Abstract
Background
Clinical evidence supports the use of genetic counselling and BRCA1/2 testing for women at risk for hereditary breast and ovarian cancer. Currently, screen-and-treat strategies are not reimbursed in the Brazilian Unified Healthcare System (SUS). The aim of this modelling study was to evaluate the cost effectiveness of a gene-based screen-and-treat strategy for BRCA1/2 in women with a high familial risk followed by preventive interventions compared with no screening.
Methods
Adopting the SUS perspective, a Markov model with a lifelong time horizon was developed for a cohort of healthy women aged 30 years that fulfilled the criteria for BRCA1/2 testing according to the National Comprehensive Cancer Network (NCCN) guideline. For women who tested positive, preventive options included intensified surveillance, risk-reducing bilateral mastectomy and bilateral salpingo-oophorectomy. The Markov model comprised the health states ‘well’, ‘breast cancer’, ‘death’ and two post-cancer states. Outcomes were the incremental costs per quality-adjusted life-year (QALY) and the incremental costs per life-year gained (LYG). Data were mainly obtained by a literature review. Deterministic and probabilistic sensitivity analyses were performed to assess the robustness of the results.
Results
In the base case, the screen-and-treat strategy resulted in additional costs of 3515 Brazilian reais (R$) (US$1698) and a gain of 0.145 QALYs, compared with no screening. The incremental cost-effectiveness ratio (ICER) was R$24,263 (US$21,724) per QALY and R$27,258 (US$24,405) per LYG. Applying deterministic sensitivity analyses, the ICER was most sensitive to the probability of a positive test result and the discount rate. In the probabilistic sensitivity analysis, a willingness to pay of R$25,000 per QALY gained for the screen-and-treat strategy resulted in a probability of cost effectiveness of 80%.
Conclusion
Although there is no rigorous cost-effectiveness threshold in Brazil, the result of this cost-effectiveness analysis may support the inclusion of BRCA1/2 testing for women at high-risk of cancer in the SUS. The ICER calculated for the provision of genetic testing for BRCA1/2 approximates the cost-effectiveness threshold proposed by the World Health Organization (WHO) for low- and middle-income countries.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Genetic testing for BRCA1/2 for healthy women with high familiar risk results in more quality-adjusted life years at a moderately higher cost. |
This economic modelling shows that a screen-and-treat strategy for women at risk for hereditary breast and ovarian cancer might be cost-effective from the perspective of the Brazilian Unified Healthcare System (SUS). |
1 Introduction
Breast cancer is the main cause of cancer-related death in Brazilian women [1]. According to the Brazilian National Cancer Institute, in 2018, the incidence of breast cancer amounted to 56 cases per 100,000 women, resulting in 59,700 new cases annually [1]. Additionally, 6150 incident cases of ovarian cancer were diagnosed in Brazil in 2018 [1]. Although there has been a trend toward stabilization in female breast cancer mortality rates in Brazil, the state-specific mortality rates show considerable inequalities that may indicate disparities in healthcare availability [2].
The risk of breast cancer or ovarian cancer increases with the number of affected relatives in the family and the closeness of the relationship (i.e. first- or second-degree relative). Additionally, the younger a woman is diagnosed with cancer, the more likely a genetic component will be found [3]. Hereditary breast and ovarian cancer (HBOC) are mainly associated with germline mutations in the genes BRCA1 and BRCA2 (collectively named BRCA hereafter). Pathogenic variants in the BRCA genes are important predictors of breast and ovarian cancer, with a 40–80% lifetime risk of breast cancer and an 11–50% lifetime risk of ovarian cancer, respectively [4]. In case of a BRCA mutation, a non-directive counselling session should be provided to inform women of their absolute individual risk. For women who have tested positive, different preventive options are recommended. According to published evidence, risk-reducing surgery (bilateral mastectomy or bilateral salpingo-oophorectomy), chemoprevention and enhanced surveillance are effective for reducing the incidence and mortality of cancer [5, 6]. However, the decision between the available risk-reducing strategies is highly preference-sensitive [7, 8].
In order to identify high-risk women for genetic testing and counselling, the National Comprehensive Cancer Network (NCCN) guideline proposes to select women based on their personal and familial history of cancer [7]. These criteria are currently applied to reimburse genetic testing for privately insured women in Brazil [9]. Whereas private insurance offers supplementary coverage, about 70% of the population is exclusively insured within the Brazilian Unified Healthcare System (SUS). The SUS is organized according to three levels of complexity of care. In the primary level, general practitioners are responsible for basic care. If further laboratory testing and imaging is required, patients are referred to high complexity centres. Cancer care in Brazil is performed on the third level (i.e. specialized care units and hospital complexes), which is largely financed by the federal government and reimbursed according to disease-related package sums. Among the high-complexity oncology centres, only a few with research motivation offer genetic counselling and testing [10]. As a result, in Brazil there is limited access to genetic counselling and BRCA testing for women at increased familial risk.
Recently, a Markov model for screen-and-treat strategies offered to Brazilian women at risk for BRCA mutation revealed an incremental cost-effectiveness ratio of R$910 (Brazilian reais) per cancer case avoided. However, the cohort simulated in this study represented only first-degree relatives of women with ovarian cancer and aspects of health-related quality of life were not reflected [11].
The aim of this modelling study was to evaluate the costs and effects of a genetic-based screen-and-treat strategy compared with standard care. The screen-and-treat strategy will be provided to healthy women with a first- or second-degree relative affected by BRCA-associated cancer (i.e. breast, ovarian, pancreatic and prostate cancer) [7]. The prophylactic treatments considered in this model were intensified surveillance, risk-reducing bilateral mastectomy or risk-reducing bilateral salpingo-oophorectomy.
2 Methods
Using TreeAge Pro (TreeAge® ProSuite 2018), a Markov model was developed to assess the cost and effectiveness of a screen-and-treat strategy for women with a first- or second-degree relative affected by BRCA-associated cancer compared with no screening (i.e. no prophylactic treatment). To reflect long-term consequences of breast and ovarian cancer, the model had a time horizon of 70 years. Using a 1-year cycle length, the analysis was performed from the perspective of the SUS.
The selection of the model population was based on the clinical criteria for BRCA testing established by the NCCN. The NCCN guideline criteria (version 2.2017) consider several aspects of a woman’s personal and family history including different HBOC-related tumours (i.e. prostate and pancreatic cancers) (Table A1, appendix—see electronic supplementary material [ESM]). Thus, the model population reflects Brazilian women still unaffected by breast or ovarian cancer with first- or second-degree relatives who have BRCA-related cancer [7]. Due to the low incidence of breast and ovarian cancer at younger ages, all women enter the model at the age of 30 years.
2.1 Strategies for the Comparison
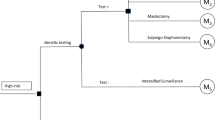
The intervention is defined as a stepwise process that starts by the identification of women at high risk of HBOC due to familial or personal history. The three main components are genetic counselling by a trained health professional (i.e. evaluation of individual risk and patient education of risk-reducing alternatives), genetic testing and different surgical/non-surgical preventive options [12]. The preventive options for women testing positive were (i) intensified surveillance, defined as yearly breast examination by physicians and alternate mammography and magnetic resonance imaging (MRI) every 6 months, (ii) salpingo-oophorectomy and (iii) bilateral mastectomy. For women choosing (ii) or (iii), it was possible to delay the preventive surgery for 5–10 years for the purpose of childbearing and other personal reasons. Women who postpone the preventive surgery were assumed to receive intensified surveillance. Women in the intervention group who test negative and women in the control group are treated in accordance with the standard breast-cancer—screening programme provided by the SUS, which does not include intensive surveillance for familial syndromes (i.e. biennial mammography between 50 and 69 years) (Fig. 1).
2.2 Model Overview
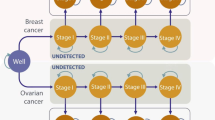
The structure of the Markov model was based on a previous publication and on consultancy with Brazilian oncologists [13, 14]. The Markov model comprised the health states ‘well’, ‘breast cancer’, ‘ovarian cancer’, ‘death’ and two-post cancer states. Women diagnosed with breast cancer were assumed to undergo either unilateral mastectomy (i.e. simple and radical) or breast-conserving surgery in the same proportion [15]. Women with contralateral breast cancer returned to the initial breast cancer state, with treatment costs and utilities assumed to be the same as for their first breast cancer. The state ‘death’ was modelled as an absorbing state. Transition from ovarian cancer to breast cancer was not included, considering the low incidence of ovarian cancer and its high rates of recurrence and mortality [16] (Fig. 2).
2.3 Input Parameters
Several systematic literature searches in Medline and BIREME (a Latin American health database) were performed to identify input data on clinical efficacy of surgical treatment options, transition probabilities and utilities. If possible, data reflecting the Brazilian context were preferred. Quality of studies was assessed following the recommendations of the Cochrane non-Randomized Studies Methods Group [17]. In addition, clinical experts were asked about the suitability of data in the specific clinical context. Table 1 summarizes all input data and data sources, whereas Table A2 (Appendix, see ESM) provides additional information on the way the literature review was conducted.
2.4 Probabilities
Data on the prevalence of BRCA variants among the at-risk population were extracted from a cohort of healthy women that fulfilled the HBOC testing criteria in Southern Brazil according to the NCCN. This reference study applied next-generation sequencing or Sanger sequencing to analyse the coding region of the BRCA genes and intron–exon junctions [18].
For women who tested BRCA positive, the probabilities of moving from the ‘well’ state to the states ‘breast cancer’ or ‘ovarian cancer’ and data on the recurrence of breast cancer were derived from a retrospective study including 7666 female mutation-carriers from the United Kingdom, Netherlands, France, Australia, New Zealand, United States and Canada [19]. For women who tested BRCA negative and for the control group, we applied the breast and ovarian cancer risk of the general Brazilian population [20,21,22]. Similarly, the probability of recurrent cancer (for both breast and ovarian) was assumed to be equal to that of the general Brazilian population [23, 24]. Mortality data on breast and ovarian cancer were obtained from a Southwest Brazilian cohort and a national registry [25], 26.
2.5 Risk Reduction Strategies
The uptake of risk-reducing surgery for our model was based on data from a prospective cohort from the United States and the United Kingdom with a sample of 1499 BRCA carriers [27]. Because randomization was not possible due the nature of the intervention, data on effectiveness of risk-reducing mastectomy (breast cancer incidence) and salpingo-oophorectomy (both breast and ovarian cancer incidence) were taken from systematic reviews from observational trials. These studies included only women who tested positive for BRCA [5, 6, 28, 29].
2.6 Utilities
In the model, utilities decreased as a result of a positive genetic test, prophylactic surgery and breast or ovarian cancer. Utilities for the prophylactic strategies were obtained from Grann et al. [30]. In that study, a Time Trade-Off instrument (TTO) was applied to 83 female Canadian BRCA carriers to reflect health states such as tested gene-positive, breast cancer and ovarian cancer. In this survey, women were asked how many years of their life expectancy they would trade to be free of these states. In accordance with assumptions of other modelling studies and based on findings from a retrospective cohort, decreased utilities following preventive surgery and a positive test result were assumed to increase in a linear manner for 5 years to regain the age-specific utility of an otherwise healthy woman carrying a mutation [13, 31, 32]. To women with contralateral breast cancer, recurrent ipsilateral breast cancer or recurrent ovarian cancer, the utilities and costs of the first year of disease were once more applied. Although metastatic breast cancer was not included as a separated health state, an additional decrease of utilities due to metastasis was reflected in a proportion (i.e. 8.4%) of patients within the breast cancer state [33]. Similarly, lower utility values due to end-stage ovarian cancer were applied to the proportion of women in the ‘ovarian cancer’ state, whose survival was expected to be < 1 year (i.e. proportional to the age-adjusted mortality applied to each cycle).
Data on utility in case of breast cancer or metastatic breast cancer were obtained from a meta-regression of studies eliciting utilities with the Standard Gamble (SG) approach [34]. It was assumed that a woman’s utility declines as a result of breast cancer and then increases linearly for 5 years to regain the age-specific utility of a post-cancer state as determined by Grann et al. [30]. Utilities for ovarian cancer were taken from a single study based on the SG approach. The utility values in that study were associated with five clusters of patients with different levels of toxicity and symptoms [35]. To ensure a consistent set of utilities, these were combined and age-adjusted using the multiplicative method [36].
2.7 Costs
Cost data were mainly expressed in Brazilian currency (Reais, R$), as suggested by the Brazilian guideline of economic evaluations [37]. Costs of preventive strategies for the year 2019 (i.e. genetic counselling, mammography, breast MRI, prophylactic mastectomy and prophylactic oophorectomy) were obtained from the Table of Procedures, Medications and Ortheses, Prostheses, and Special Materials for the National Health System (DATASUS Tabnet) [38]. The prices for genetic testing were based on commercial proposals from local distributors, which were similar to the costs currently applied in the private sector.
All other cost data were extracted from a recent analysis that used a macro costing approach to estimate the mean reimbursement per case of breast and ovarian cancer to a hospital in Sao Paulo [11]. In that study, data on costs were collected from a cohort of 100 patients during a mean follow-up of 3.3 years. The mean costs of treatment for ovarian cancer amounted to R$12,958 for the first year, and decreased from R$3960 in year 2 to R$3508 in year 5 (breast cancer: R$9120 in the first year, R$4059 to R$1278 in years 2 to 5). These costs include the costs of palliative care, adjuvant and neo-adjuvant chemotherapy, radiotherapy, diagnostic and surgical procedures. (Table 2 and Table A3 in the Appendix, see ESM) Because in this study estimates of resource consumption and costs were made for the year 2014, these were adjusted to unitary prices established by the SUS for 2019 [38]. For women in the post-cancer states, the costs associated with cancer treatment decreased yearly up to year 5. From the fifth year on, no additional costs were considered.
In order to allow comparisons with other settings, conversion to United States dollars (US$) was performed by using a web-based tool for adjusting for currency and inflation. This web tool adjusts estimates of costs for currency and/or price year using a two-stage computation. Stage one adjusts the original estimate of cost from the original price year to a target price year, using a Gross Domestic Product deflator index (‘GDPD values’). Stage two converts the price-year–adjusted cost estimate from the original currency to a target currency, using rates based on Purchasing Power Parities for GDP (‘PPP values’) [39].
2.8 Model Outcomes
The model outputs were measured as an incremental cost-effectiveness ratio (ICER) and expressed in R$ per quality-adjusted life-year (QALY) and R$ per life-year gained (LYG). In line with recommendations from the Brazilian guideline for economic evaluations, costs and benefits were discounted at 5% and a half-cycle correction was applied [37].
2.9 Model Validation and Sensitivity Analyses
In order to explore sources of uncertainty and heterogeneity, several one-way deterministic sensitivity analyses were applied. In addition, the following structural sensitivity analyses were performed: (i) women in the control group received standard care whereas 50% of women in the intervention group who tested negative for BRCA received intensive surveillance and 50% standard care; (ii) the target group was limited to women fulfilling the NCCN criteria and a pre-test probability of BRCA mutation of 10% (according to the Manchester tool) [40]; and (iii) a higher cancer risk for women who tested negative, due to family history of cancer. The Manchester tool is a manual scoring system that can be easily applied in counselling sessions to quantify the likelihood of identifying a BRCA1 or BRCA2 mutation. In order to assess how a simultaneous change of several variables affected the cost-effectiveness ratio, a probabilistic sensitivity analysis (10,000 iterations) was conducted. Gamma distributions were used for modelling cost parameters, while probabilities and utilities were assumed to be beta-distributed.
In order to validate the model, we consulted experts on the adequacy of input data and the conceptual appropriateness of the model. Technical accuracy was checked regarding data entry and potential programming errors (computerized model validation). For cross-model validation, we assessed the extent to which other models for breast cancer prevention came to different conclusions. To make our model more comparable to others, we increased the starting age (i.e. all women underwent surgery at the age of 35). The ‘Assessment of the Validation Status of Health-Economic decision models’ (AdViSHE) was used to provide structured information regarding the validation status of this model [41] (Table A5, appendix, see ESM).
3 Results
3.1 Base-Case Analysis
The screen-and-treat strategy costs R$5995 for the base-case scenario, resulting in an incremental cost of R$3515 (US$1698) compared with the no-testing strategy. Correspondingly, women that were offered the genetic screening strategy had an incremental gain of 0.145 QALYs. The ICER for the base-case analysis was R$24,263 (US$11,726) per QALY. The costs per LYG amount to R$27,258 (US$13,174). Base-case results are described in Table 3.
3.2 Sensitivity Analyses
The probability of a positive test result and the discounting rate were the variables with the largest impact on the ICER. In a scenario where the probability of a positive test result was 50% lower, the ICER increased to R$40,715/QALY (US$19,678). In contrast, if the probability of a positive test result was 50% higher, the ICER would decrease to R$17,228/QALY (US$8326). A discount rate of zero would reduce the ICER to R$3459/QALY (US$1672), while a discount rate of 10% would result in an ICER of R$101,857/QALY (US$49,230). The cost-effectiveness ratio was also sensitive to data on effectiveness of prophylactic surgical procedures. Assuming a higher impact on incidence of breast cancer (i.e. a hazard ratio [HR] of 0.01 for mastectomy and of 0.04 for salpingo-oophorectomy), the ICER would decrease by 48%. Assuming a higher impact on incidence of ovarian cancer by salpingo-oophorectomy (HR of ovarian cancer = 0.12), the ICER would drop by 20% (Table 4A and Fig. 1a Tornado Diagram, appendix [ESM]).
If all women underwent surgical prevention at the age of 35 years, the ICER would decrease by 6%. If the provision of genetic testing were restricted to women with a 10% pre-test probability or more, the ICER would decrease by 40% (R$14,467 or US$10,489 per QALY). If the risk of breast and ovarian cancer in BRCA-negative women were considered two-fold the risk of the general population, the ICER would decrease by only 2%. The ICER almost doubled in the scenario where patients in the intervention group that tested negative would be followed with intensive surveillance instead of intensive surveillance (Table 5A, appendix [see ESM]).
In the probabilistic sensitivity analysis, all 10,000 iterations fell in the northeast quadrant, indicating that the screen-and-treat strategy is certainly more expensive and generates more QALYs (Fig. 2a, appendix [see ESM]). The cost-effectiveness acceptability curve showed a probability of genetic testing being cost effective of 21%, 80% and 99% at a willingness to pay (WTP) of R$23,000, R$25,000 and R$27,000/QALY, respectively. Figure 3 shows the cost-effectiveness acceptability curve for the genetic screen-and-treat strategy provided to Brazilian women.
4 Discussion
Currently, within the Brazilian SUS, genetic testing is exclusively provided in the context of scientific research (e.g. clinical trials). Because risk-reducing strategies for BRCA carriers are clinically established and adopted by international guidelines [7, 42], the objective of this modelling study was to evaluate the costs and effects of a genetic-based screen-and-treat strategy provided to healthy women at risk for HBOC, as defined by the NCCN criteria, from the perspective of the SUS. The analysis revealed that the provision of genetic testing for BRCA in Brazilian woman with a high familial risk would result in both additional benefits and costs.
The calculated cost-effectiveness ratio in this analysis is below the cost-effectiveness threshold proposed by the World Health Organization (WHO) recommended for middle-income countries (i.e. 1 to 3 of the Gross Domestic Product [GDP] per capita) [43]. Because the ICER of our analysis is 1.04 times the GDP per capita, a genetic-based screen-and-treat strategy for Brazilian women at increased risk of cancer might be considered cost effective. Nonetheless, a reliable threshold for Brazil is yet to be defined.
The cost-effectiveness threshold from the WHO has been discredited, as have other estimates. One estimate based on per capita health expenditures and life expectancy at birth suggested a threshold for Brazil between 0.62 and 1.05 GDP per capita per QALY [44]. Using data from several low- and middle-income countries combined with several assumptions, an opportunity-cost–based threshold was proposed to lay between 0.18 and 0.71 of the GDP per capita [45]. Based on these thresholds, the genetic-based screen-and-treat strategy for Brazilian women at risk for hereditary breast and ovarian cancer would not be cost effective. However, a systematic review of health economic evaluations in Brazil showed that many interventions with an ICER above the upper limit of these two thresholds still received supporting decisions [46]. Therefore, decision making concerning health interventions in Brazil appears to be based on factors including but not limited to thresholds (e.g. budgetary impact, magnitude of clinical effectiveness, epidemiological relevance, among others).
Because data on utilities specific to the Brazilian population is not available, it is perceived that decision-making agencies in Brazil prefer to take into account additional measures of clinical benefit (e.g. life-years gained, deaths averted, or new cases of cancer averted) [47]. Therefore, the ICER calculated as cost per LYG may contribute to allocation decisions (R$27,258 per LYG).
The structure of the model had a number of strengths. First, unlike previous modelling studies, this model considered the women’s option to postpone the surgery by 5 or 10 years after genetic counselling and testing. As many women might prefer to delay a prophylactic surgery to allow for childbearing or for aesthetic or other personal reasons, the adopted structure reflects women’s options more accurately. Second, data on costs were based on a reference case (DATASUS Tabnet) that corresponds to the main components of health financing in Brazil (including geographic discrepancies). In addition, most utility data applied to the model were generated using the SG approach, resulting in homogeneous parameters on quality of life. However, utilities on prophylactic surgeries and end-stage ovarian cancer had to be based on TTO.
Finally, data on reimbursement for oncological procedures and medications in the SUS were in accordance with the disease-related package of standard reimbursement sums. Although the prices used for this analysis do not cover the costs of all available chemotherapy regimens, they were assumed to adequately reflect the additional burden for the SUS [48].
A limitation of the model is in the unknown risk of BRCA-negative tested women with a family history of cancer compared with cancer risks observed in the general population. Two studies found an increased risk only for relatives of BRCA2 [49] or BRCA1 [50] carriers, while more recent studies based on larger cohorts showed that noncarriers of either genetic mutation did not show an increased risk for HBOC [20,21,22]. Due to this uncertainty, we followed the guideline recommendation by assuming standard care for women who tested negative [7]. However, the uncertainty of the risk in women testing negative can lead to a more conservative approach by the physician where the decision between standard care and intensified surveillance is concerned. Therefore, an unknown proportion of BRCA-negative tested women may receive intensive surveillance, which would increase the cost-effectiveness ratio. Assuming a scenario of intensive surveillance for 50% of negative tested women (which would reflect a more cautious treatment attitude of physicians), this would increase the ICER to R$38,968 per QALY. A structural sensitivity analysis reflecting a higher cancer risk in BRCA-negative tested women did not affect the cost-effectiveness ratio.
A further limitation of the present model might be the lack of Brazilian data on penetrance of BRCA. Whereas the variability on penetrance was similar between the United States, the United Kingdom, Spain, Australia and New Zealand [19, 51,52,53,54,55,56,57], a cohort of Colombian mutation carriers appeared to be associated with lower penetrance [58]. Nevertheless, studies that reflect the genetic profile of the Latin American population are limited and their application to the Brazilian population is difficult. The reason is that the Hispanic influence on the Brazilian population structure is considerably lower than that on neighbour countries [59]. In order to account for the heterogeneity and variability of BRCA penetrance, the data applied to this model were selected from a retrospective international cohort. Although we cannot rule out having misjudged the risk profile of Brazilian women, several sensitivity analyses showed similar results.
In addition, the rates of uptake of prophylactic procedures used for the model could not be obtained from the Brazilian population. Although this choice of prophylactic strategies by women at risk is contingent on personal beliefs and physician recommendations, a healthcare system’s structure and access to service are the main determinants for the surgical uptake rates of different countries [60]. Given the lack of Brazilian data, we relied on data from the United Kingdom, as the structure of the healthcare system is similar to that of the Brazilian system [27]. Furthermore, a sensitivity analysis simulating a lower uptake of prophylactic surgeries (20%) than that applied to the base case resulted in only a slight change of the ICER.
A further limitation is the exclusion of a transition from ovarian cancer to breast cancer. The rationale for this omission was (i) the low incidence of ovarian cancer [25, 26] and (ii) high rates of mortality and recurrence of ovarian cancer [27]. Thus, an impact of breast cancer in women with ovarian cancer on the overall results of the model is not expected.
Among the previous Markov models that evaluated the cost effectiveness of genetic BRCA testing for breast or ovarian cancer in high-risk women, Ramos et al. concluded BRCA testing to be cost effective from the perspective of the SUS. This conclusion was drawn from the comparison of the ICER of R$908 per case of cancer prevented with the usual sums typically spent on treatment of breast and ovarian cancer in Brazil. Data on costs used for that analysis were the same as were applied to our model, while the model structure differed to some extent, compromising comparability of results. In detail, the population analysed in the model was restricted to only first-degree relatives of patients affected by ovarian cancer. Additionally, women at risk were offered surgical procedures at the age of 35 years (i.e. a delayed prophylactic procedure was not considered) and the time horizon was shorter (i.e. 40 years) [11]. Hence, the comparability of the results of our study with those of Ramos et al. might be limited. Different methodological approaches were also observed for models developed for other countries. Holland et al. assessed a cost-effectiveness evaluation of BRCA testing for US women from the societal perspective. In that model, a long-term decrease of utility due to prophylactic surgeries was assumed (i.e. 60 years) and a combined state of breast and ovarian cancer was considered. The results of that model were most sensitive to a utility decrease due to a BRCA mutation or a prophylactic mastectomy and a utility gain from a negative test result [61]. In the model designed by Müller et al., the option to undergo both procedures simultaneously was provided and a separate state for metastatic breast cancer was included [13]. These and other methodological discrepancies affect the comparability of the results. However, most analyses showed similar results with a tendency in favour of genetic screening being cost effective.
Gene mutations are detectable by standard methods such as Sanger Sequencing of polymerase chain reaction, amplified DNA sequencing or alternative assays. For the last decades, few facilities in Brazil have offered genetic testing, motivated either by research purposes or by reimbursement for privately insured women [62]. For low- and middle-income countries, multigene panels that incorporate recurrent mutations are an alternative to more expensive standard techniques. A panel assay designed for recurrent Hispanic BRCA mutations (HISPANEL) was already implemented throughout Latin America [63, 64]. In a sample of 193 Brazilian women, however, the panel assay showed a sensitivity of only 27% for detecting a BRCA mutation [65]. Nevertheless, the development of a genetic panel that accounts for the heterogeneous structure of the Brazilian population has the potential to be a cost-effective alternative and should be further investigated in future studies [66].
5 Conclusion
The present model showed that a screen-and-treat strategy for healthy women at risk for HBOC results in additional QALYs/LYG and moderately more costs, with an ICER of R$24,264 per QALY gained. The cost effectiveness of the screen-and-treat intervention depends on a still undecided cost-effectiveness threshold for Brazil. Further studies should assess the cost effectiveness of multigene test strategies, which are a viable and promising option for preventing breast and ovarian cancer in low- and middle-income countries. Although the choice of a risk-reducing surgery depends on the woman’s preferences and her current personal situation, increased accessibility of genetic testing should support her decision.
Data Availability
The model used in the current study can be made available to researchers upon request.
References
Estimativa 2018: incidência de câncer no Brasil. Instituto Nacional de Câncer José Alencar Gomes da Silva Ministério da Saúde. 2017;ISBN 978–85–7318–362–7.
Gonzaga CM, Freitas-Junior R, Curado MP, Sousa AL, Souza-Neto JA, Souza MR. Temporal trends in female breast cancer mortality in Brazil and correlations with social inequalities: ecological time-series study. BMC public health. 2015;15:96. https://doi.org/10.1186/s12889-015-1445-7.
Colditz GA, Willett Wc Fau - Hunter DJ, Hunter Dj Fau - Stampfer MJ, Stampfer Mj Fau - Manson JE, Manson Je Fau - Hennekens CH, Hennekens Ch Fau - Rosner BA et al. Family history, age, and risk of breast cancer. Prospective data from the Nurses' Health Study. (0098–7484 (Print)).
Ford D, Easton DF, Stratton M, Narod S, Goldgar D, Devilee P, et al. Genetic heterogeneity and penetrance analysis of the BRCA1 and BRCA2 genes in breast cancer families. Am J Hum Genet. 1998;62(3):676–89. https://doi.org/10.1086/301749.
De Felice F, Marchetti C, Musella A, Palaia I, Perniola G, Musio D, et al. Bilateral risk-reduction mastectomy in BRCA1 and BRCA2 mutation carriers: a meta-analysis. Ann Surg Oncol. 2015;22(9):2876–80. https://doi.org/10.1245/s10434-015-4532-1.
Domchek SM, Friebel Tm Fau - Singer CF, Singer Cf Fau - Evans DG, Evans Dg Fau - Lynch HT, Lynch Ht Fau - Isaacs C, Isaacs C Fau - Garber JE et al. Association of risk-reducing surgery in BRCA1 or BRCA2 mutation carriers with cancer risk and mortality. (1538–3598 (Electronic)).
Mary BD, Robert P, Michael B, Saundra SB, Meagan F, Susan F, et al. NCCN guidelines insights: genetic/familial high-risk assessment: breast and ovarian, Version 2.2017. J Natl Compr Canc Netw. 2017;15(1):9–20. https://doi.org/10.6004/jnccn.2017.0003.
Nelson HD, Pappas M, Zakher B, Mitchell JP, Okinaka-Hu L, Fu R. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: a systematic review to update the U.S. Preventive Services Task Force recommendation. Ann Intern Med. 2014;160(4):255–66. https://doi.org/10.7326/m13-1684.
Diretrizes de utilizacao para cobertura de procedimentos da saúde suplementar Agência Nacional de Saúde Suplementar. 2018;Anexo II.
Ashton-Prolla P, Goldim Jr Fau - Vairo FPE, Vairo Fp Fau - da Silveira Matte U, da Silveira Matte U Fau - Sequeiros J, Sequeiros J. Genomic analysis in the clinic: benefits and challenges for health care professionals and patients in Brazil. (1868–310X (Print)).
Ramos MCA, Folgueira M, Maistro S, Campolina AG, Soarez PC, Bock GH, et al. Cost effectiveness of the cancer prevention program for carriers of the BRCA1/2 mutation. Rev Saude Publica. 2018;52:94. https://doi.org/10.11606/S1518-8787.2018052000643.
Nelson HD, Pappas M, Cantor A, Haney E, Holmes R. Risk assessment, genetic counseling, and genetic testing for BRCA-related cancer in women: updated evidence report and systematic review for the US preventive services task force. JAMA. 2019;322(7):666–85. https://doi.org/10.1001/jama.2019.8430.
Müller D, Danner M, Rhiem K, Stollenwerk B, Engel C, Rasche L, et al. Cost-effectiveness of different strategies to prevent breast and ovarian cancer in German women with a BRCA 1 or 2 mutation. Eur J Health Econ. 2018;19(3):341–53. https://doi.org/10.1007/s10198-017-0887-5.
Cancer DfNbtNCCf. Familial Breast Cancer: Full cost effectiveness evidence review & reports. 2013;Update of clinical guideline 14 and 41.
Freitas-Junior R, Gagliato DM, Moura Filho JWC, Gouveia PA, Rahal RMS, Paulinelli RR, et al. Trends in breast cancer surgery at Brazil's public health system. J Surg Oncol. 2017;115(5):544–9. https://doi.org/10.1002/jso.24572.
Domchek SM, Jhaveri K Fau - Patil S, Patil S Fau - Stopfer JE, Stopfer Je Fau - Hudis C, Hudis C Fau - Powers J, Powers J Fau - Stadler Z et al. Risk of metachronous breast cancer after BRCA mutation-associated ovarian cancer. (1097–0142 (Electronic)).
Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ. 2016;355:i4919–i. https://doi.org/10.1136/bmj.i4919.
Alemar B, Gregorio C, Herzog J, Matzenbacher Bittar C, Brinckmann O, Netto C, Artigalas O, et al. BRCA1 and BRCA2 mutational profile and prevalence in hereditary breast and ovarian cancer (HBOC) probands from Southern Brazil: Are international testing criteria appropriate for this specific population? PLoS ONE. 2017;12(11):e0187630. https://doi.org/10.1371/journal.pone.0187630.
Kuchenbaecker KB, Hopper JL, Barnes DR, Phillips KA, Mooij TM, Roos-Blom MJ, et al. Risks of breast, ovarian, and contralateral breast cancer for BRCA1 and BRCA2 mutation carriers. JAMA. 2017;317(23):2402–16. https://doi.org/10.1001/jama.2017.7112.
Girardi F, Barnes DR, Barrowdale D, Frost D, Brady AF, Miller C, et al. Risks of breast or ovarian cancer in BRCA1 or BRCA2 predictive test negatives: findings from the EMBRACE study. Genet Med. 2018;20(12):1575–82. https://doi.org/10.1038/gim.2018.44.
Domchek SM, Gaudet MM, Stopfer JE, Fleischaut MH, Powers J, Kauff N, et al. Breast cancer risks in individuals testing negative for a known family mutation in BRCA1 or BRCA2. Breast Cancer Res Treat. 2010;119(2):409–14. https://doi.org/10.1007/s10549-009-0611-y.
Harvey SL, Milne RL, McLachlan SA, Friedlander ML, Birch KE, Weideman P, et al. Prospective study of breast cancer risk for mutation negative women from BRCA1 or BRCA2 mutation positive families. Breast Cancer Res Treat. 2011;130(3):1057–61. https://doi.org/10.1007/s10549-011-1733-6.
Giornelli GH. Management of relapsed ovarian cancer: a review. SpringerPlus. 2016;5(1):1197. https://doi.org/10.1186/s40064-016-2660-0.
Paes MF, Renata D, Klesia P. A retrospective analysis of clinicopathological and prognostic characteristics of ovarian tumors in the State of Espírito Santo, Brazil. J Ovar Res. 2011;4:14.
Schneider JC. Five-year survival and prognostic factors in women with breast cancer in Santa Catarina State, Brazil. Cad Saúde Pública, Rio de Janeiro. 2009;25(6):1285–96.
Allemani C, Matsuda, Tomohiro, Di Carlo, Veronica, Harewood, Rhea, Matz, Melissa, Nikšić, Maja, Bonaventure, Audrey, Valkov, Mikhail , Christopher J Johnson JE, Olufemi J Ogunbiyi, Gulnar Azevedo e Silva, Wan-Qing Chen, Sultan Eser, Gerda Engholm, Charles A Stiller,, Alain Monnereau RRW, Otto Visser, Gek Hsiang Lim, Joanne Aitken, Hannah K Weir, Michel P Coleman, CONCORD Working Group*. Global surveillance of trends in cancer survival 2000–14 (CONCORD-3): analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet. 2018; 391(1023–75). doi:10.1016/S0140–6736(17)33326–3.
Chai X, Friebel TM, Singer CF, Evans DG, Lynch HT, Isaacs C, et al. Use of risk-reducing surgeries in a prospective cohort of 1,499 BRCA1 and BRCA2 mutation carriers. Breast Cancer Res Treat. 2014;148(2):397–406. https://doi.org/10.1007/s10549-014-3134-0.
Eleje GU, Eke AC, Ezebialu IU, Ikechebelu JI, Ugwu EO, Okonkwo OO. Risk-reducing bilateral salpingo-oophorectomy in women with BRCA1 or BRCA2 mutations. Cochrane Database Syst Rev. 2018;8:12464. https://doi.org/10.1002/14651858.CD012464.pub2.
Evans DG, Ingham SL, Baildam A, Ross GL, Lalloo F, Buchan I, et al. Contralateral mastectomy improves survival in women with BRCA1/2-associated breast cancer. Breast Cancer Res Treat. 2013;140(1):135–42. https://doi.org/10.1007/s10549-013-2583-1.
Grann VR, Patel P, Bharthuar A, Jacobson JS, Warner E, Anderson K, et al. Breast cancer-related preferences among women with and without BRCA mutations. Breast Cancer Res Treat. 2010;119(1):177–84. https://doi.org/10.1007/s10549-009-0373-6.
Familial Breast Cancer: Classification and Care of People at Risk of Familial Breast Cancer and Management of Breast Cancer and Related Risks in People with a Family History of Breast Cancer. National Collaborating Centre for Cancer (UK) CTI.
Halbert CH, Stopfer JE, McDonald J, Weathers B, Collier A, Troxel AB, et al. Long-term reactions to genetic testing for BRCA1 and BRCA2 mutations: does time heal women's concerns? J Clin Oncol. 2011;29(32):4302–6. https://doi.org/10.1200/jco.2010.33.1561.
Freitas RJ, Nunes RD, Martins E, Curado MP, Freitas NMA, Soares LR, et al. Prognostic factors and overall survival of breast cancer in the city of Goiania, Brazil: a population-based study. Revista do Colegio Brasileiro de Cirurgioes. 2017;44(5):435–43. https://doi.org/10.1590/0100-69912017005003.
Peasgood T, Ward SE, Brazier J. Health-state utility values in breast cancer. Exp Rev Pharmacoecon Outcomes Res. 2010;10(5):553–66. https://doi.org/10.1586/erp.10.65.
Stein K, Sugar C, Velikova G, Stark D. Putting the 'Q' in quality adjusted life years (QALYs) for advanced ovarian cancer - An approach using data clustering methods and the internet. Eur J Cancer. 2007;43(1):104–13. https://doi.org/10.1016/j.ejca.2006.09.007.
Ara R, Wailoo A. Using health state utility values in models exploring the cost-effectiveness of health technologies. (1524–4733 (Electronic)).
Ministério da Saúde (BR) SdC, Tecnologia e Insumos Estratégicos,, Tecnologia. DdCe. Diretrizes metodológicas: diretriz de avaliação econômica. 2014;2.ed.(Brasília (DF)).
DATASUS. Sistema de Informação sobre Mortalidade. Ministério da Saúde (BR). 2018. https://www2.datasus.gov.br/DATASUS/index.php?area=060701.2008.
Shemilt I, Thomas J, Morciano M. A web-based tool for adjusting costs to a specific target currency and price year. Evid Policy A J Res Debate Pract. 2010;6:51–60. https://doi.org/10.1332/174426410X482999.
Evans DG, Eccles DM, Rahman N, Young K, Bulman M, Amir E, et al. A new scoring system for the chances of identifying a BRCA1/2 mutation outperforms existing models including BRCAPRO. J Med Genet. 2004;41(6):474–80. https://doi.org/10.1136/jmg.2003.017996.
Vemer P, Corro Ramos I, van Voorn GA, Al MJ, Feenstra TL. AdViSHE: a validation-assessment tool of health-economic models for decision makers and model users. PharmacoEconomics. 2016;34(4):349–61. https://doi.org/10.1007/s40273-015-0327-2.
Paluch-Shimon S, Cardoso F, Sessa C, Balmana J, Cardoso MJ, Gilbert F et al. Prevention and screening in BRCA mutation carriers and other breast/ovarian hereditary cancer syndromes: ESMO Clinical Practice Guidelines for cancer prevention and screening. (1569–8041 (Electronic)).
World Health Organization. Macroeconomics and health: investing in health for economic development. Geneva: World Health Organization. 2001. https://www1.worldbank.org/publicsector/pe/PEAMMarch2005/CMHReport.pdf.
Pichon-Riviere A AF, Marti SG The efficiency path to universal health coverage: derivation of cost-effectiveness thresholds based on health expenditures and life expectancy.Updated country-level estimates for 194 countries. 6th ISPOR Latin America; Sao Paulo: ISPOR; 2017.
Woods B, Revill P, Sculpher M, Claxton K. Country-level cost-effectiveness thresholds: initial estimates and the need for further research. Value Health. 2016;19(8):929–35. https://doi.org/10.1016/j.jval.2016.02.017.
Yuba TY, Novaes HMD, de Soarez PC. Challenges to decision-making processes in the national HTA agency in Brazil: operational procedures, evidence use and recommendations. Health Res Policy Syst. 2018;16(1):40. https://doi.org/10.1186/s12961-018-0319-8.
Elias FT, Araujo DV. How health economic evaluation (HEE) contributes to decision-making in public health care: the case of Brazil. Zeitschrift fur Evidenz, Fortbildung und Qualitat im Gesundheitswesen. 2014;108(7):405–12. https://doi.org/10.1016/j.zefq.2014.08.021.
Silva MJSd, Osorio-de-Castro CGS. Organização e práticas da assistência farmacêutica em oncologia no âmbito do Sistema Único de Saúde. Interface - Comunicação, Saúde, Educação. 2019. doi:10.1590/interface.180297.
Evans DG, Ingham SL, Buchan I, Woodward ER, Byers H, Howell A, et al. Increased rate of phenocopies in all age groups in BRCA1/BRCA2 mutation kindred, but increased prospective breast cancer risk is confined to BRCA2 mutation carriers. Cancer Epidemiol Biomark Prevent. 2013;22(12):2269–76. https://doi.org/10.1158/1055-9965.EPI-13-0316-T.
Vos JR, de Bock GH, Teixeira N, van der Kolk DM, Jansen L, Mourits MJ, et al. Proven non-carriers in BRCA families have an earlier age of onset of breast cancer. Eur J Cancer. 2013;49(9):2101–6. https://doi.org/10.1016/j.ejca.2013.02.018.
Gabai-Kapara E, Lahad A, Kaufman B, Friedman E, Segev S, Renbaum P, et al. Population-based screening for breast and ovarian cancer risk due to BRCA1 and BRCA2. Proc Natl Acad Sci USA. 2014;111(39):14205–10. https://doi.org/10.1073/pnas.1415979111.
Mavaddat N, Peock S, Frost D, Ellis S, Platte R, Fineberg E, et al. Cancer risks for BRCA1 and BRCA2 mutation carriers: results from prospective analysis of EMBRACE. J Natl Cancer Inst. 2013;105(11):812–22. https://doi.org/10.1093/jnci/djt095.
Brohet RM, Velthuizen ME, Hogervorst FB, Meijers-Heijboer HE, Seynaeve C, Collee MJ, et al. Breast and ovarian cancer risks in a large series of clinically ascertained families with a high proportion of BRCA1 and BRCA2 Dutch founder mutations. J Med Genet. 2014;51(2):98–107. https://doi.org/10.1136/jmedgenet-2013-101974.
Chen S, Iversen ES, Friebel T, Finkelstein D, Weber BL, Eisen A, et al. Characterization of BRCA1 and BRCA2 mutations in a large United States sample. J Clin Oncol. 2006;24(6):863–71. https://doi.org/10.1200/JCO.2005.03.6772.
Milne RL, Osorio A, Cajal TR, Vega A, Llort G, de la Hoya M, et al. The average cumulative risks of breast and ovarian cancer for carriers of mutations in BRCA1 and BRCA2 attending genetic counseling units in Spain. Clin Cancer Res. 2008;14(9):2861–9. https://doi.org/10.1158/1078-0432.CCR-07-4436.
Antoniou AC, Cunningham AP, Peto J, Evans DG, Lalloo F, Narod SA, et al. The BOADICEA model of genetic susceptibility to breast and ovarian cancers: updates and extensions. Br J Cancer. 2008;98(8):1457–66. https://doi.org/10.1038/sj.bjc.6604305.
Hopper JL, Southey Mc Fau - Dite GS, Dite Gs Fau - Jolley DJ, Jolley Dj Fau - Giles GG, Giles Gg Fau - McCredie MR, McCredie Mr Fau - Easton DF et al. Population-based estimate of the average age-specific cumulative risk of breast cancer for a defined set of protein-truncating mutations in BRCA1 and BRCA2. Australian Breast Cancer Family Study. (1055–9965 (Print)).
Torres D, Bermejo JL, Rashid MU, Briceño I, Gil F, Beltran A, et al. Prevalence and Penetrance of BRCA1 and BRCA2 Germline Mutations in Colombian Breast Cancer Patients. Sci Rep. 2017;7(1):4713. https://doi.org/10.1038/s41598-017-05056-y.
Jara L, Morales S, de Mayo T, Gonzalez-Hormazabal P, Carrasco V, Godoy R. Mutations in BRCA1, BRCA2 and other breast and ovarian cancer susceptibility genes in Central and South American populations. (0717–6287 (Electronic)).
Metcalfe KA, Birenbaum-Carmeli D, Lubinski J, Gronwald J, Lynch H, Moller P, et al. International variation in rates of uptake of preventive options in BRCA1 and BRCA2 mutation carriers. Int J Cancer. 2008;122(9):2017–22. https://doi.org/10.1002/ijc.23340.
Holland ML, Huston A Fau - Noyes K, Noyes K. Cost-effectiveness of testing for breast cancer susceptibility genes. (1524–4733 (Electronic)).
Koike Folgueira MAA, Maistro S, Teixeira N, Mourits MJE, Chammas R, Hendrika de Bock G. How should genetic counseling for ovarian cancer be implemented in a middle-income country? An insight based on the Brazilian scenario. J Glob Oncol. 2018;4:1–3. doi:10.1200/JGO.17.00230.
Hernandez JE, Llacuachaqui M, Palacio GV, Figueroa JD, Madrid J, Lema M et al. Prevalence of BRCA1 and BRCA2 mutations in unselected breast cancer patients from medellin, Colombia. (1731–2302 (Print)).
Abugattas J, Llacuachaqui M, Allende YS, Velasquez AA, Velarde R, Cotrina J et al. Prevalence of BRCA1 and BRCA2 mutations in unselected breast cancer patients from Peru. (1399–0004 (Electronic)).
Alemar B, Herzog J, Brinckmann Oliveira Netto C, Artigalas O, Schwartz IVD, Matzenbacher Bittar C et al. Prevalence of Hispanic BRCA1 and BRCA2 mutations among hereditary breast and ovarian cancer patients from Brazil reveals differences among Latin American populations. 2016(2210–7762 (Print)).
Chavarri-Guerra Y, Blazer KR, Weitzel JN. Genetic Cancer Risk Assessment for Breast Cancer in Latin America. (0034–8376 (Print)).
Registro de Câncer de São Paulo - Departamento de Epidemiologia da Faculdade de Saúde Pública da Universidade de Sao Paulo. https://tabnet.saude.prefeitura.sp.gov.br/cgi/deftohtm3.exe?secretarias/saude/TABNET/CA/cancer.def.
Peres VC. Veloso, Danyelle Lorrane Carneiro, Xavier, Raphaela Maioni, Salge, Ana Karina Marques, Guimarães, Janaína Valadares Breast cancer in women: recurrence and survival at five years. Text Context Nursing. 2015;24(3):740–7. https://doi.org/10.1590/0104-0707201501123_014.
Schneider IJCO. Eleonora Five-year survival and prognostic factors in women with breast cancer in Santa Catarina State, Brazil. Cad Saúde Pública, Rio de Janeiro. 2009;25(6):1285–96.
Metcalfe KA, Lynch Ht Fau - Ghadirian P, Ghadirian P Fau - Tung N, Tung N Fau - Olivotto IA, Olivotto Ia Fau - Foulkes WD, Foulkes Wd Fau - Warner E et al. The risk of ovarian cancer after breast cancer in BRCA1 and BRCA2 carriers. (0090–8258 (Print)).
Sullivan PW, Lawrence Wf Fau - Ghushchyan V, Ghushchyan V. A national catalog of preference-based scores for chronic conditions in the United States. (0025–7079 (Print)).
Havrilesky LJ, Broadwater G, Davis DM, Nolte KC, Barnett JC, Myers ER, et al. Determination of quality of life-related utilities for health states relevant to ovarian cancer diagnosis and treatment. Gynecol Oncol. 2009;113(2):216–20. https://doi.org/10.1016/j.ygyno.2008.12.026.
Acknowledgements
Open Access funding provided by Projekt DEAL. The model used in this study was provided to the journal’s peer reviewers for their reference when reviewing the manuscript.
Funding
The German Chancellor Fellowship from the Alexander von Humboldt Foundation supported Julia Correa-Galendi during the development of this research. Project funded by the German Chancellor Fellowship from the Alexander from Humboldt Foundation.
Author information
Authors and Affiliations
Contributions
Conception and design: JC-G, DM, SS, MPED. Collection and assembly of data: JC-G, MPED. Data analysis and interpretation: JC-G, DM. Manuscript writing: JC-G, DM, SS. Final approval of manuscript: JC-G, DM, MPED, SS.
Corresponding author
Ethics declarations
Conflict of Interest
The authors confirm that they have no conflicts of interest to declare.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Simoes Correa-Galendi, J., del Pilar Estevez Diz, M., Stock, S. et al. Economic Modelling of Screen-and-Treat Strategies for Brazilian Women at Risk of Hereditary Breast and Ovarian Cancer. Appl Health Econ Health Policy 19, 97–109 (2021). https://doi.org/10.1007/s40258-020-00599-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40258-020-00599-0