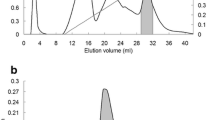
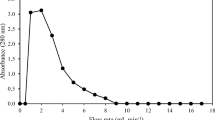
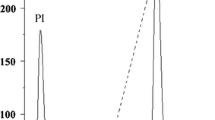
Abstract
A 35 kDa rabbit erythrocyte agglutinating lectin from the seeds of Cicer arietinum was purified and designated as CAL. The lectin was inhibited by fetuin and N-acetyl-d-galactosamine at a concentration of 20 and 50 mM respectively, but not by simple mono or oligosaccharides. CAL is active between pH 5 and 10 presented thermo stability up to 50 °C and demonstrated DNA damage inhibition at 30 µg concentration. The lectin elicited maximum mitogenic activity towards mice splenocytes at 7.5 µg ml− 1. CAL exerted an inhibitory activity on HIV-1 reverse transcriptase with IC50 of 180 µM. CAL abilities in animal bioassay resulted decreased levels of total triglyceride and creatinine. In vitro and in vivo studies revealed that CAL may constitute an important role impending biomedical applications.









Similar content being viewed by others
References
Agrawal P, Kumar S, Jaiswal YK, Das HR, Das RH (2011) A Mesorhizobium lipopolysaccharide (LPS) specific lectin (CRL) from the roots of nodulating host plant, Cicer arietinum. Biochimie 93:440–449. https://doi.org/10.1016/j.biochi.2010.10.017
Ahmad SA, Khan RHB, Ahmad A (1999) Physicochemical characterization of Cajanus cajan lectin: effect of pH and metal ions on lectin carbohydrate interaction. Biochem Biophys Acta 1427:378–384 (Pub Med PMID: 10350653)
Alatorre-Cruz JM, Pita-López W, López-Reyes RG, Ferriz-Martínez RA, Cervantes-Jiménez R et al (2017) Effects of intragastrically-administered tepary bean lectins on digestive and immune organs: preclinical evaluation. Toxicol Rep 12:56–64. https://doi.org/10.1016/j.toxrep.2017.12.008
Bardocz P, Grant G, Ewen SWB, Duguid TJ, Brown DS, Englyst K, Pusztai A (1995) Reversible effect of phytohaemagglutinin on the growth and metabolism of rat gastrointestinal tract. Gut 37:353–360
Bausch JN, Poretz RD (1977) Purification and properties of the hemagglutinin from Maclura pomifera seeds. Biochemistry 16(26):5790–5794. https://doi.org/10.1021/bi00645a023
Bhagyawant SS, Srivastava N (2008) Assessment of antinutritional factors and protein content in the seeds of chickpea cultivars and their divergence. J Cell Tissue Res 8(1):1333–1338
Borrebaeck CAK, Lonnerdal B, Etzler ME (1981) Metal chelate affinity chromatography of the Dolichosbz florus seed lectin and its subunits. FEBS Lett 130(2):194–196
Boyd MR, Gustafson KR, McMahon JB, Shoemaker RH, O’Keefe BR, Mori T, Wu RJ, Gulakowski L, Rivera MI, Laurencot CM (1997) Discovery of cyanovirin-N, a novel human immunodeficiency virus-inactivating protein that binds viral surface envelope glycoprotein gp120: potential applications to microbicide development. Antimicrob Agents Chemother 41:1521–1530
Chakraborti D. Sarkar A, Mondal HA, Das S (2009) Tissue specific expression of potent insecticidal, Allium sativumleaf agglutinin (ASAL) in important pulse crop, chickpea (Cicer arietinum L.) to resist the phloem feeding Aphis craccivora. Transgenic Res 18:529–544. https://doi.org/10.1007/s11248-009-9242-7
Chan YS, Wong JH, Ng TB (2011) A glucuronic acid binding leguminous lectin with mitogenic activity toward mouse splenocytes. Protein Pept Lett 18:194–202
Chan YS, Wong JH, Fang EF, Pan W, Ng TB (2012) Isolation of a glucosamine binding leguminous lectin with mitogenic activity towards splenocytes and anti-proliferative activity towards tumor cells. PLoS One 7(6):e38961. https://doi.org/10.1371/journal.pone.0038961
Coelho LC, Silva PM, Lima VL, Pontual EV, Paiva PM, Napoleão TH, Correia MT (2017) Lectins, interconnecting proteins with biotechnological/pharmacological and therapeutic applications. Evid Based Complement Alternat Med. https://doi.org/10.1155/2017/1594074
Collins RA, Ng TB, Fong WP, Wan CC, Yeung HW (1997) A comparison of human immunodeficiency virus type 1 inhibition by partially purified aqueous extracts of Chinese medicinal herbs. Life Sci 60:PL345–351
Decastel M, Boeck HD, Goussault Y, Bruyne DCK, Loontiens FG, Frenoy JP (1985) Effect of pH on oligomeric equilibrium and saccharide-binding properties of peanut agglutinin. Arch Biochem Biophys 240:811–819
Diaz IL, Partida ANG, Moreno LV (2017) Legume lectins: proteins with diverse applications. Int J Mol Sci 18(6):1242.
Drury RA, Wallington EA (1980) Carleton’s histological techniques, 5th edn. Oxford University Press, New York, p 195
Esteban R, Dopico B, Mun˜oz FJ, Romo S, Labrador E (2002) A seedling specific vegetative lectin gene is related to development in Cicer arietinum. Phys Plant 114:619–626
Estrada-Martínez LA, Moreno-Celis U, Cervantes-Jiménez R, Ferriz Martínez RA, Blanco-Labra A, García-Gasca T (2017) Plant lectins as medical tools against digestive system cancers. Int J Mol Sci 18:1403. https://doi.org/10.3390/ijms18071403
Faheina-Martins GV, da Silveira AL, Cavalcanti Márcio V, Ramos BC, Moraes MO, Pessoa C, Araújom DAM (2012) Antiproliferative effects of lectins from Canavalia ensiformis and Canavalia brasiliensis in human leukemia cell lines. Toxicol In Vitro 26:1161–1169. https://doi.org/10.1016/j.tiv.2012.06.017
Ferriz-Martínez R, García-García K, Torres-Arteaga I, Rodriguez-Mendez AJ, Guerrero-Carrillo MJ et al (2015) Tolerability assessment of a lectin fraction from Tepary bean seeds (Phaseolus acutifolius) orally administered to rats. Toxicol Rep 2:63–69. https://doi.org/10.1016/j.toxrep.2014.10.015
Goldstein IJ, Hayes CE (1978) The lectins: carbohydrate-binding proteins of plants and animals. Adv Carbohydr Chem Bio Chem 35:127–340. https://doi.org/10.1016/S0065-2318(08)60220-6
Guha G, Rajkumar V, Kumar RA, Mathew L (2011) The antioxidant and DNA protection potential of Indian tribal medicinal plants. Turk J Biol 35:233–242. https://doi.org/10.3906/biy-0906-64
Gurjar MM, Khan MI, Gaikwad SM (1998) Alpha-Galactoside binding lectin from Artocarpus hirsuta: characterization of the sugar specificity and binding site. Biochem Biophys Acta 1381:256–264
Jaffe CL, Rogozinski SE, Lis H, Sharon N (1977) Transition metal requirements of soybean agglutinin. FEBS Lett 82(2):191–196. https://doi.org/10.1016/0014-5793(77)80582-6
Kalita S, Kumar G, Karthik L, Rao KBV (2012) In vitro antioxidant and DNA damage inhibition activity of aqueous extract of Lantana camara L. (Verbenaceae) leaves. Asian Pacific J Trop Biomed 2:S1675–S1679. https://doi.org/10.1016/S2221-1691(12)60476-6
Katre UV, Gaikwad SM, Bhagyawant SS, Deshpande UD, Khan MI, Suresh CG (2005) Crystallization and preliminary X-ray characterization of a lectin from Cicer arietinum (chickpea). Acta Crystallogr F61:141–143. https://doi.org/10.1107/S1744309104032166
Kawagishi H, Nomura A, Mizuno T, Kimura A, Chiba S (1990) Isolation and characterization of a lectin from Grifola frondosa fruiting bodies. Biochem Biophys Acta 1034:247–252
Koike T, Beppu H, Kuzuya H, Maruta K, Shimpo K et al (1995) A 35 kDa mannose-binding lectin with hemagglutinating and mitogenic activities from “Kidachi Aloe” (Aloe arborescens Miller var. natalensis Berger). J Biochem 118:1205–1210
Kornfeld R, Gregory WT, Kornfeld SA (1972) Red Kidney Bean (Phaseolus vulgatis) Phytohemagglutinin. Methods Enzymol 28:344–349
Koval’chuk NV (2006) Dynamic of lectin activity during germination of bean seeds (Phaseolus vulgaris L.). Ukr Biokhim Zh 78:130–134
Kumar A, Chattopadhyay S (2007) DNA damage protecting activity and antioxidant potential of pudina extract. Food Chem 100:1377–1384. https://doi.org/10.1016/j.foodchem.2005.12.015
Laemmli UK, Favre M (1973) Gel electrophoresis of proteins. J Mol Biol 80:575–599. https://doi.org/10.1371/journal.pone.0054212
Liener IE (1962) Toxic factors in edible legumes and their elimination. Am J Clin Nutr 11:281–298
Loris R, Hamelryck T, Bouckaert J, Wyns L (1998) Legume lectin structure. Biochem Biophys Acta Protein Struct Mol Enzymol 1383:9–36. https://doi.org/10.1016/S0167-4838(97)00182-9
Monsigny M, Sene C, Obrenovitch A, Roche AC, Delmotte F, Boschetti E (1979) Properties of succinylated wheat-germ agglutinin. Eur J Biochem 98:39–45
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assay. J Immunol Methods 65(1–2):55–63
Nakagawa R, Yasokawa D, Ikeda T, Nagashima K (1996) Purification and characterization of two lectins from calllus of Helianthus tuberosus. Biosci Biotechnol Biochem 60:259–262
O’Farrell PH (1975) High resolution two-dimensional electrophoresis of proteins. J Biol Chem 250(10):4007–4021
Peumans WJ, Van Damme EJM (1995) Lectins as plant defense proteins. Plant Physiol 109:347–352
Peumans WJ, Van Damme EJ, Barre A, Rouge P (2001) Classification of plant lectins in families of structurally and evolutionary related proteins. Adv Exp Med Biol 491:27–54. https://doi.org/10.1007/978-1-4615-1267-7-3
Pusztai A (1991) General effect on animal cells. In: Pusztai A (ed) Plant lectins. Cambridge University Press, Cambridge, pp 105–159
Pusztai A (1993) Dietary lectins are metabolic signals foe the gut and modulate immune and hormone functions. Eur J Med Chem 47:691–699
Qureshi IA, Dash P, Srivastava PS, Koundal KR (2006) Purification and characterization of an N-acetyl-d-galactosamine-specific lectin from seeds of chickpea (Cicer arietinum L.). Phytochem Anal 17:350–356. https://doi.org/10.1002/pca.925
Richmond W (1973) Preparation and properties of a cholesterol oxidase from Nocardia sp. and its application to the enzymatic assay of total cholesterol in serum. Clin Chem 19:1350–1356
Rodrigues e Lacerda R, Silva do Nascimento E et al (2017) Lectin from seeds of a Brazilian lima bean variety (Phaseolus lunatus L.var. cascavel) presents antioxidant, antitumour and gastroprotective activities. Int J Biol Macromol 95:1072–1081. https://doi.org/10.1016/j.ijbiomac.2016.10.097
Rudiger H, Gabius HJ (2001) Plant lectins: occurrence, biochemistry, functions and applications. Glycoconjugate J 18(8):589–613
Salahuddin A (1992) Legume Lectins: homologous proteins with similar structure but distinct carbohydrate binding specificity. Indian J Biochem Biophys 29:388–393
Sharma U, Katre UV, Suresh CG (2015) Crystal structure of a plant albumin from Cicer arietinum (chickpea) possessing hemopexin fold and hemagglutination activity. Planta 241(5):1061–1073. https://doi.org/10.1007/s00425-014-2236-6
Sharon N, Lis H (1972) Lectins: cell-agglutinating and sugar-specific proteins. Science 177:949–959. https://doi.org/10.1126/science.177.4053.949
Sharon N, Lis H (1989) Lectins as cell recognition molecules. Science 246:227–234
Sharon N, Lis H (2004) History of lectins: from hemagglutinins to biological recognition molecules. Glycobiology 14(11):53–62. https://doi.org/10.1093/glycob/cwh122
Shevchenko A, Tomas H, Havliˇs J, Olsen JV, Mann M (2007) In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat Protoc 1:2856–2860. https://doi.org/10.1038/nprot.2006.468
Sonntag VC (1987) The chemical basis of radiation biology. Taylor & Francis, London
Suseelan KN, Mitra R (2001) Purification and characterization of a hemagglutinin isolated from the leaves of chenopodium (Chenopodium amaranticolor). Indian J Biochem Biophys 38:186–192
Suseelan KN, Bhatia CR, Mitra R (1997) Purification and characterization of two major lectins from Vigna mungo (blackgram). J Biosci 22:439–455. https://doi.org/10.1007/BF02703190
Verma K, Shrivastava D, Kumar G (2015) Antioxidant activity and DNA damage inhibition in vitro by a methanolic extract of Carissa carandas (Apocynaceae) leaves. J Taibah Univ Sci 9:34–40. https://doi.org/10.1016/j.jtusci.2014.07.001
Villanueva AC, Ortega HC, Jafarova FA, Garfias Y et al (2007) Lectin from Phaseolus acutifolius var. escumite: chemical characterization, sugar specificity, and effect on human T-lymphocytes. J Agric Food Chem 55:5781–5787
Wakankar MS, Krishnasastry MV, Jaokar TM, Patel KA, Gaikwad SM (2013) Solution and in silico studies on the recombinant lectin from Cicer arietinum seeds. Int J Biol Macromol 56:149–155. https://doi.org/10.1016/j.ijbiomac.2013.02.015
Wang HX, Ng TB (2001) Examination of lectins, polysaccharo peptide, alkaloid, coumarin and trypsin inhibitor for inhibitory activity toward immunodeficiency virus reverse transcriptase and glycohydrolases. Planta Med 67:669–672. https://doi.org/10.1055/s-2001-17359
Wang Z, Chen M, Zhu Y, Qian P, Zhou Y, Wei J, Shen Y, Mijiti A, Gu A, Wang Z, Zhang H, Ma H (2017) Isolation, identification and characterization of a new type of lectin with α-amylase inhibitory activity in chickpea (Cicer arietinum L.). Protein Pept Lett 24(11):1008–1020. https://doi.org/10.2174/0929866524666170711120501)
Wong JH, Ng TB (2006) Isolation and characterization of a glucose/mannose-specific lectin with stimulatory effect on nitric oxide production by macrophages from the emperor banana. Int J Biochem Cell Biol 38:234–243. https://doi.org/10.1016/j.biocel.2005.09.004
Xu JG, Hu QP, Liu Y (2012) Antioxidant and DNA-protective activities of chlorogenic acid isomers. J Agric Food Chem 60:11625–11630. https://doi.org/10.1021/jf303771s
Ye XY, Ng TB (2002) Isolation of a new cyclophilin-like protein from chickpeas with mitogenic, antifungal and anti-HIV-1 reverse transcriptase activities. Life Sci 70:1129–1138
Zentian L, Li D, Qiao S, Zhu X, Huang C (2003) Anti-nutritional effects of a moderate dose of soybean agglutinin in the rat. Arch Anim Nutr 57:267–277. https://doi.org/10.1080/00039420310001594414
Acknowledgements
Authors are grateful to UGC, New Delhi for providing research funding (Grant No: 40–154/2011). Ajay Kumar Gautam in particular thankful to UGC for providing research fellowship. Thanks are also due to Dr. P.K. Dash, Head-Virology Division, DRDE, Gwalior for providing expertise. Providing the seed samples by Mahatma PhuleKrishiVidyapeeth, Rahuri, Maharashtra is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There is no conflict of interest.
Rights and permissions
About this article
Cite this article
Gautam, A.K., Srivastava, N., Nagar, D.P. et al. Biochemical and functional properties of a lectin purified from the seeds of Cicer arietinum L.. 3 Biotech 8, 272 (2018). https://doi.org/10.1007/s13205-018-1272-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1272-5