Abstract
Purpose of Review
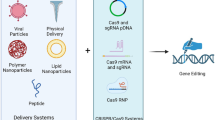
This review will highlight how gene editing technology using clustered regularly interspaced short palindromic repeats (CRISPR) and CRISPR-associated protein 9 (Cas9) has revolutionized the xenotransplantation field, leading to the first pig-to-human kidney and heart xenotransplants.
Recent Findings
CRISPR/Cas9 gene editing technology has significantly accelerated the development of multi-gene modified pigs to address the major immunological and physiological incompatibilities between pigs and humans. These gene edits include the knockout (KO) of the three porcine-specific glycan epitopes responsible for hyperacute rejection and human transgene expression targeting the coagulation and complement pathways. CRISPR/Cas9 genetic editing has also addressed a critical concern for the potential for cross-species transmission of porcine endogenous retroviruses (PERVs) by allowing the successful generation of pigs with genomically inactivated PERVs to eliminate the risk of viral transmission.
Summary
CRISPR/Cas9 multi-gene edited pigs are likely to be used in the first human clinical xenotransplant trials. While genetic modifications will help protect pig xenografts from innate immune responses, genetic engineering alone will not be sufficient to prevent chronic rejection, given the overwhelming number of possible xenoantigens that can trigger adaptive immune responses and antibody-mediated rejection. Additional immunomodulatory strategies, such as targeted immunosuppression or tolerance induction, will be required for long-term survival of porcine xenografts.
Similar content being viewed by others
Abbreviations
- 10-GE:
-
10 Gene edited pig
- β4GalNT2 :
-
Gene encoding β-1,4-N-acetyl-galactosaminyltransferase 2
- Cas:
-
CRISPR-associated protein
- CMAH :
-
Gene encoding cytidine monophosphate-N-acetylneuraminic acid hydroxylase
- CRISPR:
-
Clustered regularly interspaced short palindromic repeats
- DAF:
-
Decay-accelerating factor
- DSB:
-
Double-stranded DNA break
- ECMO:
-
Extracorporeal membrane oxygenation
- EPCR:
-
Endothelial cell protein C receptor
- GAL:
-
Galactose-α-1,3-galactose glycan
- GGTA1 :
-
Gene encoding α-1,3-galactosyltransferase
- GHR:
-
Growth hormone receptor
- HDR:
-
Homology-directed repair
- HO1:
-
Hemoxygenase-1
- KO:
-
Knockout
- Neu5Gc:
-
N-glycolylneuraminic acid glycan
- NHEJ:
-
Nonhomologous end-joining
- NHP:
-
Non-human primate
- NYU:
-
New York University
- PAM:
-
Protospacer adjacent motif
- PERV:
-
Porcine endogenous retrovirus
- POD:
-
Post-operative day
- RVD:
-
Repeat-variable di-residue
- SDa:
-
Xenoantigen produced by β-1,4-N-acetyl-galactosaminyltransferase 2
- sgRNA:
-
Single guide RNA
- TALEN:
-
Transcription activator-like effector nuclease
- TBM:
-
Thrombomodulin
- UAB:
-
University of Alabama
- UMMC:
-
University of Maryland Medical Center
- ZFN:
-
Zinc finger nuclease
References
Papers of particular interest, published recently, have been highlighted as: • Of importance
Israni AK. OPTN/SRTR 2020 annual data report: introduction. Am J Transplant. 2022;22(Suppl 2):11–20. https://doi.org/10.1111/ajt.16974.
Klymiuk N, Aigner B, Brem G, Wolf E. Genetic modification of pigs as organ donors for xenotransplantation. Mol Reprod Dev. 2010. https://doi.org/10.1002/mrd.21127.
Galili U, Shohet SB, Kobrin E, Stults CL, Macher BA. Man, apes, and Old World monkeys differ from other mammals in the expression of alpha-galactosyl epitopes on nucleated cells. J Biol Chem. 1988. https://www.ncbi.nlm.nih.gov/pubmed/2460463. Accessed 21 July 2022.
Salama A, Evanno G, Harb J, Soulillou JP. Potential deleterious role of anti-Neu5Gc antibodies in xenotransplantation. Xenotransplantation. 2015. https://doi.org/10.1111/xen.12142.
Byrne G, Ahmad-Villiers S, Du Z, McGregor C. B4GALNT2 and xenotransplantation: a newly appreciated xenogeneic antigen. Xenotransplantation. 2018. https://doi.org/10.1111/xen.12394.
Sykes M, Sachs DH. Transplanting organs from pigs to humans. Sci Immunol. 2019. https://www.science.org/doi/10.1126/sciimmunol.aau6298. Accessed 21 July 2022.
Montgomery RA, Stern JM, Lonze BE, Tatapudi VS, Mangiola M, Wu M, et al. Results of two cases of pig-to-human kidney xenotransplantation. N Engl J Med. 2022. https://www.nejm.org/doi/full/10.1056/NEJMoa2120238. Accessed 21 July 2022.
Porrett PM, Orandi BJ, Kumar V, Houp J, Anderson D, Cozette Killian A, et al. First clinical-grade porcine kidney xenotransplant using a human decedent model. Am J Transplant. 2022. https://doi.org/10.1111/ajt.16930.
Griffith BP, Goerlich CE, Singh AK, Rothblatt M, Lau CL, Shah A, et al. Genetically modified porcine-to-human cardiac xenotransplantation. N Engl J Med. 2022. https://www.nejm.org/doi/10.1056/NEJMoa2201422. Accessed 21 July 2022.
Gupta RM, Musunuru K. Expanding the genetic editing tool kit: ZFNs, TALENs, and CRISPR-Cas9. J Clin Invest. 2014. https://doi.org/10.1172/JCI72992.
Kim YG, Cha J, Chandrasegaran S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proc Natl Acad Sci. 1996. https://doi.org/10.1073/pnas.93.3.1156.
Bibikova M, Beumer K, Trautman JK, Carroll D. Enhancing gene targeting with designed zinc finger nucleases. Science. 2003. https://doi.org/10.1126/science.1079512.
Porteus MH, Baltimore D. Chimeric nucleases stimulate gene targeting in human cells. Science. 2003. https://doi.org/10.1126/science.1078395.
Orlando SJ, Santiago Y, DeKelver RC, Freyvert Y, Boydston EA, Moehle EA, et al. Zinc-finger nuclease-driven targeted integration into mammalian genomes using donors with limited chromosomal homology. Nucleic Acids Res. 2010. https://doi.org/10.1093/nar/gkq512.
Ramirez CL, Foley JE, Wright DA, Muller-Lerch F, Rahman SH, Cornu TI, et al. Unexpected failure rates for modular assembly of engineered zinc fingers. Nat Methods. 2008. https://doi.org/10.1038/nmeth0508-374.
Boch J, Scholze H, Schornack S, Landgraf A, Hahn S, Kay S, et al. Breaking the code of DNA binding specificity of TAL-type III effectors. Science. 2009. https://doi.org/10.1126/science.1178811.
Moscou MJ, Bogdanove AJ. A simple cipher governs DNA recognition by TAL effectors. Science. 2009. https://doi.org/10.1126/science.1178817.
Valton J, Dupuy A, Daboussi F, Thomas S, Marechal A, Macmaster R, et al. Overcoming transcription activator-like effector (TALE) DNA binding domain sensitivity to cytosine methylation. J Biol Chem. 2012. https://doi.org/10.1074/jbc.C112.408864.
Chen L, Tang L, Xiang H, Jin L, Li Q, Dong Y, et al. Advances in genome editing technology and its promising application in evolutionary and ecological studies. Gigascience. 2014. https://doi.org/10.1186/2047-217X-3-24.
Ishino Y, Shinagawa H, Makino K, Amemura M, Nakata A. Nucleotide sequence of the iap gene, responsible for alkaline phosphatase isozyme conversion in Escherichia coli, and identification of the gene product. J Bacteriol. 1987. https://doi.org/10.1128/jb.169.12.5429-5433.1987.
Jansen R, Embden JD, Gaastra W, Schouls LM. Identification of genes that are associated with DNA repeats in prokaryotes. Mol Microbiol. 2002. https://doi.org/10.1046/j.1365-2958.2002.02839.x.
Barrangou R, Fremaux C, Deveau H, Richards M, Boyaval P, Moineau S, et al. CRISPR provides acquired resistance against viruses in prokaryotes. Science. 2007. https://doi.org/10.1126/science.1138140.
Brouns SJ, Jore MM, Lundgren M, Westra ER, Slijkhuis RJ, Snijders AP, et al. Small CRISPR RNAs guide antiviral defense in prokaryotes. Science. 2008. https://doi.org/10.1126/science.1159689.
Garneau JE, Dupuis ME, Villion M, Romero DA, Barrangou R, Boyaval P, et al. The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature. 2010. https://doi.org/10.1038/nature09523.
Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012. https://doi.org/10.1126/science.1225829.
Ledford H, Callaway E. Pioneers of revolutionary CRISPR gene editing win chemistry Nobel. Nature. 2020. https://doi.org/10.1038/d41586-020-02765-9.
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 2013. https://doi.org/10.1126/science.1231143.
Mali P, Yang L, Esvelt KM, Aach J, Guell M, DiCarlo JE, et al. RNA-guided human genome engineering via Cas9. Science. 2013. https://doi.org/10.1126/science.1232033.
Cho SW, Kim S, Kim JM, Kim JS. Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nat Biotechnol. 2013. https://doi.org/10.1038/nbt.2507.
Hou Z, Zhang Y, Propson NE, Howden SE, Chu LF, Sontheimer EJ, et al. Efficient genome engineering in human pluripotent stem cells using Cas9 from Neisseria meningitidis. Proc Natl Acad Sci. 2013. https://doi.org/10.1073/pnas.1313587110.
Esvelt KM, Mali P, Braff JL, Moosburner M, Yaung SJ, Church GM. Orthogonal Cas9 proteins for RNA-guided gene regulation and editing. Nat Methods. 2013. https://doi.org/10.1038/nmeth.2681.
Tsai SQ, Wyvekens N, Khayter C, Foden JA, Thapar V, Reyon D, et al. Dimeric CRISPR RNA-guided FokI nucleases for highly specific genome editing. Nat Biotechnol. 2014. https://doi.org/10.1038/nbt.2908.
Guilinger JP, Thompson DB, Liu DR. Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat Biotechnol. 2014. https://doi.org/10.1038/nbt.2909.
Komor AC, Kim YB, Packer MS, Zuris JA, Liu DR. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature. 2016. https://doi.org/10.1038/nature17946.
Anzalone AV, Randolph PB, Davis JR, Sousa AA, Koblan LW, Levy JM, et al. Search-and-replace genome editing without double-strand breaks or donor DNA. Nature. 2019. https://doi.org/10.1038/s41586-019-1711-4.
Wang H, Yang H, Shivalila CS, Dawlaty MM, Cheng AW, Zhang F, et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell. 2013. https://doi.org/10.1016/j.cell.2013.04.025.
Shalem O, Sanjana NE, Hartenian E, Shi X, Scott DA, Mikkelson T, et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science. 2014. https://doi.org/10.1126/science.1247005.
Koike-Yusa H, Li Y, Tan EP, Velasco-Herrera Mdel C, Yusa K. Genome-wide recessive genetic screening in mammalian cells with a lentiviral CRISPR-guide RNA library. Nat Biotechnol. 2014. https://doi.org/10.1038/nbt.2800.
Zhou Y, Zhu S, Cai C, Yuan P, Li C, Huang Y, et al. High-throughput screening of a CRISPR/Cas9 library for functional genomics in human cells. Nature. 2014. https://doi.org/10.1038/nature13166.
Li W, Teng F, Li T, Zhou Q. Simultaneous generation and germline transmission of multiple gene mutations in rat using CRISPR-Cas systems. Nat Biotechnol. 2013. https://doi.org/10.1038/nbt.2652.
Fu Y, Sander JD, Reyon D, Cascio VM, Joung JK. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat Biotechnol. 2014. https://doi.org/10.1038/nbt.2808.
Dai Y, Vaught TD, Boone J, Chen SH, Phelps CJ, Ball S, et al. Targeted disruption of the alpha1,3-galactosyltransferase gene in cloned pigs. Nat Biotechnol. 2002. https://doi.org/10.1038/nbt0302-251.
Phelps CJ, Koike C, Vaught TD, Boone J, Wells KD, Chen SH, et al. Production of alpha 1,3-galactosyltransferase-deficient pigs. Science. 2003. https://doi.org/10.1126/science.1078942.
Chen G, Qian H, Starzl T, Sun H, Garcia B, Wang X, et al. Acute rejection is associated with antibodies to non-Gal antigens in baboons using Gal-knockout pig kidneys. Nat Med. 2005. https://doi.org/10.1038/nm1330.
Kuwaki K, Tseng YL, Dor FJ, Shimizu A, Houser SL, Sanderson TM, et al. Heart transplantation in baboons using alpha1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med. 2005. https://doi.org/10.1038/nm1171.
Estrada JL, Martens G, Li P, Adams A, Newell KA, Ford ML, et al. Evaluation of human and non-human primate antibody binding to pig cells lacking GGTA1/CMAH/beta4GalNT2 genes. Xenotransplantation. 2015. https://doi.org/10.1111/xen.12161.
Loveland BE, Milland J, Kyriakou P, Thorley BR, Christiansen D, Lanteri MB, et al. Characterization of a CD46 transgenic pig and protection of transgenic kidneys against hyperacute rejection in non-immunosuppressed baboons. Xenotransplantation. 2004. https://doi.org/10.1046/j.1399-3089.2003.00103.x.
Azimzadeh AM, Kelishadi SS, Ezzelarab MB, Singh AK, Stoddard T, Iwase H, et al. Early graft failure of GalTKO pig organs in baboons is reduced by expression of a human complement pathway-regulatory protein. Xenotransplantation. 2015. https://doi.org/10.1111/xen.12176.
• Mohiuddin MM, Goerlich CE, Singh AK, Zhang T, Tatarov I, Lewis B, et al. Progressive genetic modifications of porcine cardiac xenografts extend survival to 9 months. Xenotransplantation. 2022. https://doi.org/10.1111/xen.12744. (GHR KO added to a multi-gene edited cardiac xenograft model resulted in significantly extended graft survival by preventing graft hypertrophy.)
Petersen B, Ramackers W, Tiede A, Lucas-Hahn A, Herrmann D, Barg-Kues B, et al. Pigs transgenic for human thrombomodulin have elevated production of activated protein C. Xenotransplantation. 2009. https://doi.org/10.1111/j.1399-3089.2009.00537.x.
Singh AK, Chan JL, DiChiacchio L, Hardy NL, Corcoran PC, Lewis BGT, et al. Cardiac xenografts show reduced survival in the absence of transgenic human thrombomodulin expression in donor pigs. Xenotransplantation. 2019. https://doi.org/10.1111/xen.12465.
Ide K, Wang H, Tahara H, Liu J, Wang X, Asahara T, et al. Role for CD47-SIRPalpha signaling in xenograft rejection by macrophages. Proc Natl Acad Sci. 2007. https://doi.org/10.1073/pnas.0609661104.
Petersen B, Ramackers W, Lucas-Hahn A, Lemme E, Hassel P, Queisser AL, et al. Transgenic expression of human heme oxygenase-1 in pigs confers resistance against xenograft rejection during ex vivo perfusion of porcine kidneys. Xenotransplantation. 2011. https://doi.org/10.1111/j.1399-3089.2011.00674.x.
Langin M, Mayr T, Reichart B, Michel S, Buchholz S, Guethoff S, et al. Consistent success in life-supporting porcine cardiac xenotransplantation. Nature. 2018. https://doi.org/10.1038/s41586-018-0765-z.
Goerlich CE, Griffith B, Hanna P, Hong SN, Ayares D, Singh AK, et al. The growth of xenotransplanted hearts can be reduced with growth hormone receptor knockout pig donors. J Thorac Cardiovasc Surg. 2021. https://doi.org/10.1016/j.jtcvs.2021.07.051.
Niu D, Wei HJ, Lin L, George H, Wang T, Lee IH, et al. Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science. 2017. https://doi.org/10.1126/science.aan4187.
• Yue Y, Xu W, Kan Y, Zhao HY, Zhou Y, Song X, et al. Extensive germline genome engineering in pigs. Nat Biomed Eng. 2021. https://doi.org/10.1038/s41551-020-00613-9. (CRISPR/Cas9 and transposon technologies were used to produce PERV KO pigs with 13 additional genetic modifications.)
Wang ZY, Burlak C, Estrada JL, Li P, Tector MF, Tector AJ. Erythrocytes from GGTA1/CMAH knockout pigs: implications for xenotransfusion and testing in non-human primates. Xenotransplantation. 2014. https://doi.org/10.1111/xen.12106.
• Ariyoshi Y, Takeuchi K, Pomposelli T, Ekanayake-Alper DK, Shimizu A, Boyd L, et al. Antibody reactivity with new antigens revealed in multi-transgenic triple knockout pigs may cause early loss of pig kidneys in baboons. Xenotransplantation. 2021. https://doi.org/10.1111/xen.12642. (Baboons transplanted with triple glycan KO pig kidneys and vascularized thymic grafts demonstrated unmasking of neoantigens that were associated with accelerated graft rejection.)
Yamada K, Shimizu A, Ierino FL, Utsugi R, Barth RN, Esnaola N, et al. Thymic transplantation in miniature swine. I. Development and function of the “thymokidney.” Transplantation. 1999. https://doi.org/10.1097/00007890-199912150-00011.
Griesemer AD, Hirakata A, Shimizu A, Moran S, Tena A, Iwaki H, et al. Results of gal-knockout porcine thymokidney xenografts. Am J Transplant. 2009. https://doi.org/10.1111/j.1600-6143.2009.02849.x.
• Takeuchi K, Ariyoshi Y, Shimizu A, Okumura Y, Cara-Fuentes G, Garcia GE, et al. Expression of human CD47 in pig glomeruli prevents proteinuria and prolongs graft survival following pig-to-baboon xenotransplantation. Xenotransplantation. 2021. https://doi.org/10.1111/xen.12708. (Podocyte-specific expression of human CD47 in pig kidney xenografts prevented development of proteinuria, but concurrent expression of human CD47 in pig kidney xenografts tubules was associated with a destructive inflammatory process.)
Reyes LM, Estrada JL, Wang ZY, Blosser RJ, Smith RF, Sidner RA, et al. Creating class I MHC-null pigs using guide RNA and the Cas9 endonuclease. J Immunol. 2014. https://doi.org/10.4049/jimmunol.1402059.
Fischer K, Rieblinger B, Hein R, Sfriso R, Zuber J, Fischer A, et al. Viable pigs after simultaneous inactivation of porcine MHC class I and three xenoreactive antigen genes GGTA1, CMAH and B4GALNT2. Xenotransplantation. 2020. https://doi.org/10.1111/xen.12560.
Yamada K, Ariyoshi Y, Pomposelli T, Sekijima M. Co-transplantation of vascularized thymic graft with kidney in pig-to-nonhuman primates for the induction of tolerance across xenogeneic barriers. Methods Mol Biol. 2020. https://doi.org/10.1007/978-1-0716-0255-3_11.
Tasaki M, Wamala I, Tena A, Villani V, Sekijima M, Pathiraja V, et al. High incidence of xenogenic bone marrow engraftment in pig-to-baboon intra-bone bone marrow transplantation. Am J Transplant. 2015. https://doi.org/10.1111/ajt.13070.
Watanabe H, Ariyoshi Y, Pomposelli T, Takeuchi K, Ekanayake-Alper DK, Boyd LK, et al. Intra-bone bone marrow transplantation from hCD47 transgenic pigs to baboons prolongs chimerism to >60 days and promotes increased porcine lung transplant survival. Xenotransplantation. 2020. https://doi.org/10.1111/xen.12552.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The author declares no competing interests.
Human and Animal Rights and Informed Consent
All reported studies/experiments with human or animal subjects performed by the authors have been previously published and complied with all applicable ethical standards (including the Helsinki Declaration and its amendments, institutional research committee standards, and international/national/institutional guidelines).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Expanding Role of Technology in Organ Transplant
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Stewart, Z.A. Xenotransplantation: The Contribution of CRISPR/Cas9 Gene Editing Technology. Curr Transpl Rep 9, 268–275 (2022). https://doi.org/10.1007/s40472-022-00380-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40472-022-00380-3