Abstract
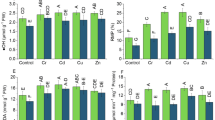
The aim is to analyze the Pb-tolerance of three commonly found tree species and determine the Pb-toxicity and impacts of Pb on growth, physiological and biochemical parameters. In the present study, we examine the response of Pb-induced stress in three commonly growing tree species i.e., Terminalia arjuna, Cassia fistula, and Pongamia pinnata based on various characteristics at an interval of 90 days for 360 days. Plant seedlings were exposed to 0 (control), 200, 450, and 600 mg of Pb/kg dry soil. Our results showed that Pb significantly reduced growth, photosynthetic pigments, and physiological parameters in all studied species. These reductions were highest in P. pinnata followed by C. fistula, and T. arjuna. Besides, Pb-induced toxicity caused many fold rise in biochemicals such as malondialdehyde, proline, phenolics, thiols, and others. These elevations were highest in T. arjuna followed by C. fistula and P. pinnata. Further, it was also observed that with an increase in the number of days of year (DOY) of Pb exposure, plants showed a reduction in physiological parameters but an increase in growth and biochemical parameters in all studied species. Furthermore, the physiological parameters showed a negative and the biochemical parameters showed a positive correlation with both DOY and treatments, while the growth parameters showed a negative correlation with treatments and a positive correlation with DOY. Hence, we can conclude that the three selected species are promising candidates to be recommended for plantation in Pb-polluted urban areas due to their Pb-tolerance. Overall, the study provides a better understanding of the impacts of Pb on tree species in the long-term and in assessing their potential for sustainable plantations in urban areas.
Highlights
• Pb reduced growth, and affected physiology and biochemistry in all studied species.
• P. pinnata was most affected followed by C. fistula and T. arjuna.
• Physiology was diminished with the number of days of Pb exposure and treatments.
• Physiology had negative and biochemistry had positive correlation with Pb exposure.
• Growth was decreased due to Pb exposure but increased with number of days.






Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Abrol E, Vyas D, Koul S (2012) Metabolic shift from secondary metabolite production to induction of anti-oxidative enzymes during NaCl stress in Swertia chirata Buch.-Ham. Acta Physiol Plantarum 34(2):541–546. https://doi.org/10.1007/s11738-011-0851-4
Ahmad P, Jaleel CA, Azooz MM, Nabi G (2009) Generation of ROS and non-enzymatic antioxidants during abiotic stress in plants. Botany Res Int 2(1):11–20
Ahmad I, Gul I, Irum S, Manzoor M, Arshad M (2022) Accumulation of heavy metals in wild plants collected from the industrial sites - potential for phytoremediation. Int J Environ Sci Tech 1–12. https://doi.org/10.1007/s13762-022-04340-3
Ainsworth AE, Gillespie MK (2007) Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat Protoc 2:875–877. https://doi.org/10.1038/nprot.2007.102
Arnon ID (1949) Copper enzymes in isolated chloroplasts polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15. https://doi.org/10.1104/pp.24.1.1
Ashraf U, Kanu AS, Deng Q, Mo Z, Pan S, Tian H, Tang X (2017) Lead (Pb) toxicity; physio-biochemical mechanisms, grain yield, quality, and Pb distribution proportions in scented rice. Front Plant Sci 8:259. https://doi.org/10.3389/fpls.2017.00259
Awashthi SK (2000) Prevention of Food Adulteration Act no 37 of 1954. Central and State Rules as Amended for 1999, 3rd edn. Ashoka Law House, New Delhi
Baek SA, Han T, Ahn SK, Kang H, Cho MR, Lee SC, Im KH (2012) Effects of heavy metals on plant growths and pigment contents in Arabidopsis thaliana. Plant Pathol J 28(4):446–452. https://doi.org/10.5423/PPJ.NT.01.2012.0006
Baldi A, Cecchi S, Grassi C, Zanchi CA, Orlandini S, Napoli M (2021) Lead bioaccumulation and translocation in herbaceous plants grown in urban and peri-urban soil and the potential human health risk. Agronomy 11(12):2444. https://doi.org/10.3390/agronomy11122444
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/bf00018060
Białońska D, Zobel AM, Kuraś M, Tykarska T, Sawicka-Kapusta K (2007) Phenolic compounds and cell structure in bilberry leaves affected by emissions from a Zn–Pb smelter. Water Air Soil Pollut 181(1):123–133. https://doi.org/10.1007/s11270-006-9284-x
Bidalia A, Okram Z, Hanief M, Rao KS (2018) Assessment of tolerances in Mitragyna parvifolia (Roxb.) Korth. and Syzygium cumini Keels. seedlings to waterlogging. Photosynthetica 56(2):707–717. https://doi.org/10.1007/s11099-017-0724-1
Björkman O, Demmig B (1987) Photon yield of O2 evolution and chlorophyll fluorescence characteristics at 77 K among vascular plants of diverse origins. Planta 170(4):489–504. https://doi.org/10.1007/BF00402983
Bond BJ (2000) Age-related changes in photosynthesis of woody plants. Trends Plant Sci 5(8):349–353. https://doi.org/10.1016/S1360-1385(00)01691-5
Cheeseman JM (2006) Hydrogen peroxide concentrations in leaves under natural conditions. J Exp Bot 57(10):2435–2444. https://doi.org/10.1093/jxb/erl004
Chen L, Larson SL, Ballard JH, Ma Y, Zhang Q, Li J, Wu L, Arslan Z, Han FX (2019) Laboratory spiking process of soil with various uranium and other heavy metals. MethodsX 6:734–739. https://doi.org/10.1016/j.mex.2019.03.026
de Souza SCR, de Andrade SAL, de Souza LA, Schiavinato MA (2012) Lead tolerance and phytoremediation potential of Brazilian leguminous tree species at the seedling stage. J Environ Manage 110:299–307. https://doi.org/10.1016/j.jenvman.2012.06.015
Delmail D, Labrousse P, Hourdin P, Larcher L, Moesch C, Botineau M (2011) Physiological, anatomical and phenotypical effects of a cadmium stress in different-aged chlorophyllian organs of Myriophyllum alterniflorum DC (Haloragaceae). Environ Exp Bot 72(2):174–181. https://doi.org/10.1016/j.envexpbot.2011.03.004
Dezhban A, Shirvany A, Attarod P, Delshad M, Matinizadeh M, Khoshnevis M (2015) Cadmium and lead effects on chlorophyll fluorescence, chlorophyll pigments and proline of Robinia pseudoacacia. J for Res 26(2):323–329. https://doi.org/10.1007/s11676-015-0045-9
Doğru A, Çakirlar H (2020) Effects of leaf age on chlorophyll fluorescence and antioxidant enzymes activity in winter rapeseed leaves under cold acclimation conditions. Rev Bras Bot 43(1):11–20. https://doi.org/10.1007/s40415-020-00577-9
Drzewiecka K, Mleczek M, Gąsecka M, Magdziak Z, Budka A, Chadzinikolau T, Kaczmarek Z, Goliński P (2017) Copper and nickel co-treatment alters metal uptake and stress parameters of Salix purpurea× viminalis. J Plant Physiol 216:125–134. https://doi.org/10.1016/j.jplph.2017.04.020
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82(1):70–77. https://doi.org/10.1016/0003-9861(59)90090-6
FAO/WHO (2007) Codex Alimentarius Commission, Joint FAO/WHO Food Standards Programme and World Health Organization, 2007. Codex Alimentarius Commission: Procedural Manual. Food & Agriculture Org
Gill RA, Zang L, Ali B, Farooq MA, Cui P, Yang S, Ali S, Zhou W (2015) Chromium-induced physio-chemical and ultrastructural changes in four cultivars of Brassica napus L. Chemosphere 120:154–164. https://doi.org/10.1016/j.chemosphere.2014.06.029
Gillespie KM, Ainsworth EA (2007) Measurement of reduced, oxidized and total ascorbate content in plants. Nat Protoc 2(4):871–874. https://doi.org/10.1038/nprot.2007.101
Greenwood MS, Hopper CA, Hutchison KW (1989) Maturation in larch: I. Effect of age on shoot growth, foliar characteristics, and DNA methylation. Plant Physiol 90(2):406–412. https://doi.org/10.1104/pp.90.2.406
Gul I, Manzoor M, Hashim N, Shah GM, Waani SPT, Shahid M, Antoniadis V, Rinklebe J, Arshad M (2021) Challenges in microbially and chelate-assisted phytoextraction of cadmium and lead–A review. Environmen Pollut 287:117667. https://doi.org/10.1016/j.envpol.2021.117667
Gul I, Manzoor M, Kallerhoff J, Arshad M (2020) Enhanced phytoremediation of lead by soil applied organic and inorganic amendments: Pb phytoavailability, accumulation and metal recovery. Chemosphere 258:127405. https://doi.org/10.1016/j.chemosphere.2020.127405
Hatamian M, Nejad AR, Kafi M, Souri MK, Shahbazi K (2019) Growth characteristics of ornamental Judas tree (Cercis siliquastrum L.) seedling under different concentrations of lead and cadmium in irrigation water. Acta Scientiarum Polonorum Hortorum Cultus 18(2): 87–96. https://doi.org/10.24326/asphc.2019.2.8
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57:1332–1334. https://doi.org/10.1139/b79-163
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611. https://doi.org/10.1007/s004250050524
Hou X, Han H, Cai L, Liu A, Ma X, Zhou C, Wang G, Meng F (2018) Pb stress effects on leaf chlorophyll fluorescence, antioxidative enzyme activities, and organic acid contents of Pogonatherum crinitum seedlings. Flora 240:82–88. https://doi.org/10.1016/j.flora.2018.01.006
John R, Ahmad P, Gadgil K, Sharma S (2009) Heavy metal toxicity: Effect on plant growth, biochemical parameters and metal accumulation by Brassica juncea L. Int J Plant Prod 3(3):65–76
Kaur G, Singh HP, Batish DR, Kohli RK (2012) A time course assessment of changes in reactive oxygen species generation and antioxidant defense in hydroponically grown wheat in response to lead ions (Pb2+). Protoplasma 249(4):1091–1100. https://doi.org/10.1007/s00709-011-0353-7
Khan M, Al Azzawi TNI, Imran M, Hussain A, Mun BG, Pande A, Yun BW (2021) Effects of lead (Pb)-induced oxidative stress on morphological and physio-biochemical properties of rice. Biocell 45(5): 1413-1423. https://doi.org/10.32604/biocell.2021.015954
Kinuthia GK, Ngure V, Kamau L (2021) Urban mosquitoes and filamentous green algae: their biomonitoring role in heavy metal pollution in open drainage channels in Nairobi industrial area. Kenya BMC Ecol Evol 21(1):1–15. https://doi.org/10.1186/s12862-021-01913-7
Kopittke PM, Blamey FPC, Asher CJ, Menzies NW (2010) Trace metal phytotoxicity in solution culture: a review. J Exp Bot 61(4):945–954. https://doi.org/10.1093/jxb/erp385
Ma T, Christie P, Teng Y, Luo Y (2013) Rape (Brassica chinensis L.) seed germination, seedling growth, and physiology in soil polluted with di-n-butyl phthalate and bis (2-ethylhexyl) phthalate. Environ Sci Poll Res 20(8):5289–5298. https://doi.org/10.1007/s11356-013-1520-5
Mahdavian K, Ghaderian SM, Schat H (2016) Pb accumulation, Pb tolerance, antioxidants, thiols, and organic acids in metallicolous and non-metallicolous Peganum harmala L. under Pb exposure. Environ Exp Bot 126:21–31. https://doi.org/10.1016/j.envexpbot.2016.01.010
Malar S, Manikandan R, Favas PJ, Sahi SV, Venkatachalam P (2014) Effect of lead on phytotoxicity, growth, biochemical alterations and its role on genomic template stability in Sesbania grandiflora: a potential plant for phytoremediation. Ecotoxicol Environ Saf 108:249–257. https://doi.org/10.1016/j.ecoenv.2014.05.018
Mallick N, Mohn FH (2003) Use of chlorophyll fluorescence in metal-stress research: a case study with the green microalga Scenedesmus. Ecotoxicol Environ Saf 55(1):64–69. https://doi.org/10.1016/S0147-6513(02)00122-7
Manzoor M, Gul I, Manzoor A, Kallerhoff J, Arshad M (2021) Optimization of integrated phytoremediation system (IPS) for enhanced lead removal and restoration of soil microbial activities. Chemosphere 277:130243. https://doi.org/10.1016/j.chemosphere.2021.130243
Nadgórska-Socha A, Kafel A, Kandziora-Ciupa M, Gospodarek J, Zawisza-Raszka A (2013) Accumulation of heavy metals and antioxidant responses in Vicia faba plants grown on monometallic contaminated soil. Environ Sci Poll Res 20(2):1124–1134. https://doi.org/10.1007/s11356-012-1191-7
Pal R, Kaur R, Rajwar D, Narayan Rai JP (2019) Induction of non-protein thiols and phytochelatins by cadmium in Eichhornia crassipes. Int J Phytoremediation 21(8):790–798. https://doi.org/10.1080/15226514.2019.1566881
Patel K, Tripathi I, Chaurasia M, Rao KS (2021a) Phytoremediation: Status and Outlook. In: Singh P, Singh R, Singh VK, Bhadouria R (eds) Pollutants and Water Management: Resources, Strategies and Scarcity. John Wiley & Sons Ltd., New York, pp 67–94. https://doi.org/10.1002/9781119693635.ch4
Patel K, Bidalia A, Tripathi I, Gupta Y, Arora P, Rao KS (2021b) Effect of heat stress on wild type and A7a knockout mutant Arabidopsis thaliana plants. Vegetos 35:168–178. https://doi.org/10.1007/s42535-021-00272-4
Patel K, Chaurasia M, Rao KS (2022) Heavy metal accumulation in leaves of selected plant species in urban areas of Delhi. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-022-24157-4
Prasad TK, Anderson MD, Martin BA, Stewart CR (1994) Evidence for chilling-induced oxidative stress in maize seedlings and a regulatory role for hydrogen peroxide. Plant Cell 6:65–74. https://doi.org/10.1105/tpc.6.1.65
Pulavarty A, Kukde S, Shinde VM, Sarangi BK (2016) Morphological, physiological and biochemical adaptations of Eucalyptus citriodora seedlings under NaCl stress in hydroponic conditions. Acta Physiol Plantarum 38(1):1–12. https://doi.org/10.1007/s11738-015-2042-1
Rastgoo L, Alemzadeh A (2011) Biochemical responses of Gouan (‘Aeluropus littoralis’) to heavy metals stress. Aust J Crop Sci 5(4):375–383. https://doi.org/10.3316/informit.281334528451448
Rebbeck J, Jensen KF, Greenwood MS (1993) Ozone effects on grafted mature and juvenile red spruce: photosynthesis, stomatal conductance, and chlorophyll concentration. Can J for Res 23(3):450–456. https://doi.org/10.1139/x93-063
Rossato LV, Nicoloso FT, Farias JG, Cargnelluti D, Tabaldi LA, Antes FG, Dressler VL, Morsch VM, Schetinger MRC (2012) Effects of lead on the growth, lead accumulation and physiological responses of Pluchea sagittalis. Ecotoxicology 21(1):111–123. https://doi.org/10.1007/s10646-011-0771-5
Sengar RS, Gautam M, Sengar RS, Garg SK, Sengar K, Chaudhary R (2008) Lead stress effects on physiobiochemical activities of higher plants. Rev Environ Contam Toxicol 196:73–93. https://doi.org/10.1007/978-0-387-78444-1_3
Seth CS, Misra V, Singh RR, Zolla L (2011) EDTA-enhanced lead phytoremediation in sunflower (Helianthus annuus L.) hydroponic culture. Plant Soil 347(1):231–242. https://doi.org/10.1007/s11104-011-0841-8
Shin YK, Bhandari SR, Jo JS, Song JW, Lee JG (2021) Effect of drought stress on chlorophyll fluorescence parameters, phytochemical contents, and antioxidant activities in lettuce seedlings. Horticulturae 7(8):238. https://doi.org/10.3390/horticulturae7080238
Shu X, Yin L, Zhang Q, Wang W (2012) Effect of Pb toxicity on leaf growth, antioxidant enzyme activities, and photosynthesis in cuttings and seedlings of Jatropha curcas L. Environ Sci Pollut Res 19(3):893–902. https://doi.org/10.1007/s11356-011-0625-y
Sidhu GPS, Singh HP, Batish DR, Kohli RK (2016) Effect of lead on oxidative status, antioxidative response and metal accumulation in Coronopus didymus. Plant Physiol Biochem 105:290–296. https://doi.org/10.1016/j.plaphy.2016.05.019
Singh S, Parihar P, Singh R, Singh VP, Prasad SM (2016) Heavy metal tolerance in plants: role of transcriptomics, proteomics, metabolomics, and ionomics. Front Plant Sci 6:1143. https://doi.org/10.3389/fpls.2015.01143
Suryawanshi PV, Rajaram BS, Bhanarkar AD, Chalapati Rao CV (2016) Determining heavy metal contamination of road dust in Delhi, India. Atmósfera, 29(3):221–234. https://doi.org/10.20937/ATM.2016.29.03.04
Sung DY, Guy CL (2003) Physiological and molecular assessment of altered expression of Hsc70-1 in Arabidopsis: Evidence for pleiotropic consequences. Plant Physiol 132:979–987. https://doi.org/10.1104/pp.102.019398
Yang N, Zhou FR, Wang JX (2016) Eco-toxicological effects of two kinds of lead compounds on forest tree seed in alkaline soil. Environ Monit Assess 188(3):1–9. https://doi.org/10.1007/s10661-016-5198-2
Zhou J, Zhang Z, Zhang Y, Wei Y, Jiang Z (2018) Effects of lead stress on the growth, physiology, and cellular structure of privet seedlings. PLoS ONE 13(3):0191139. https://doi.org/10.1371/journal.pone.0191139
Acknowledgements
The authors express their sincere gratitude to University Grant Commission, New Delhi for financial support in the form of Junior research fellowship to Kajal Patel (reference ID-322696). We thank Dr. Rahul Bhadouria for the help with statistical analysis.
Funding
The study was funded by University Grant Commission, New Delhi (Government of India) in the form of CSIR-UGC JRF (Ref no. 322696) fellowship granted to Kajal Patel.
Author information
Authors and Affiliations
Contributions
KSR and KP contributed to the study’s conceptualization and design. Material preparation, data collection, and analysis were performed by KP and MC. The final drafting of the manuscript was done by KP and KSR.
Corresponding author
Ethics declarations
Ethics Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Author Credit Statement
KSR and KP contributed to the study’s conceptualization and design. Material preparation, data collection, and analysis were performed by KP and MC. The final drafting of the manuscript was done by KP and KSR.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, K., Chaurasia, M. & Rao, K.S. Impacts of Pb-Induced Oxidative Stress on Morphological, Physiological and Biochemical Properties of Tree Species. Environ. Process. 9, 60 (2022). https://doi.org/10.1007/s40710-022-00616-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40710-022-00616-5