Abstract
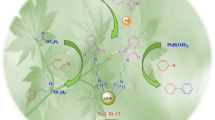
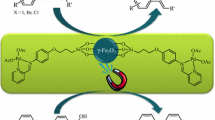
A simple, efficient and less expensive protocol for the phosphine-free C–C coupling reactions and synthesis of anilines in the presence of 2-aminobenzamide complex of palladium supported on Fe3O4 magnetic nanoparticles (Pd(0)-ABA-Fe3O4) has been reported. The Suzuki reaction was carried out in water or PEG using phenylboronic acid (PhB(OH)2) or sodium tetraphenyl borate (NaBPh4). Pd(0)-ABA-Fe3O4 has been found promising for Heck reaction of butyl acrylate, styrene or acrylonitrile with aryl halides (including Cl, Br and I). Also, Pd(0)-ABA-Fe3O4 has been found as efficient catalyst for the amination of aryl halides using aqueous ammonia. The products have been obtained in short reaction times and high yields. The catalyst was easily separated using an external magnet from the reaction mixture and reused for several runs without significant loss of its catalytic efficiency or palladium leaching. The leaching of catalyst has been examined by hot filtration and ICP-OES technique. The nanomagnetical catalyst was characterized by FTIR, TGA, XRD, VSM, TEM, SEM, EDS, DLS and ICP-OES techniques.













Similar content being viewed by others
References
S. Das, S. Bhunia, T. Maity, S. Koner, J. Mol. Catal. A Chem. 394, 188 (2014)
M. Wena, S. Takakura, K. Fuku, K. Moria, H. Yamashita, Catal. Today 242, 381 (2015)
S. Kazemi Movahed, R. Esmatpoursalmani, A. Bazgir, RSC Adv. 4, 14586 (2014)
N. Miyaura, A. Suzuki, Chem. Rev. 95, 2457 (1995)
G. Herve, G. Sartori, G. Enderlin, G. Mackenzie, C. Len, RSC Adv. 4, 18558 (2014)
D.A. Alonso, C. Najera, Chem. Soc. Rev. 39, 2891 (2010)
X. Le, Z. Dong, Z. Jin, Q. Wang, J. Ma, Catal. Commun. 53, 47 (2014)
D. Zhang, Q. Wang, Coord. Chem. Rev. 286, 1 (2015)
A. Ghorbani-Choghamarani, P. Moradi, B. Tahmasbi, RSC Adv. 6, 56638 (2016)
A. Ghorbani-Choghamarani, B. Tahmasbi, P. Moradi, N. Havasi, Appl. Organomet. Chem. 30, 619 (2016)
M. Opanasenko, P. Stepnicka, J. Cejka, RSC Adv. 4, 65137 (2014)
P. Sharma, A.P. Singh, Catal. Sci. Technol. 4, 2978 (2014)
S.S. Soni, D.A. Kotadia, Catal. Sci. Technol. 4, 510 (2014)
M.M. Heravi, S. Sadjadi, J. Iran. Chem. Soc. 6, 1 (2009)
P. Nehra, B. Khungar, K. Pericherla, S.C. Sivasubramanian, A. Kumar, Green Chem. 16, 4266 (2014)
P.M. Uberman, L.A. Perez, S.E. Martın, G.I. Lacconi, RSC Adv. 4, 12330 (2014)
M. Gholinejad, B. Karimi, F. Mansouri, J. Mol. Catal. A Chem. 386, 20 (2014)
B. Atashkar, A. Rostami, B. Tahmasbi, Catal. Sci. Technol. 3, 2140 (2013)
R.K. Sharma, M. Yadav, R. Gaur, Y. Monga, A. Adholeya, Catal. Sci. Technol. 5, 2728 (2015)
V. Polshettiwar, R. Luque, A. Fihri, H. Zhu, M. Bouhrara, J.M. Basset, Chem. Rev. 111, 3036 (2011)
B. Atashkar, A. Rostami, H. Gholami, B. Tahmasbi, Res. Chem. Intermed. 41, 3675 (2015)
A. Rostami, B. Tahmasbi, A. Yari, Bull. Korean Chem. Soc. 34, 1521 (2013)
C.W. Lim, I.S. Lee, Nano Today 5, 412 (2010)
K. Kulkarni, J. Friend, L. Yeo, P. Perlmutter, Ultrason. Sonochem. 21, 1305 (2014)
T. Yu, X.Y. Wu, J. Yang, Tetrahedron Lett. 55, 4071 (2014)
R.B. Nasir Baig, R.S. Varma, RSC Adv. 4, 6568 (2014)
F. Havasi, A. Ghorbani-Choghamarani, F. Nikpour, N. J. Chem. 39, 6504 (2015)
R.A. Green, J.F. Hartwig, Angew. Chem. Int. Ed. 54, 1 (2015)
B. Yang, L. Liao, Y. Zeng, X. Zhu, Y. Wan, Catal. Commun. 45, 100 (2014)
J. Li, L. Liu, RSC Adv. 2, 10485 (2012)
A. Ghorbani-Choghamarani, Z. Darvishnejad, B. Tahmasbi, Inorg. Chim. Acta 435, 223 (2015)
M. Hajjami, B. Tahmasbi, RSC Adv. 5, 59194 (2015)
W. Zhang, X. Chen, T. Tang, E. Mijowska, Nanoscale 6, 12884 (2014)
M. Nikoorazm, A. Ghorbani-Choghamarani, M. Khanmoradi, Appl. Organomet. Chem. 30, 236 (2016)
A. Ghorbani-Choghamarani, M. Hajjami, B. Tahmasbi, N. Noori, J. Iran. Chem. Soc. 13, 2193 (2016)
A. Ghorbani-Choghamarani, B. Tahmasbi, F. Arghand, S. Faryadi, RSC Adv. 5, 92174 (2015)
A. Ghorbani-Choghamarani, B. Tahmasbi, N. J. Chem. 40, 1205 (2016)
Y.Y. Peng, J. Liu, X. Lei, Z. Yin, Green Chem. 12, 1072 (2010)
L. Baia, J.X. Wanga, Adv. Synth. Catal. 350, 315 (2007)
X. Zheng, Q. Yang, Z. Li, Z. Zhu, X. Cui, H. Fu, H. Chen, R. Li, Catal. Commun. 57, 143 (2014)
M. Nasrollahzadeh, S.M. Sajadi, M. Maham, J. Mol. Catal. A: Chem. 396, 297 (2015)
V.W. Faria, D.G.M. Oliveira, M.H.S. Kurz, F.F. Goncalves, C.W. Scheeren, G.R. Rosa, RSC Adv. 4, 13446 (2014)
S.J. Sabounchei, A. Hashemi, Inorg. Chem. Commun. 47, 123 (2014)
M. Nasrollahzadeh, A. Azarian, M. Maham, A. Ehsani, J. Ind. Eng. Chem. 21, 746 (2015)
T. Chen, J. Gao, M. Shi, Tetrahedron 62, 6289 (2006)
N. Iranpoor, H. Firouzabadi, A. Tarassoli, M. Fereidoonnezhad, Tetrahedron 66, 2415 (2010)
Q. Xu, W.L. Duan, Z.Y. Lei, Z.B. Zhu, M. Shi, Tetrahedron 61, 11225 (2005)
Y.P. Wang, H.M. Lee, J. Organomet. Chem. 791, 90 (2015)
Q. Du, W. Zhang, H. Ma, J. Zheng, B. Zhou, Y. Li, Tetrahedron 68, 3577 (2012)
Y. Leng, F. Yang, K. Wei, Y. Wu, Tetrahedron 66, 1244 (2010)
L. Wang, H. Li, P. Li, Tetrahedron 65, 364 (2009)
A. Ghorbani-Choghamarani, B. Tahmasbi, P. Moradi, RSC Adv. 6, 43205 (2016)
A. Ghorbani-Choghamarani, B. Tahmasbi, P. Moradi, Appl. Organomet. Chem. 30, 422 (2016)
K.G. Thakur, K.S. Srinivas, K. Chiranjeevi, G. Sekar, Green Chem. 13, 2326 (2011)
M. Nikoorazm, A. Ghorbani-Choghamarani, N. Noori, B. Tahmasbi, Appl. Organomet. Chem. 30, 843 (2016)
M. Gholinejad, H.R. Shahsavari, Inorg. Chim. Acta 421, 433 (2014)
H.A. Patel, A.L. Patel, A.V. Bedekar, Appl. Organomet. Chem. 29, 1 (2015)
Acknowledgements
This work was supported by the research facilities of Ilam University, Ilam, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghorbani-Choghamarani, A., Tahmasbi, B., Noori, N. et al. A new palladium complex supported on magnetic nanoparticles and applied as an catalyst in amination of aryl halides, Heck and Suzuki reactions. J IRAN CHEM SOC 14, 681–693 (2017). https://doi.org/10.1007/s13738-016-1020-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-016-1020-x