez MV, Denoya GI, Jagus RJ, et al. Microbiological, antioxidant and physicochemical stability of a fruit and vegetable smoothie treated by high pressure processing and stored at room temperature. LWT. 2019;105(1):206–10. https://doi.org/10.1016/j.lwt.2019.02.030.
Machado KIA, Roquetto AR, Moura CS, et al. Comparative impact of thermal and high isostatic pressure inactivation of gram-negative microorganisms on the endotoxic potential of reconstituted powder milk. LWT. 2019;106(7):78–82. https://doi.org/10.1016/j.lwt.2019.02.064.
Stratakos AC, Inguglia ES, Linton M, et al. Effect of high-pressure processing on the safety, shelf life and quality of raw milk. Innov Food Sci Emerg Technol. 2019;52(1):325–33. https://doi.org/10.1016/j.ifset.2019.01.009.
Sanchez L, Pérez MD, Parron JA. HHP in dairy products: impact on quality and applications. In: Barba Francisco J, editor. Present and future of high pressure processing. Amsterdam: Elsevier; 2020.
Razali MF, Narayanan S, MdHazmi NA, et al. Minimal processing for goat milk preservation: effect of high-pressure processing on its quality. J Food Process Preserv. 2021;45(2):e15590. https://doi.org/10.1111/jfpp.15590.
Torres B, Tiwari B, Patras A, et al. Stability of anthocyanins and ascorbic acid of high pressure processed blood orange juice during storage. Innov Food Sci Emerg Technol. 2011;12(2):93–7. https://doi.org/10.1016/j.ifset.2011.01.005.
Aghajanzadeh S, Ziaiifar AM, Verkerk R. Effect of thermal and non-thermal treatments on the color of citrus juice: a review. Food Rev Intl. 2021;31(1):1–23. https://doi.org/10.1080/87559129.2021.2012799.
Inácio RS, Barros R, Saraiva JA, et al. Optimization of raw ewes’ milk high-pressure pre-treatment for improved production of raw milk cheese. Foods. 2022;11(3):435.
Lai YY, Chen JH, Liu YC, et al. Evaluation of microbiological safety, physicochemical and aromatic qualities of shiikuwasha (Citrus depressaHayata) juice after high pressure processing. J Food Sci Technol. 2022;59(3):990–1000. https://doi.org/10.1007/s13197-021-05103-7.
Pokhrel PR, Boulet C, Yildiz S, Sablani S, Tang J, Barbosa Canovas GV. Effect of high hydrostatic pressure on microbial inactivation and quality changes in carrot-orange juice blends at varying pH. LWT. 2022;159:113219. https://doi.org/10.1016/j.lwt.2022.113219.
Xia Qile, Liu Chenxing, Cao Yan, Zhao Yuqi, Shengmin Lu, Dan Wu, Aniya Rongfa Guan. Improving quality of sea buckthorn juice by high-pressure processing. LWT. 2023;185:115149. https://doi.org/10.1016/j.lwt.2023.115149.
Błaszczak Wioletta, Amarowicz Ryszard, Adrian Górecki R. Antioxidant capacity, phenolic composition and microbial stability of aronia juice subjected to high hydrostatic pressure processing. Innov IFSET. 2017;39:141–7. https://doi.org/10.1016/j.ifset.2016.12.005.
Porębska I, Sokołowska B, Skąpska S, Rzoska SJ. Treatment with high hydrostatic pressure and supercritical carbon dioxide to control alicyclobacillus acidoterrestris spores in apple juice. Food Control. 2016. https://doi.org/10.1016/j.foodcont.2016.06.005.
Sokołowska B, Woźniak Ł, Skąpska S, Porębska I, Nasiłowska J, Rzoska SJ. Evaluation of quality changes of beetroot juice after high hydrostatic pressure processing. High Press Res. 2017;37(2):214–22. https://doi.org/10.1080/08957959.2017.1302443.
Xinxing Xu, Deng J, Luo D, Bao Y, et al. Comparative study of high hydrostatic pressure and high temperature short time processing on quality of clear and cloudy Se-enriched kiwifruit juices. IFSET. 2018;49:1–12. https://doi.org/10.1016/j.ifset.2018.07.010.
Patrignani F, Mannozzi C, Tappi S, Tylewicz U, et al. (Ultra) High pressure homogenization potential on the shelf-life and functionality of kiwifruit juice. Front Microbiol. 2019;10:246. https://doi.org/10.3389/fmicb.2019.00246.
Zhuojia Xu, Zhao M, Chen Q, et al. High hydrostatic pressure improved properties of peanut sprout juice and its effect on resveratrol. Food Biosci. 2023;56:2212–4292. https://doi.org/10.1016/j.fbio.2023.103061.
TorresOssandón MJ, Castillo L, Uribe E, Bilbao-Sainz C, Ah-Hen KS, Vega-Gálvez A. Combined effect of high hydrostatic pressure and proteolytic fraction P1G10 from Vasconcellea cundinamarcensis latex against Botrytis cinerea in grape juice. Foods. 2023;12(18):3400. https://doi.org/10.3390/foods12183400.
Sourri P, Argyri AA, Nychas G-JE, Tassou CC, Panagou EZ. The effect of temperature-assisted high hydrostatic pressure on the survival of alicyclobacillus acidoterrestris inoculated in orange juice throughout storage at different isothermal conditions. Fermentation. 2022;8(7):308. https://doi.org/10.3390/fermentation8070308.
Buitimea-Cantúa GV, Rico-Alderete IA, Rostro-Alanís MdJ, Welti-Chanes J, Escobedo-Avellaneda ZJ, Soto-Caballero MC. Effect of high hydrostatic pressure and pulsed electric fields processes on microbial safety and quality of black/red raspberry juice. Foods. 2022;11(15):2342. https://doi.org/10.3390/foods11152342.
Raghubeer EV, Phan BN, Onuoha E, Diggins S, Aguilar V, Swanson S, Lee A. The use of high-pressure processing (HHP) to improve the safety and quality of raw coconut (Cocos nucifera L) water. Int J Food Microbiol. 2020;331:108697.
Lou X, Jin Y, Tian H, et al. High-pressure and thermal processing of cloudy hawthorn berry (Crataegus pinnatifida) juice: Impact on microbial shelf-life, enzyme activity and quality-related attributes. Food Chem. 2022;372(1):e131313. https://doi.org/10.1016/j.foodchem.2021.131313.
Popović V, Koutchma T, Pagan J. Emerging applications of ultraviolet light-emitting diodes for foods and beverages. Innov Food Sci Technol Voctoria. 2021;1(1):335–44. https://doi.org/10.1016/B978-0-08-100596-5.22667-X.
Guerrero-Beltran JA, Ochoa-Velasco CE. Ultraviolet-C light technology and systems for preservation of fruit juices and beverages. Innov Food Process Technol Voctoria. 2021. https://doi.org/10.1016/B978-0-08-100596-5.22937-5.
Koutchma T, Bissonnette S, Popovic V. An update on research, development and implementation of UV and pulsed light technologies for nonthermal preservation of milk and dairy products. Innov Food Process Technol Voctoria. 2021. https://doi.org/10.1016/B978-0-08-100596-5.22680-2.
Nicorescu I, Nguyen B, Moreau-Ferret M, et al. Pulsed light inactivation of Bacillus subtilis vegetative cells in suspensions and spices. Food Control. 2013;31(1):151–7. https://doi.org/10.1016/j.foodcont.2012.09.047.
John D, Ramaswamy HS. Pulsed light technology to enhance food safety and quality: a mini-review. Curr Opin Food Sci. 2018;23(1):70–9. https://doi.org/10.1016/j.cofs.2018.06.004.
Vorobiev E, Lebovka N. Pulsed electric field in green processing and preservation of food products. In: Chemat F, Vorobiev E, editors. Green food processing techniques. France: Elsevier Inc.; 2019.
Niu D, Zeng XA, Ren EF, et al. Review of the application of pulsed electric fields (PEF) technology for food processing in China. Food Res Int. 2020;137(1):109715. https://doi.org/10.1016/j.foodres.2020.109715.
Ekezie CFG, Sun DW, Cheng JH. A review on recent advances in cold plasma technology for the food industry: current applications and future trends. Trends Food Sci. 2017;69:46–58. https://doi.org/10.1016/j.tifs.2017.08.007.
Pataro G, Munoz A, Palgan I, et al. Bacterial inactivation in fruit juices using a continuous flow pulsed light (PL) system. Food Res Int. 2011;44(6):1642–8. https://doi.org/10.1016/j.foodres.2011.04.048.
Siddique MAB, Maresca P, Pataro G, et al. Effect of pulsed light treatment on structural and functional properties of whey protein isolate. Food Res Int. 2016;87(1):189–96. https://doi.org/10.1016/j.foodres.2016.07.017.
Kwaw E, Ma Y, Tchabo W, et al. Effect of pulsed light treatment on the phytochemical, volatile, and sensorial attributes of lactic-acid-fermented mulberry juice. Intl J Food Prop. 2018;21(1):213–28. https://doi.org/10.1080/10942912.2018.1446024.
Preetha P, Pandiselvam R, Varadharaju N, et al. Effect of pulsed light treatment on inactivation kinetics of Escherichia coli (MTCC 433) in fruit juices. Food Control. 2020;121(1):107547. https://doi.org/10.1016/j.foodcont.2020.107547.
Rahaman A, Zeng X-A, Farooq MA, et al. Effect of pulsed electric fields processing on physiochemical properties and bioactive compounds of apricot juice. J Food Process Eng. 2020;43(8):e13449. https://doi.org/10.1111/jfpe.13449.
Dziadek K, Kopec A, Dróżdż T, et al. Effect of pulsed electric field treatment on shelf life and nutritional value of apple juice. J Food Sci Techol. 2019;56(3):1184–91. https://doi.org/10.1007/s13197-019-03581-4.
Yousuf A, Rahman MA, Uddin MR, et al. Pineapple juice preservation by pulsed electric field treatment. Open J Biol Sci. 2020;5(1):006–12. https://doi.org/10.1735/ojbs.000016.
Tamilselvan K, Kumar R, Lakshmana JH, et al. Pulsed electric field processing of functional drink based on tender coconut water (Cococus nucifera L)—nannari (Hemidesmus indicus) blended beverage. CFJST. 2014;6(2):84–96. https://doi.org/10.1750/CJFST.2014.6.2.04.
Aslam Raouf, Alam Mohammed Shafiq, Ali Asgar, Tao Yang, Sivakumar Manickam A. chemometric approach to evaluate the effects of probe-type ultrasonication on the enzyme inactivation and quality attributes of fresh amla juice. Ultrason Sonochem. 2023;92(1):106268. https://doi.org/10.1016/j.ultsonch.2022.106268.
Dhar R, Chakraborty S. Influence of voltage and distance on quality attributes of mixed fruit beverage during pulsed light treatment and kinetic modeling. J Food Process Eng. 2020;43(11):e13517. https://doi.org/10.1111/jfpe.13517.
Dhar R, Chakraborty S. Pasteurization of bael fruit (Aegle marmelos) juice using high-intensity pulsed light treatment. Food Control. 2023;152(1):109826. https://doi.org/10.1016/j.foodcont.2023.109826.
Shaik Lubna, Chakraborty Snehasis. Ultrasound processing of sweet lime juice: effect of matrix pH on microbial inactivation, enzyme stability, and bioactive retention. J Food Process Eng. 2022;46(6):14231. https://doi.org/10.1111/jfpe.14231.
Preetha P, Varadharaju N, Jeevarathinam G, Deepa J, Kumar AM, Balakrishnan M, Rajkumar P, Pandiselvam R. Optimization of continuous flow pulsed light system process parameters for microbial inactivation in tender coconut water, pineapple and orange juice. J Food Process Eng. 2023;46(3):e14254. https://doi.org/10.1111/jfpe.14254.
Basak S, Jha T, Chakraborty S. Pasteurization of tender coconut water by pulsed light treatment: microbial safety, enzymatic inactivation, and impact on physicochemical properties. IFSET. 2023;84(3):103302. https://doi.org/10.1016/j.ifset.2023.103302.
Bhagat B, Chakraborty S. Potential of pulsed light treatment to pasteurize pomegranate juice: microbial safety, enzyme inactivation, and phytochemical retention. LWT Food Sci Technol. 2022;159(1):113215. https://doi.org/10.1016/j.lwt.2022.113215.
Vollmer K, Chakraborty S, Bhalerao PP, et al. Effect of pulsed light treatment on natural microbiota, enzyme activity, and phytochemical composition of pineapple (Ananas comosus [L.] Merr.) juice. Food Bioproc Technol. 2020;13(1):1095–109. https://doi.org/10.1007/s11947-020-02460-7.
Kwaw E, Ma Y, Tchabo W, Apaliya MT, Sackey AS, Wu M, et al. Impact of ultrasonication and pulsed light treatments on phenolics concentration and antioxidant activities of lactic-acid-fermented mulberry juice. LWT Food Sci Technol. 2018;92(3):61–6. https://doi.org/10.1016/j.lwt.2018.02.016.
Kaya Z, Unluturk S, Martin-Belloso O, Soliva-Fortuny R. Effectiveness of pulsed light treatments assisted by mild heat on Saccharomyces cerevisiae inactivation in verjuice and evaluation of its quality during storage. IFSET. 2020;66(1):102517. https://doi.org/10.1016/j.ifset.2020.102517.
Wang B, Wei W, Zhang Y, Xu H, Ma H. Decontamination and quality assessment of freshly squeezed grape juice under spiral continuous flow-through pulsed light (SCFPL) treatment. J Food Process Preserv. 2022;46(1):e16186. https://doi.org/10.1111/jfpp.16186.
Pravallika K, Shaik L, Chakraborty S. Changes in the quality attributes of pulsed light and thermally pasteurized pomegranate (Punica granatum) juice stored at refrigerated condition (4 °C). Food Meas. 2023;17(6):1–19. https://doi.org/10.1007/s11694-023-02132-y.
Strathmann H, Giorno L, Drioli E. An introduction to membrane science and technology. In: Uragami Tadashi, editor. Consiglio nazionale delle ricerche. Rome: Wiley; 2006. https://doi.org/10.1002/9781118932551.ch1.
Kostantinos BP, Harris NL. Osmotic concentration of liquid foods. J Food Eng. 2001;49(2):201–6. https://doi.org/10.1016/S0260-8774(00)00222-3.
Jiao B, Cassano A, Drioli E. Recent advances on membrane processes for the concentration of fruit juices: a review. J Food Eng. 2004;63(3):303–24. https://doi.org/10.1016/j.jfoodeng.2003.08.003.
Crespo J, Brazinha C. Fundamentals of pervaporation. In: Basile A, Figoli A, Khayet M, editors. Pervaporation, vapour permeation and membrane distillation. Cambridge: Elsevier Ltd.; 2015. p. 1–17.
Wijmans JG, Baker RW. The solution-diusion model: a review. J Membr Sci. 1995;107(1–2):1–21. https://doi.org/10.1016/0376-7388(95)00102-I.
McCarron A, Donnelley M, McIntyre C, Parsons D. Challenges of up-scaling lentivirus production and processing. J Biotechnol. 2016;240(1):23–30. https://doi.org/10.1016/j.jbiotec.2016.10.016.
Li J, Wang Z, Ge Y, Sun Q, Hu X. Clarification and sterilization of raw depectinized apple juice by ceramic ultrafiltration membranes. J Sci Food Agric. 2006;86(1):148–55. https://doi.org/10.1002/jsfa.2281.
Jeong SJ, Jeong HS, Woo SH, Shin CS. Consequences of ultrafltration and ultraviolet on the quality of white birch (Betula Platyphylla var japonica) sap during storage. Aust J Crop Sci. 2013;7(8):1072–7. https://doi.org/10.3316/informit.409070807017399.
Sagu ST, Karmakar S, Nso EJ, Kapseu C, De S. Ultrafiltration of banana (Musa acuminata) juice using hollow fibers for enhanced shelf life. Food Bioproc Process. 2014;7(9):2711–22. https://doi.org/10.1007/s11947-014-1309-7.
Mondal M, Biswas PP, De S. Clarification and storage study of bottle gourd (Lagenaria siceraria) juice by hollow fibre ultrafiltration. Food Bioprod Process. 2016;100(1):1–15. https://doi.org/10.1016/j.fbp.2016.06.010.
Ghosh P, Garg S, Mohanty I, Sahoo D, Pradhan RC. Comparison and storage study of ultra-filtered clarified jamun (Syzygium cumini) juice. J Food Sci Technol. 2019;56(4):1877–89. https://doi.org/10.1007/s13197-019-03648-2.
Lamdande AG, Mittal R, Raghavarao KSMS. Flux evaluation based on fouling mechanism in acoustic field-assisted ultrafiltration for cold sterilization of tender coconut water. IFSET. 2020;61(1):102312. https://doi.org/10.1016/j.ifset.2020.102312.
Mahnot NK, Kalita D, Mahanta CL, Chaudhuri MK. Effect of additives on the quality of tender coconut water processed by nonthermal two stage microfiltration technique. LWT Food Sci Technol. 2014;59(2):1191–5. https://doi.org/10.1016/j.lwt.2014.06.040.
Carneiro L, Sa I, Gomes FS, Matta VM, Cabral LMC. Cold sterilization and clarifcation of pineapple juice by tangential microfltration. Desalination. 2002;148(1–3):93–8. https://doi.org/10.1016/S0011-9164(02)00659-8.
Bhattacharjee C, Saxena VK, Dutta S. Analysis of fouling and juice quality in crossflow ultrafiltration of watermelon juice. Food Sci Technol. 2017;38(1):71–6. https://doi.org/10.1590/1678-457x.15217.
Le TT, Vu LT, Le NL. Effects of membrane pore size and transmembrane pressure on ultrafiltration of red-fleshed dragon fruit (Hylocereus polyrhizus) juice. J Chem Technol Biotechnol. 2021;96(6):1561–72. https://doi.org/10.1002/jctb.6672.
Yilmaz E, Bagci PO. Ultrafiltration of broccoli juice using polyethersulfone membrane: fouling analysis and evaluation of the juice quality. Food Bioproc Technol. 2019;12(12):1–11. https://doi.org/10.1007/s11947-019-02292-0.
Qaid S, Zait M, El Kacemi K, El Midaoui A, El Hajjil H, Taky M. Ultrafiltration for clarification of valencia orange juice: comparison of two flat sheet membranes on quality of juice production. JMES. 2017;8(4):1186–94.
Loizzo MR, Sicari V, Tundis R, Leporini M, Falco T, Calabrò V. The influence of ultrafiltration of Citrus limon L. Burm. cv femminello comune juice on its chemical composition and antioxidant and hypoglycemic properties. Antioxidants. 2019;8(1):23. https://doi.org/10.3390/2Fantiox8010023.
Abdullah S, Karmakar S, Mishra S, et al. Ultrafiltration of cashew apple juice using hollow fibers for shelf life extension: process optimization, flux modelling and storage study. J Food Meas Charact. 2023;17(1):2182–92. https://doi.org/10.1007/s11694-022-01790-8.
Cai M, Xie C, Lv Y, Yang K, Sun P. Changes in physicochemical profiles and quality of apple juice treated by ultrafiltration and during its storage. Food Sci Nutr. 2020;8(6):2913–9. https://doi.org/10.1002/2Ffsn3.1593.
Panigrahi C, Mondal M, Karmakar S, Mishra HN, De S. Shelf life extension of sugarcane juice by cross flow hollow fibre ultrafiltration. J Food Eng. 2020;274:109880. https://doi.org/10.1016/j.jfoodeng.2019.109880.
Baranska A, Kot A, Samborska K. Ultrafiltration as a method to obtain sugar reduced cloudy juices a research on juice’s properties. Zesz Probl Postepow Nauk Roln. 2020;600(1):3–11. https://doi.org/10.2263/ZPPNR.2020.600.1.
Li F, Yan H, Li W, Zhao J, Ming J. A comparative study of the effects of ultrafiltration membranes and storage on phytochemical and color properties of mulberry juice. J Food Sci. 2019;84(12):3565–72. https://doi.org/10.1111/1750-3841.14935.
Doan NT, Lai QD, Nguyen HD, Hiroshi N. Enrichment of lycopene in watermelon juice by ultrafiltration: technical assessment and fouling analysis. Int J Food Sci Technol. 2023;58(12):6285–95. https://doi.org/10.1111/ijfs.16732.
Basak S. The potential of pulsed magnetic field to achieve microbial inactivation and enzymatic stability in foods: A concise critical review. Fut Foods. 2023;7(2):100230. https://doi.org/10.1016/j.fufo.2023.100230.
Qian J, Yan G, Huo S, Dai C, Ma H, Kan J. Effects of pulsed magnetic field on microbial and enzymic inactivation and quality attributes of orange juice. J Food Process Preserv. 2021;45(6):e15533. https://doi.org/10.1111/jfpp.15533.
Lin L, Wang X, Cui H. Synergistic efficacy of pulsed magnetic fields and Litseacubeba essential oil treatment against Escherichia coli O157: H7 in vegetable juices. Food Control. 2019;106(3):106686. https://doi.org/10.1016/j.foodcont.2019.06.012.
Lin L, Wang X, He R, Cui H. Action mechanism of pulsed magnetic field against E. coli O157: H7 and its application in vegetable juice. Food Control. 2019;95:150–6. https://doi.org/10.1016/j.foodcont.2018.08.011.
Luo X, Ma H, Gao M. Sterilization test colostrums by pulse magnetic field. China Dairy Ind. 2004;32(8):22–3.
Guo D, Xiao K. Non-thermal sterilization of milk by intense pulsed magneic field. Food Res Dev Chin. 2010;31(7):42–5.
Qian J, Chen S, Huo S, et al. Impact of pulsed magnetic field treatment on enzymatic inactivation and quality of cloudy apple juice. J Food Sci Technol. 2021;58:2982–91. https://doi.org/10.1007/s13197-020-04801-y.
Allai FM, Azad ZAA, Mir NA, Gul K. Recent advances in non-thermal processing technologies for enhancing shelf life and improving food safety. Appl Food Res. 2023;3(3):100258. https://doi.org/10.1016/j.afres.2022.100258.
Hu Y, Wu W. Application of membrane filtration to cold sterilization of drinks and establishment of aseptic workshop. Food Environ Virol. 2023;15(2):89–106. https://doi.org/10.1007/s12560-023-09551-6.
Acknowledgements
The authors are grateful to the Management of Vellore Institute of Technology, Tamil Nadu, India for providing the facilities to carry out this work.
Funding
The authors have not received funding for carrying out this work.
Author information
Authors and Affiliations
Contributions
JGC. Conceptualisation, writing, and reviewing. SR. Literature review and writing. AM. Reviewing. VKS. Reviewing.
Corresponding author
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Jeevitha, G.C., Saravanan, R., Mittal, A. et al. Non-thermal processing as a preservation tool for health-promoting beverages. Discov Food 3, 26 (2023). https://doi.org/10.1007/s44187-023-00066-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s44187-023-00066-4