Abstract
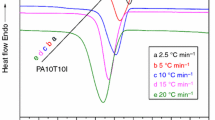
The isothermal crystallization process, kinetics, melting behavior and equilibrium melting point of bio-based semi-aromatic high-temperature polyamide PA5T/56 were studied in detail by differential scanning calorimetry. The results showed that the crystallization properties of PA5T/56 are strongly dependent on temperature, with increases in isothermal crystallization temperature, the crystallization process becomes longer and the crystallization speed slows down. The isothermal crystallization process and kinetics were analyzed by Avrami equation, the Avrami index (n value) is between 1.84 and 2.41, it is speculated that the growth mode of the crystal is the coexistence of one-dimensional needle growth and two-dimensional sheet growth, which is confirmed by two-dimensional small angle X-ray scattering instrument and field emission scanning electron microscope. At the same time, according to Turnbull–Fisher equation, it is reasonable to use Arvami equation to treat the isothermal crystallization process of PA5T/56. According to Hoffman–Weeks theory, the equilibrium melting point of PA5T/56 raises with the increase in PA5T content. The isothermal crystallization activation energy of PA5T/56 was calculated by Arrhenius equation, and the kg value of PA5T/56 was calculated by Lauritzen–Hoffman equation, both of them increase first and then decrease with the increase in PA5T content. Therefore, the study on the crystallization mechanism and crystal formation process of PA is of great significance for controlling the crystallinity of semi-crystalline polymers to obtain the required performance.
Graphical abstract













Similar content being viewed by others
References
Rwei S-P, Ranganathan P, Chiang W-Y, Lee Y-H (2018) Synthesis of low melting temperature aliphatic-aromatic copolyamides derived from novel bio-based semi aromatic monomer. Polymers 10:793–798
Gao J, Huang W, He W, Long L, Qin S (2021) Superior flame retardancy of glass fiber-reinforced polyamide 6T composites by synergism between DOPO-based derivative and carbon nanotube. J Thermal Anal Calorim 9:121–130
Watt E, Abdelwahab MA, Mohanty AK, Misra M (2021) Biocomposites from biobased polyamide 4,10 and waste corn cob based biocarbon. Compos Part A Appl Sci Manuf 145:106340
Zhang XK, Xie TX, Yang GS (2006) Isothermal crystallization and melting behaviors of nylon 11/nylon 66 alloys by in situ polymerization. Polymer 47:2116–2126
Liu BX, Hu GS, Zhang JT, Yan W (2019) Study on non-isothermal crystallization kinetics of high temperature resistant PA10T/66 and PA10T/66/GF. Appl Eng Plast 47:105–109
Wang ZQ, Hu GS, Zhang JT (2017) Isothermal crystallization kinetics of melt polymerization of high temperature resistant polyamide. Mater Guide 031:137–144152
Wang J, Hu G, Zhang J, Zhang B, Wang Z (2014) Study on non-isothermal crystallization kinetics of heat-resistant PA6T/11 copolyamide and its preparation. Plast Sci Technol 11:34–38
Su CX, Wang DB, Zhang CH, Zhou ZX, Cao M, Jiang SJ, Huang XB (2019) Non-isothermal crystallization kinetics of pa10t and PA10T/1012. Appl Eng Plast 47:105–116
Hu GS, Ding ZY, Li YC, Wang BB (2009) Crystalline morphology and melting behavior of nylon11/ethylene-vinyl alcohol/dicumyl peroxide blends. J Polym Res 16:263–269
Liu HX, Huang YY, Yuan L, He P, Cai Y, Xu Y, Yu Y, Xiong H (2010) Isothermal crystallization kinetics of modified bamboo cellulose/PCL composites. Carbohydr Polym 79:513–519
Zou P, Tang S, Fu Z, Xiong H (2009) Isothermal and non-isothermal crystallization kinetics of modified rape straw flour/high-density polyethylene composites. Int J Therm Sci 48:837–846
Liu P, Xue Y, Men Y (2019) Melt memory effect beyond equilibrium melting point in commercial isotactic polybutene-1. Ind Eng Chem Res 58:44–57
Run M, Song H, Wang S, Bai L, Jia Y (2010) Crystal morphology, melting behaviors and isothermal crystallization kinetics of SCF/PTT composites. Polym Compos 30:87–94
Zhao H, Zhang D, Li Y (2020) Morphology and crystallization kinetics of rubber-modified nylon 6 prepared by anionic in-situ polymerization. Sci Eng Compos Mater 27:204–215
Liu S, Yu Y, Cui Y, Zhang HF, Mo ZS (1998) Isothermal and nonisothermal crystallization kinetics of nylon-11. J Appl Polym Sci 70:2371–2380
Lin CC (2010) The rate of crystallization of poly(ethylene terephthalate) by differential scanning calorimetry. Polym Eng Sci 23:113–116
Tjong SC, Chen SX, Li R (2015) Crystallization kinetics of compatibilized blends of a liquid crystalline polymer with polypropylene. J Appl Polym Sci 64:707–715
Biber E, Gündüz G, Mavis B, Colak U (2010) Compatibility analysis of Nylon 6 and poly(ethylene-n-butyl acrylate-maleic anhydride) elastomer blends using isothermal crystallization kinetics. Mater Chem Phys 122:93–101
Wang ZQ, Hu GS, Zhang JT, Xu JS, Shi WB (2018) Isothermal crystallization kinetics of Nylon 10T and Nylon 10T/1010 copolymers: effect of Sebacic acid as a third comonomer. J Wuhan Univ Technol (Mater Sci) 33:237–245
Neugebauer F, Ploshikhin V, Ambrosy J, Witt G (2016) Isothermal and non-isothermal crystallization kinetics of polyamide 12 used in laser sintering. J Therm Anal Calorim 124:925–933
Liu TX, Mo ZS, Wang SE, Hong F (1997) Isothermal melt and cold crystallization kinetics of poly(aryl ether ether ketone ketone) (PEEKK). Eur Polym J 33:1405–1414
Cai J, MiaoL LW, Yao K, Xiong H (2011) Isothermal crystallization kinetics of thermoplastic starch/poly(lactic acid) composites. Carbohydr Polym 86:941–947
Mondal A, Sohel MA, Arif PM, Thomas S, Sengupta A (2021) Effect of ABS on non-isothermal crystallization kinetics of polyamide 6. J Therm Anal Calorim 20:443–456
Acknowledgements
We thank all those who participated in the study. This research was supported by Shanghai Cathay Biotechnology Co., China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Meng, C., Liu, X. Isothermal crystallization kinetics of bio-based semi-aromatic high-temperature polyamide PA5T/56. Iran Polym J 31, 605–617 (2022). https://doi.org/10.1007/s13726-021-01012-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13726-021-01012-w