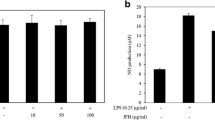
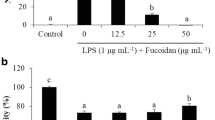
Abstract
Fish bones are the by-products of aquatic and fishery processing, which are often discarded. However, it has been considered having health-promoting by containing many essential nutrients. This study investigates the anti-inflammatory effect of fish bone fermented by Monascus purpureus (FBF) and the NF-κB pathway regulation mechanism in lipopolysaccharides (LPS)-induced RAW 264.7 cells. FBF has inhibited the production of PGE2 (prostaglandin E2), interleukin-6 (IL-6) and tumor necrosis factor-α (TNF-α) in LPS-induced RAW264.7 cells. The FBF has significantly inhibited mRNA expression of inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2). Moreover, FBF has suppressed activation of NF-κB (nuclear factor kappa-B) by increasing IκB mRNA expression and reduced of p65, p50 mRNA expression, as well as nuclear NF-κB DNA binding activity in LPS-induced RAW 246.7 cells. These findings demonstrate that FBF has inhibited LPS-induced inflammation by subsiding the activation of NF-κB in RAW 246.7 cells, implying that FBF could be employed as a promising natural product.





Similar content being viewed by others
Abbreviations
- LPS:
-
Lipopolysaccharides
- iNOS:
-
Inducible nitric oxide synthase
- PGE2 :
-
Prostaglandin E2
- COX-2:
-
Cyclooxygenase-2
- IL-6:
-
Interleukin-6
- TNF-α:
-
Tumor necrosis factor-α
- NF-κB:
-
Nuclear factor kappa-B
- DMEM:
-
Dulbecco’s Modified Eagle Medium
- PBS:
-
Phosphate-buffered saline
- MTT:
-
3-(4,5-Dimethyazol-2-yl)-2,5-diphenyltetrazolium bromide
- Real-time PCR:
-
Real-time reverse transcriptase polymerase chain reaction
References
Abdulazeez SS, Sundaram B, Ramamoorthy B, Ponnusamy P (2014) Production and antioxidant properties of protein hydrolysate from Rastrelliger kanagurta (Indian mackerel). Pak J Pharm Sci 27:1295–1302
Aktan F (2004) iNOS mediated nitric oxide production and its regulation. Life Sci 75:639–653. https://doi.org/10.1016/j.lfs.2003.10.042
Al Khawli F, Pateiro M, Domínguez R, Lorenzo JM, Gullón P, Kousoulaki K, Ferrer E, Berrada H, Barba FJ (2019) Innovative green technologies of intensification for valorization of seafood and their by-products. Mar Drugs 17:689. https://doi.org/10.3390/md17120689
Chen YT, Hsieh SL, Gao WS, Yin LJ, Dong CD, Chen CW, Singhania RR, Hsieh S, Chen SJ (2021) Evaluation of chemical compositions, antioxidant capacity and intracellular antioxidant action in fish bone fermented with Monascus purpureus. Molecules 26:5288. https://doi.org/10.3390/molecules26175288
Coppola D, Lauritano C, Palma Esposito F, Riccio G, Rizzo C, de Pascale D (2021) Fish waste: from problem to valuable resource. Mar Drugs 19:116. https://doi.org/10.3390/md19020116
Denizot F, Lang R (1986) Rapid colorimetric assay for cell growth and survival modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J Immunol Methods 89:271–277. https://doi.org/10.1016/0022-1759(86)90368-6
FAO (2020) Yearbooks of fisheries satistics food and agriculture organization of the United Nations. Rome
Flammini L, Martuzzi F, Vivo V, Ghirri A, Salomi E, Bignetti E, Barocelli E (2016) Hake fish bone as a calcium source for efficient bone mineralization. Int J Food Sci Nutr 67:265–273. https://doi.org/10.3109/09637486.2016.1150434
Hanada T, Yoshimura A (2002) Regulation of cytokine signaling and inflammation. Cytokine Growth Factor Rev 13:413–421. https://doi.org/10.1016/S1359-6101(02)00026-6
Hsu LC, Liang YH, Hsu YW, Kuo YH, Pan TM (2013) Anti-inflammatory properties of yellow and orange pigments from Monascus purpureus NTU 568. J Agric Food Chem 61:2796–2802. https://doi.org/10.1021/jf305521v
Hwang D, Kang K, Jo MJ, Seo YB, Park NG, Kim GD (2019) Anti-inflammatory activity of β-thymosin peptide derived from pacific oyster (Crassostrea gigas) on NO and PGE2 production by down-regulating NF-κB in LPS-induced RAW264.7 macrophage cells. Mar Drugs 17:129. https://doi.org/10.3390/md17020129
Jung KI, Kim BK, Kang JH, Oh GH, Kim IK, Kim M (2019) Anti-oxidant and anti-inflammatory activities of water and the fermentation liquid of sea tangle (Saccharina japonica). J Life Sci 29:596–606. https://doi.org/10.5352/JLS.2019.29.5.596
Kemp DC, Kwon JY (2021) Fish and shellfish-derived anti-inflammatory protein products: properties and mechanisms. Molecules 26:3225. https://doi.org/10.3390/molecules26113225
Kim YS, Shin WB, Dong X, Kim EK, Nawarathna WPAS, Kim H, Park PJ (2017) Anti-inflammatory effect of the extract from fermented Asterina pectinifera with Cordyceps militaris mycelia in LPS induced RAW264.7 macrophages. Food Sci Biotechnol 26:1633–1640. https://doi.org/10.1007/s10068-017-0233-9
Kim KH, Lee JH, Kwon GS, Seo EW, Lee JB (2020) Inhibitory effect of ethanol extract of Monascus fermented red yeast rice on proinflammatory iNOS and COX-2 protein expression in LPS stimulated RAW 264.7 macrophage cells. J Life Sci 30:352–358. https://doi.org/10.5352/JLS.2020.30.4.352
Kwon MS, Mun OJ, Bae MJ, Lee SG, Kim M, Lee SH, Yu KH, Kim YY, Kong CS (2015) Anti-inflammatory activity of ethanol extracts from Hizikia fusiformis fermented with lactic acid bacteria in LPS-stimulated RAW264. 7 macrophages. J Korean Soc Food Sci Nutr 44:1450–1457. https://doi.org/10.3746/jkfn.2015.44.10.1450
Li XM, Shen XH, Duan ZW, Guo SR (2011) Advances on the pharmacological effects of red yeast rice. Chin J Nat Med 9:161–166. https://doi.org/10.3724/SP.J.1009.2011.00161
Lin HTV, Lu WJ, Tsai GJ, Chou CT, Hsiao HI, Hwang PA (2016) Enhanced anti-inflammatory activity of brown seaweed Laminaria japonica by fermentation using Bacillus subtilis. Process Biochem 51:1945–1953. https://doi.org/10.1016/j.procbio.2016.08.024
Lowry OH, Rosebrough NJ, Farr AL, Ramdall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/S0021-9258(19)52451-6
Marti-Quijal FJ, Remize F, Meca G, Ferrer E, Ruiz MJ, Barba FJ (2020) Fermentation in fish and by-products processing: An overview of current research and future prospects. Curr Opin Food Sci 31:9–16. https://doi.org/10.1016/j.cofs.2019.08.001
Mun OJ, Kwon MS, Bae MJ, Ahn BN, Karadeniz F, Kim M, Lee SH, Yu KH, Kim YY, Seo Y, Kong CS (2015) Anti-inflammatory activity of Hizikia fusiformis extracts fermented with Lactobacillus casei in LPS-stimulated RAW 264.7 macrophages. Korean Soc Biotechnol Bioeng J 30:38–43. https://doi.org/10.7841/ksbbj.2015.30.1.38
Patel S (2016) Functional food red yeast rice (RYR) for metabolic syndrome amelioration: a review on pros and cons. World J Microbiol Biotechnol 32:87. https://doi.org/10.1007/s11274-016-2035-2
Pfeilschifter J, Köditz R, Pfohl M, Schatz H (2002) Changes in proinflammatory cytokine activity after menopause. Endocr Rev 23:90–119. https://doi.org/10.1210/edrv.23.1.0456
Sheriff SA, Sundaram B, Ramamoorthy B, Ponnusamy P (2014) Synthesis and in vitro antioxidant functions of protein hydrolysate from backbones of Rastrelliger kanagurta by proteolytic enzymes. Saudi J Biol Sci 21:19–26. https://doi.org/10.1016/j.sjbs.2013.04.009
Shi P, Liu M, Fan F, Yu C, Lu W, Du M (2018) Characterization of natural hydroxyapatite originated from fish bone and its biocompatibility with osteoblasts. Mater Sci Eng C Mater Biol Appl 90:706–712. https://doi.org/10.1016/j.msec.2018.04.026
Surh YJ, Chun KS, Cha HH, Han SS, Keum YS, Park KK, Lee SS (2001) Molecular mechanism underlying chemopreventive activities of anti-inflammatory phytochemicals: downregulation of COX-2 and iNOS through suppression of NF-kappa B activation. Mutat Res 480–481:243–268. https://doi.org/10.1016/S0027-5107(01)00183-X
Tang S, Dong S, Chen M, Gao R, Chen S, Zhao Y, Liua Z, Sun B (2018) Preparation of a fermentation solution of grass fish bones and its calcium bioavailability in rats. Food Funct 9:4135–4142. https://doi.org/10.1039/C8FO00674A
Tseng KC, Fang TJ, Chiang SS, Liu CF, Wu CL, Pan TM (2012) Immunomodulatory activities and antioxidant properties of polysaccharides from Monascus-fermented products in vitro. J Sci Food Agric 92:1483–1489. https://doi.org/10.1002/jsfa.4731
Vane JR, Bakhle YS, Botting RM (1998) Cyclooxygenases 1 and 2. Annu Rev Pharmacol Toxicol 38:97–120. https://doi.org/10.1146/annurev.pharmtox.38.1.97
Yin T, Park JW (2014) Effects of nano-scaled fish bone on the gelation properties of Alaska pollock surimi. Food Chem 150:463–468. https://doi.org/10.1016/j.foodchem.2013.11.041
Acknowledgements
The authors are grateful to appreciate the invaluable support of the Ministry of Science and Technology of Taiwan in this study
Funding
This study was funded with the support of the Ministry of Science and Technology of Taiwan (grant number MOST 109-2622-E-992-028-).
Author information
Authors and Affiliations
Contributions
Conceptualization, Y-TC and S-LH; methodology, Y-TC; validation, Y-TC and S-JC; formal analysis, L-JY; investigation, Y-TC, S-JC and C-YH; resources, L-JY; data curation, Y-TC and S-LH; writing—original draft preparation, Y-TC; writing—review and editing, S-LH and R-RS; supervision, C-DD and S-LH; project administration, S-LH All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Ethics approval
The work has not been published before and it is not under consideration for publication elsewhere.
Consent to participate
The MS has been approved by all authors.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, YT., Chen, SJ., Yin, LJ. et al. Anti-inflammatory effects of fish bone fermented using Monascus purpureus in LPS-induced RAW264.7 cells by regulating NF-κB pathway. J Food Sci Technol 60, 958–965 (2023). https://doi.org/10.1007/s13197-022-05413-4
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-022-05413-4