Abstract
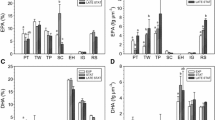
Twelve indigenous microalgae, comprising of three fresh water and nine marine strains, were evaluated for their potential as source of dietary polyunsaturated fatty acids (PUFAs) under autotrophic growth conditions. The microalgal lipids showed higher content of omega-3 PUFAs as reflected in lower omega-6 to omega-3 ratios between 0.1 and 0.75. Alpha linolenic acid (ALA), was the predominant omega-3 PUFA in fresh water green algal strains. Scenedesmus dimorphus and Chlorococcum sp. showed higher content of ALA with an average productivity of 3 mgL−1 day−1. Long chain omega-3 PUFAs, eicosapentaenoic (EPA) and docosahexaenoic acid (DHA) were observed in marine strains. Nannochloropsis and Chlorella sp. had higher content of EPA with Chlorella sp. showing an average productivity of 2 mgL−1 day−1. Isochrysis sp. showed higher DHA content with average productivity of 0.37 mgL−1 day−1. Presence of EPA and DHA contributed to higher degree of unsaturation in lipids of marine strains. Among the marine strains, the growth and lipid profile of Nannochloropsis sp. and Chlorella sp. remained unaffected by growth medium, whereas strains like T. theli showed a differential response to media. Spirulina platensis SP6 from CFTRI algal culture repository was also included as a reference strain since its nutritional benefits have been well elucidated. Gamma Linolenic acid (GLA) was the predominant PUFA in Spirulina sp. with an average productivity of 0.73 mgL−1 day−1.



Similar content being viewed by others
References
Becker W (2004) Microalgae in human and animal nutrition. In: Richmond A (ed) Microalgae for aquaculture in handbook of microalgal culture: biotechnology and applied phycology. Blackwell Science, Iowa, pp 312–352
Berges JA, Franklin DJ, Harrison PJ (2001) Evolution of an artificial sea water medium: improvements in enriched sea water, artificial water, over the last two decades. J Phycol 37:1138–1145
Borowitzka MA (1999) Commercial production of microalgae: ponds, tanks, tubes and fermenters. J Biotechnol 70:313–321
Brett M, Muller-Navarra D (1997) The role of highly unsaturated fatty acids in aquatic food web processes. Freshwater Biol 38:483–499
Christie WW (1982) Lipid analysis, 2nd edn. Pergamon press, New York, pp 93–96
Cohen Z, Ratledge C (2005) Single cell oils. AOCS Press, Illinois, Chapter 4
Deng R, Chow TJ (2010) Hypolipidemic, antioxidant, and anti-inflammatory activities of microalgae Spirulina. Cardiovasc Ther 28:33–45
Doughman SD, Krupanidhi S, Sanjeevi CB (2007) Omega-3 fatty acids for nutrition and medicine: considering microalgae oil as a vegetarian source of EPA and DHA. Curr Diabetes Rev 3:198–203
FAO Fisheries and Aquaculture Technical Paper No. 569 (2011) Review of the state of world marine fishery resources. FAO, Rome
Floreto EAT, Teshima S (1998) The fatty acid composition of seaweeds exposed to different levels of light intensity and salinity. Bot Mar 41:467–481
Fogliano V, Andreoli C, Martello A, Caiazzo M, Lobosco O, Formisano F, Carlino PA, Meca G, Graziani G, Rigano VM, Vona V, Carfagna S, Rigano C (2010) Functional ingredients produced by culture of Koliella antarctica. Aquacult 299:115–120
Fraeye I, Bruneel C, Lemahieu C, Buyse J, Muylaert K, Foubert I (2012) Dietary enrichment of eggs with omega-3 fatty acids: a review. Food Res Int 48:961–969
Guedes AC, Amaro HM, Barbosa CR, Pereira RD, Malcata FX (2011) Fatty acid composition of several wild microalgae and cyanobacteria, with a focus on eicosapentaenoic, docosahexaenoic and α-linolenic acids for eventual dietary uses. Food Res Int 44:2721–2729
Guschina IA, Harwood JL (2009) Algal lipids and effect of the environment on their biochemistry. In: Arts MT, Brett MT, Kainz M (eds) Lipids in aquatic ecosystems. Springer, New York, pp 1–24
Harwood JL, Jones AL (1989) Lipid metabolism in algae. Adv Bot Res 16:1–53
Hempel N, Petrick I, Behrendt F (2012) Biomass productivity and productivity of fatty acids and amino acids of microalgae strains as key characteristics of suitability for biodiesel production. J Appl Phycol 24:1407–1418
Jorquera O, Kiperstok A, Sales EA, Embirucu M, Ghirardi ML (2010) Comparative energy life-cycle analyses of microalgal biomass production in open ponds and photobioreactors. Bioresour Technol 101:1406–1413
Kanz T, Bold HC (1969) Morphological and taxonomical investigation of Nostoc and Anabaena in culture. In: physiological studies. University of Texas, Texas, Austin; publication No. 6924
Lichtenstein AH, Appel LJ, Brands M, Carnethon M, Daniels S, Franch HA (2006) Diet and lifestyle recommendations revision 2006 — a scientific statement from the American heart association nutrition committee. Circulation 114:82–96
Melanson SF, Lewandrowski EL, Lewandrowski KB (2005) Measurement of organochlorines in commercial over-the-counter fish oil preparations. Arch Pathol Lab Med 129:74–77
Rittmann EB (2008) Opportunities for renewable bioenergy using microorganisms. Biotechnol Bioeng 100:203–212
Sahu A, Pancha I, Jain D, Paliwal C, Ghosh T, Shailesh P, Bhattacharya S, Mishra S (2013) Fatty acids as biomarkers of algae. Phytochem 89:53–58
Simopoulos AP (2002) The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed Pharmacother 56:365–379
Simopoulos AP (2008) The importance of the omega-6/omega-3 fatty acid ratio in cardiovascular disease and other chronic diseases. Exp Biol Med 233:674–688
Storelli MM, Storelli A, Marcotrigiano GO (2004) Polychlorinated biphenyls, hexa chloro benzene, hexachloro cyclohexane isomers, and pesticide organo chlorine residues in cod-liver oil dietary supplements. J Food Protect 67:1787–1791
Teoh M, Chu W, Merchant H, Phang S (2004) Influence of culture temperature on the growth, biochemical compositions, and fatty acid profiles of six Antarctic microalgae. J Appl Phycol 16:421–430
Thompson PAM, Guo M, Harrison PJ (1992) Effects of variation in temperature on the fatty acid composition of eight species of marine phytoplankton. J Phycol 28:488–497
UTEX - The Culture collection of Algae. Artificial Sea Water Medium. http://web.biosci.utexas.edu/utex/mediaDetail.aspx?mediaID=23
Vidyashankar S, Deviprasad K, Chauhan VS, Ravishankar GA, Sarada R (2013) Selection and evaluation of CO2 tolerant indigenous microalga Scenedesmus dimorphus for unsaturated fatty acid rich lipid production under different culture conditions. Bioresour Technol 144:28–37
Wen ZY, Chen F (2003) Heterotrophic production of eicosapentaenoic acid by microalgae. Biotechnol Adv 21:273–294
Zarrouk C (1966) Contribution à l’étuded’unecyanophycée. Influence de divers’ facteurs physiques etchimiquessur la croissance et la photosynthèse de Spirulina maxima. Ph.D. Thesis, Université de Paris, Paris
Acknowledgments
Authors thank Dr. N. Bhaskar, Meat & Marine Sciences department, CSIR-CFTRI and Dr. Sugumar Gopalrajan, FCRI, Tuticorin for the help in procurement of marine microalgae strains. VS acknowledges the UGC, Govt. of India for the award of Senior Research Fellowship. Authors thank Director, CSIR-CFTRI for constant encouragement.
Author information
Authors and Affiliations
Corresponding author
Additional information
Research highlights
• 12 microalgae strains were evaluated as source of dietary PUFAs in autotrophic growth.
• Microalgal lipids had higher ratio of n-3 PUFAs as reflected by lower n-6 to n-3 ratio (0.1 to 0.75).
• ALA was predominant n-3 PUFA in freshwater while EPA was major in marine strains.
• Degree of unsaturation of PUFAs was higher in marine strains.
Rights and permissions
About this article
Cite this article
Vidyashankar, S., Sireesha, E., Chauhan, V.S. et al. Evaluation of microalgae as vegetarian source of dietary polyunsaturated fatty acids under autotrophic growth conditions. J Food Sci Technol 52, 7070–7080 (2015). https://doi.org/10.1007/s13197-015-1781-8
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-015-1781-8