Abstract
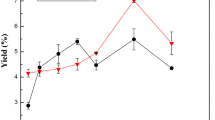
Bovine skin was incubated with plant enzymes bromelain (B) and zingibain (Z) at the level of 0, 5, 10, 15, 20 and 25 unit/g of skin and gelatin was extracted at 60 °C for 6 h. Control gelatin was extracted without enzymatic pretreatment. The yield and gel strength were 17.90% and 283.35 g for the control samples and 22.26% and 160.88 g for B20 samples. The zingibain extracted gelatin (GEZ) samples failed to form gel. Viscosities of GEZ gelatins were significantly (P < 0.05) lower than the gelatins extracted using bromelain (GEB). β and α chains were absolutely degraded in all GEB and GEZ samples. Only smear bands were observed in GEZ gelatins whereas GEB samples revealed presence of low molecular weight polypeptides. Loss of molecular order was noticed in Z5 as elaborated by Fourier transform infrared (FTIR) spectroscopy. Larger particle size, denser and inter-connected irregular network was observed in B20 under scanning electron microscopy. Based on the results obtained, bromelain, particularly at level 20, could be used to obtain a better quality gelatin with higher yield compared to zingibain.



Similar content being viewed by others
References
Ahmad M, Benjakul S (2011) Characteristics of gelatin from the skin of unicorn leatherjacket (Aluterus monoceros) as influenced by acid pretreatment and extraction time. Food Hydrocoll 25(3):381–388
Ahmad M, Benjakul S, Ovissipour M, Prodpran T (2011) Indigenous proteases in the skin of unicorn leatherjacket (Aluterus monoceros) and their influence on characteristic and functional properties of gelatin. Food Chem 127(2):508–515
Ahmad T, Ismail A, Ahmad SA, Khalil KA, Kumar Y, Adeyemi KD, Sazili AQ (2017) Recent advances on the role of process variables affecting gelatin yield and characteristics with special reference to enzymatic extraction: a review. Food Hydrocoll 63:85–96
Ahmad T, Ismail A, Ahmad SA, Khalil KA, Teik Kee L, Awad EA, Sazili AQ (2019) Physicochemical characteristics and molecular structures of gelatin extracted from bovine skin: effects of actinidin and papain enzymes pretreatment. Int J Food Prop 22(1):138–153
Badii F, Howell NK (2006) Fish gelatin: structure, gelling properties and interaction with egg albumen proteins. Food Hydrocoll 20(5):630–640
Balti R, Jridi M, Sila A, Souissi N, Nedjar-Arroume N, Guillochon D, Nasri M (2011) Extraction and functional properties of gelatin from the skin of cuttlefish (Sepia officinalis) using smooth hound crude acid protease-aided process. Food Hydrocoll 25(5):943–950
Chomarat N, Robert L, Seris JL, Kern P (1994) Comparative efficiency of pepsin and proctase for the preparation of bovine skin gelatin. Enzyme Microb Technol 16(9):756–760
Damrongsakkul S, Ratanathammapan K, Komolpis K, Tanthapanichakoon W (2008) Enzymatic hydrolysis of rawhide using papain and neutrase. J Ind Eng Chem 14(2):202–206
Duconseille A, Astruc T, Quintana N, Meersman F, Sante-Lhoutellier V (2015) Gelatin structure and composition linked to hard capsule dissolution: a review. Food Hydrocoll 43:360–376
Fan H, Dumont MJ, Simpson BK (2017) Extraction of gelatin from salmon (Salmo salar) fish skin using trypsin-aided process: optimization by Plackett–Burman and response surface methodological approaches. J Food Sci Technol 54(12):4000–4008
Fernandez-Dıaz MD, Montero P, Gomez-Guillen MC (2001) Gel properties of collagens from skins of cod (Gadus morhua) and hake (Merluccius merluccius) and their modification by the coenhancers magnesium sulphate, glycerol and transglutaminase. Food Chem 74(2):161–167
Giménez B, Turnay J, Lizarbe MA, Montero P, Gómez-Guillén MC (2005) Use of lactic acid for extraction of fish skin gelatin. Food Hydrocoll 19(6):941–950
GME-Gelatin Manufacturers of Europe (2020) Gelatine is indispensable for the food industry and for nutrition. https://www.gelatine.org/applications/food-industry.html. Accessed 04 Mar 2020
Gómez-Guillén MC, Turnay J, Fernández-Dıaz MD, Ulmo N, Lizarbe MA, Montero P (2002) Structural and physical properties of gelatin extracted from different marine species: a comparative study. Food Hydrocoll 16(1):25–34
Ha M, Bekhit AE-DA, Carne A, Hopkins DL (2012) Characterisation of commercial papain, bromelain, actinidin and zingibain protease preparations and their activities toward meat proteins. Food Chem 134(1):95–105
Hafidz RMRN, Yaakob CM, Amin I, Noorfaizan A (2011) Chemical and functional properties of bovine and porcine skin gelatin. Int Food Res J 18:813–817
Jamilah B, Tan KW, Umi Hartina MR, Azizah A (2011) Gelatins from three cultured freshwater fish skins obtained by liming process. Food Hydrocoll 25(5):1256–1260
Karim AA, Bhat R (2008) Gelatin alternatives for the food industry: recent developments, challenges and prospects. Trends Food Sci Technol 19:644–656
Ktari N, Bkhairia I, Jridi M, Hamza I, Riadh BS, Nasri M (2014) Digestive acid protease from zebra blenny (Salaria basilisca): characteristics and application in gelatin extraction. Food Res Int 57:218–224
Lassoued I, Jridi M, Nasri R, Dammak A, Hajji M, Nasri M, Barkia A (2014) Characteristics and functional properties of gelatin from thornback ray skin obtained by pepsin-aided process in comparison with commercial halal bovine gelatin. Food Hydrocoll 41:309–318
Ledward DA (1986) Gelation of gelatin. In: Mitchell JR, Ledward DA (eds) Functional properties of food macromolecules. Elsevier Applied Science Publishers, London, pp 171–201
Mariod AA, Adam HF (2013) Review: gelatin, source, extraction and industrial. Acta Sci Pol Technol Aliment 12(2):135–147
Mohtar NF, Perera C, Quek S-Y (2010) Optimisation of gelatine extraction from hoki (Macruronus novaezelandiae) skins and measurement of gel strength and SDS-PAGE. Food Chem 122(1):307–313
Muyonga JH, Cole CGB, Duodu KG (2004) Fourier transform infrared (FTIR) spectroscopic study of acid soluble collagen and gelatin from skins and bones of young and adult Nile perch (Lates niloticus). Food Chem 86(3):325–332
Nalinanon S, Benjakul S, Visessanguan W, Kishimura H (2008) Improvement of gelatin extraction from bigeye snapper skin using pepsin-aided process in combination with protease inhibitor. Food Hydrocoll 22(4):615–622
Nishimoto M, Sakamoto R, Mizuta S, Yoshinaka R (2005) Identification and characterization of molecular species of collagen in ordinary muscle and skin of the Japanese flounder Paralichthys olivaceus. J Food Chem 90:151–156
Norziah MH, Kee HY, Norita M (2014) Response surface optimization of bromelain-assisted gelatin extraction from surimi processing wastes. Food Biosci 5:9–18
Pitpreecha S (2005) Gelatin production from large animal raw hide using proteolytic enzyme extracted from papaya latex. Mater thesis, Department of Chemical Engineering, Faculty of Engineering, Chulalongkorn University, Thailand, ISBN:974-53-2940-1
Pitpreecha S, Damrongsakkul S (2006) Hydrolysis of raw hide using proteolytic enzyme extracted from papaya latex. Korean J Chem Eng 23(6):972–976
Simeonova L, Dalev P (1996) Utilization of a leather industry waste. Waste Manag 16(8):765–769
Sinthusamran S, Benjakul S, Kishimura H (2014) Characteristics and gel properties of gelatin from skin of seabass (Lates calcarifer) as influenced by extraction conditions. Food Chem 152:276–284
Statista (2020a) Pineapple production worldwide from 2002 to 2018. https://www.statista.com/statistics/298505/global-pineapple-production/. Accessed 06 Mar 2020
Statista (2020b) Production of ginger worldwide from 2010 to 2017. https://www.statista.com/statistics/1064660/ginger-production-volume-worldwide/. Accessed 06 Mar 2020
Tu Z-C, Huang T, Wang H, Sha X-m, Shi Y, Huang X-q, Man Z-z, Li D-j (2015) Physico-chemical properties of gelatin from bighead carp (Hypophthalmichthys nobilis) scales by ultrasound-assisted extraction. J Food Sci Technol 52(4):2166
Tyburcy A, Wasiak P, Cegiełka A (2010) Application of composite protective coatings on the surface of sausages with different water content. Acta Sci Pol Technol Aliment 9(2):151–159
Zhou P, Regenstein JM (2006) Determination of total protein content in gelatin solutions with the Lowry or Biuret assay. J Food Sci 71(8):C474–C479
Acknowledgements
The first author is obliged to ICAR (Indian Council of Agricultural Research) (Grant no. F. No. 29-1/2009-EQR/Edn (pt.III)), New Delhi, India for awarding ICAR-International Fellowship and Department of Agricultural Research & Education (DARE), Ministry of Agriculture and Farmers Welfare, Government of India, New Delhi for granting him permission to pursue Ph.D. in Malaysia. Biohawk, QLD, Australia is duly recognized for gifting enzyme zingibain. The research work was supported by Putra Grant (no. UPM/700-2/1/GP-IPS/2015/9467000) given by Universiti Putra Malaysia, Malaysia.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ahmad, T., Ismail, A., Ahmad, S.A. et al. Extraction, characterization and molecular structure of bovine skin gelatin extracted with plant enzymes bromelain and zingibain. J Food Sci Technol 57, 3772–3781 (2020). https://doi.org/10.1007/s13197-020-04409-2
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04409-2