Abstract
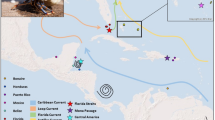
The nylon shrimp Heterocarpus reedi is the most important commercial demersal crustacean species along the central coast of Chile in the southeast Pacific, with an intense fishery since the 1950s. It inhabits between 155 and 800 m of depth and is harvested along most of its distributional range (21°S to 39°S) by bottom trawling. Current management policy considers a single population unit on the lower continental shelf margin and upper slope along the exploitation area. The goal of this study was to assess the genetic diversity and differentiation of H. reedi along the geographic area of exploitation, representing the first insights into population genetics of a demersal crustacean from Chile. We analyzed 446 partial sequences of the mitochondrial COI gene from individuals captured using bottom trawls from 14 sites separated by 1° of latitude, covering the exploitation area and bathymetric range of the benthic adults. Data showed high genetic diversity despite an extensive fishing pressure and absence of genetic differentiation among sites. Haplotype diversity showed a negative correlation with depth of the trawling, consistent with lower abundance of individuals at depths greater than 400 m. Also, a high effective population size and a post-LGM demographic expansion was detected. Lack of spatial and bathymetrical genetic differentiation and extensive gene flow reflect that the species is composed of a single panmictic unit. Phylogeographic structure suggests considering latitude, depth, and temporal variation in future assessments of the species.




Similar content being viewed by others
Data availability
Sequence data can be retrieved from GenBank Accession Numbers OK647412 - OK647622.
References
Acuña, E., Alarcón, R., Arancibia, H., Barros, M., Cid, L., Cortés, A., Cubillos, L., & Pineda, J. (2009). Evaluación directa de camarón nailon entre la II y VIII Regiones, año 2008, FIP Nº 2008–17. Final Report FIP 2008–17 (p. 274). Retrieved October 15, 2020, from https://www.subpesca.cl/fipa/613/w3-article-89228.html
Acuña, E., Alarcón, R., Arancibia, H., Cid, L., Cortés, A., & Cubillos, L. (2012). Evaluación directa de camarón nailon entre la II y VIII regiones, año 2009. Final Report FIP 2009–16 (p. 279). Retrieved October 15, 2020, from https://www.subpesca.cl/fipa/613/w3-article-89282.html
Acuña, E., Alarcón, R., Cortés, A., Arancibia, H., Haye, P. A, Segovia, N., & Petitgas, P. (2019). Evaluación directa de camarón nailon entre la II y VIII Regiones, año 2018. Final Report IFOP 2018–2019 (p. 296). Retrieved September 20, 2020, from https://www.ifop.cl/wp-content/contenidos/uploads/RepositorioIfop/InformeFinal/2019/P-682058.pdf
Acuña, E., Arancibia, H., Roa, R., Alarcón, R., Díaz, C., Mujica, A., Winkler, F. Lépez, I., & Cid, L. (1997). Análisis de la pesquería y evaluación indirecta del stock de camarón nailon (II a VIII regiones). Final Report FIP-IT 95–06 (p. 186). Retrieved September 20, 2020, from https://www.subpesca.cl/fipa/613/w3-article-89510.html
Acuña, E., González, M. T., & González, M. (2003). Pesquerías de langostinos y camarón nailon en el Norte de Chile, 263–287. In E. Yáñez (Ed.), Actividad Pesquera y de Acuicultura en Chile. Pontificia Universidad Católica de Valparaíso. Escuela de Ciencias del Mar.
Alam, M. M., Westfall, K. M., & Palsson, S. (2015). Mitochondrial DNA variation reveals cryptic species in Fenneropenaeus indicus. Bulletin of Marine Science, 91, 15–31. https://doi.org/10.5343/bms.2014.1036
Allendorf, F. W., England, P. R., Luikart, G., Ritchie, P. A., & Ryman, N. (2008). Genetic effects of harvest on wild animal populations. Trends in Ecology and Evolution, 23, 327–337. https://doi.org/10.1016/j.tree.2008.02.008
Arana, P., Melo, T., Noziglia, L., Sepúlveda, I., Silva, N., Yany, G., & Yánez, E. (1975). Los recursos demersales de la región de Valparaíso. Revista de la Comisión Permanente del Pacífico Sur, 3, 39–61.
Arana, P., & Nakamishi, A. (1971). La pesquería del camarón nailon (Heterocarpus reedi) frente a la costa de Valparaíso. Investigaciones Marinas, Valparaíso, 2, 61–92.
Arana, P., Noziglia, L., & Yany, G. (1976). Crecimiento, reproducción, factor de condición y estructura poblacional del camarón nylon (Heterocarpus reedi) (Crustacea, Decapoda, Caridea). Ciencia y Tecnología del Mar, 2, 3–98.
Aschan, M., & Ingvaldsen, R. (2009). Recruitment of shrimp (Pandalus borealis) in the Barents Sea related to spawning stock and environment. Deep-Sea Research II Topical Studies in Oceanography, 56, 2012–2022. https://doi.org/10.1016/j.dsr2.2008.11.012
Atencia-Galindo, M. A., Narvaéz, J. C., Ramírez, A., Paramo, J., & Aguire-Pabón, J. C. (2021). Genetic structure of the pink shrimp Penaeus (Farfantepenaeus) notialis (Péres-Farfante, 1967) (Decapoda: Penaeidae) in the Colombian Caribbean. Fisheries Research, 243, 106052. https://doi.org/10.1016/j.fishres.2021.106052
Azuma, N., & Chiba, S. (2017). Genetic population structure of the Hokkai shrimp Pandalus latirostris Rathbun, 1902 (Decapoda: Caridea: Pandalidae) from Zostera seagrass beds in Hakkaido, Japan. Journal of Crustacean Biology, 38, 1–9. https://doi.org/10.1093/jcbiol/rux116
Ball, A. O., & Chapman, R. W. (2003). Population genetic analysis of white shrimp, Litopenaeus setiferus, using microsatellite genetic markers. Molecular Ecology, 12, 2319–2330. https://doi.org/10.1046/j.1365-294x.2003.01922.x
Barbieri, M. A., Canales, C., Leiva, B., Bahamonde, R., Peñailillo, T., Pool, H., & Montenegro, C. (2001). Evaluación directa de langostino colorado de la I a IV Regiones, 1999. Final Report FIP 99–30 (p. 170). Retrieved September 20, 2020, from https://www.subpesca.cl/fipa/613/articles-89668_informe_final.pdf
Beerli, P., & Palczewski, M. (2010). Unified framework to evaluate panmixia and migration direction among multiple sampling locations. Genetics, 185, 313–326. https://doi.org/10.1534/genetics.109.112532
Blanco, J. L., Thomas, A. C., Carr, M. E., & Strub, P. T. (2001). Seasonal climatology of hydrographic conditions in the upwelling region off northern Chile. Journal of Geophysical Research: Oceans, 106(451–11), 467. https://doi.org/10.1029/2000JC000540
Borrell, Y. J., Arenal, F., Mbemba, Z. M., Santana, O., Díaz-Fernández, R., Vázquez, E., Blanco, G., Sánchez, J. A., & Espinosa, G. (2007). Spatial and temporal genetic analysis of the Cuban white shrimp Penaeus (Litopenaeus) schmitti. Aquaculture, 272, S125–S138. https://doi.org/10.1016/j.aquaculture.2007.08.015
Bouzat, J. L. (2010). Conservation genetics of population bottlenecks: the role of chance, selection, and history. Conservation Genetics, 11, 463–478. https://doi.org/10.1007/s10592-010-0049-0
Bradbury, I. R., Laurel, B., Snelgrove, P. V., Bentzen, P., & Campana, S. E. (2008). Global patterns in marine dispersal estimates: the influence of geography, taxonomic category and life history. Proceedings of the Royal Society of London b: Biological Sciences, 275, 1803–1809. https://doi.org/10.1098/rspb.2008.0216
Canales, C. M., Company, J. B., & Arana, P. M. (2016). Population structure of nylon shrimp Heterocarpus reedi (Crustacea: Caridea) and its relationship with environmental variables off Chile. Latin American Journal of Aquatic Research, 44, 144–154. https://doi.org/10.3856/vol44-issue1-fulltext-15
Cannas, R., Sacco, F., Follesa, M. C., Sabatini, A., Arculeo, M., Lo Brutto, S., Maggio, T., Deiana, A. M., & Cau, A. (2012). Genetic variability of the blue and red shrimp Aristeus antennatus in the Western Mediterranean Sea inferred by DNA microsatellite loci. Marine Ecology, 33, 350–363. https://doi.org/10.1111/j.1439-0485.2011.00504.x
Cárdenas, L., Castilla, J. C., & Viard, F. (2009). A phylogeographical analysis across three biogeographical provinces of the south-eastern Pacific: the case of the marine gastropod Concholepas concholepas. Journal of Biogeography, 36, 969–981. https://doi.org/10.1111/j.1365-2699.2008.02056.x
Chase, J. M., Kraft, N. J., Smith, K. G., Vellend, M., & Inouye, B. D. (2011). Using null models to disentangle variation in community dissimilarity from variation in α−diversity. Ecosphere, 2, 1–11. https://doi.org/10.1890/ES10-00117.1
Chu, T.-J., Wang, D., Huang, H.-L., Lin, F.-J., & Tzeng, T.-D. (2012). Population structure and historical demography of the whiskered velvet shrimp (Metapenaeopsis barbata) off China and Taiwan inferred from the mitochondrial control region. Zoological Studies, 51, 99–107.
Cowen, R. K., & Sponaugle, S. (2009). Larval dispersal and marine population connectivity. Annual Review of Marine Science, 1, 443–466. https://doi.org/10.1146/annurev.marine.010908.163757
Darriba, D., Taboada, G. L., Doallo, R., & Posada, D. (2012). jModelTest 2: More models, new heuristics and parallel computing. Nature Methods, 9, 772–772. https://doi.org/10.1038/nmeth.2109
De Croos, M., & Pálsson, S. (2012). Population biology and genetic diversity of two adjacent shrimp (Parapenaeopsis coromandelica) populations exploited under different fishing pressures in the coastal waters of Sri Lanka. Journal of the Marine Association of the UK, 92, 819–829. https://doi.org/10.1017/S0025315411000828
Drummond, A. J., Ashton, B., Cheung, M., Heled, J., Kearse, M., Moir, R., & Wilson, A. (2009). Geneious v4.7. http://www.geneious.com
Drummond, A. J., & Rambaut, A. (2007). BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evolutionary Biology, 7, 214. https://doi.org/10.1186/1471-2148-7-214
Drummond, A. J., Suchard, M. A., Xie, D., & Rambaut, A. (2012). Bayesian phylogenetics with BEAUti and the BEAST 1.7. Molecular Biology and Evolution, 29, 1969–1973. https://doi.org/10.1093/molbev/mss075
Dupanloup, I., Schneider, S., & Excoffier, L. (2002). A simulated annealing approach to define the genetic structure of populations. Molecular Ecology, 11, 2571–2581. https://doi.org/10.1046/j.1365-294x.2002.01650.x
Excoffier, L., & Lischer, H. E. (2010). Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Molecular Ecology Resources, 10, 564–567. https://doi.org/10.1111/j.1755-0998.2010.02847.x
Fernández, M. V., Heras, S., Vinas, J., Maltagliati, F., & Roldán, M. I. (2014). Multilocus comparative phylogeography of two aristeid shrimps of high commercial interest (Aristeus antennatus and Aristaeomorpha foliacea) reveals different responses to past environmental changes. PLoS One, 8, e59033. https://doi.org/10.1371/journal.pone.0059033
Folmer, O., Black, M., Hoeh, W., Lutz, R., & Vrijemhoek, R. (1994). DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Molecular Marine Biology and Biotechnology, 3, 294–299.
Fonseca, T., & Farías, M. (1987). Estudio del proceso de surgencia en la costa chilena utilizando percepción remota. Investigaciones Pesqueras, 34, 33–46.
Fratini, S., Ragionieri, L., Deli, T., Harrer, A., Marino, I. A. M., Cannicci, S., Zane, L., & Schubart, C. D. (2016). Unravelling population genetic structure with mitochondrial DNA in a notional panmictic coastal crab species: Sample size makes the difference. BMC Evolutionary Biology, 16, 150. https://doi.org/10.1186/s12862-016-0720-2
Fu, Y., & Li, W. (1993). Statistical tests of neutrality of mutations. Genetics, 133, 693–709.
Haye, P. A., & Muñoz-Herrera, N. C. (2013). Isolation with differentiation followed by expansion with admixture in the tunicate Pyura chilensis. BMC Evolutionary Biology, 13, 252. https://doi.org/10.1186/1471-2148-13-252
Haye, P. A., Segovia, N. I., Muñoz-Herrera, N. C., Gálvez, F. E., Martínez, A., Meynard, A., Pardo-Gandarillas, M. C., Poulin, E., & Faugeron, S. (2014). Phylogeographic structure in benthic marine invertebrates of the southeast pacific coast of Chile with differing dispersal potential. PLoS One, 9, e88613. https://doi.org/10.1371/journal.pone.0088613
Haye, P. A., Segovia, N. I., Varela, A. I., Rojas, R., Rivadeneira, M. M., & Thiel, M. (2019). Genetic and morphological divergence at a biogeographic break in the beach-dwelling brooder Excirolana hirsuticauda Menzies (Crustacea, Peracarida). BMC Evolutionary Biology, 19, 118. https://doi.org/10.1186/s12862-019-1442-z
Hedgecock, D., Barber, P. H., & Edmands, S. (2007). Genetic approaches to measuring connectivity. Oceanography, 20, 70–79. https://doi.org/10.5670/oceanog.2007.30
Heras, S., Planella, L., García-Marín, J.-L., Vera, M., & Roldán, M. I. (2019). Genetic structure and population connectivity of the blue and red shrimp Aristeus antennatus. Scientific Reports, 9, 13531. https://doi.org/10.1038/s41598-019-49958-5
Hauser, L., Adcock, G. J., Smith, P. J., Bernal Ramírez, J. H., & Carvalho, G. R. (2002). Loss of microsatellite diversity and low effective population size in an overexploited population of New Zealand snapper (Pagurus auratus). Proceedings of the National Academy of Sciences of the USA, 99, 11742–11747. https://doi.org/10.1073/pnas.172242899
Ho, S., Lanfear, R., Bromham, L., Phillips, M., Soubrier, J., Rodrigo, A., & Cooper, A. (2011). Time-dependent rates of molecular evolution. Molecular Ecology, 20, 3087–3101. https://doi.org/10.1111/j.1365-294X.2011.05178.x
Ho, S., Phillips, M., Cooper, A., & Drummond, A. (2005). Time dependency of molecular rate estimates and systematic overestimation of recent divergence times. Molecular Biology and Evolution, 22, 1561–1568. https://doi.org/10.1093/molbev/msi145
Ho, S., Shapiro, B., Phillips, M., Cooper, A., & Drummond, A. (2007). Evidence for time dependency of molecular rates. Systematic Biology, 56, 515–522. https://doi.org/10.1080/10635150701435401
Hoffman, J. I., Peck, L. S., Linse, K., & Clarke, A. (2010). Strong population genetic structure in a Broadcast-spawning Antarctic marine invertebrate. Journal of Heredity, 102, 55–56. https://doi.org/10.1093/jhered/esq094
Hudson, R. R. (2000). A new statistic for detecting genetic differentiation. Genetics, 155, 2001–2014.
Hulton, N. R. J., Purves, R. S., McCulloch, R. D., Sugden, D. E., & Bentley, M. J. (2002). The last glacial maximum and deglaciation in southern South America. Quaternary Science Reviews, 21, 233–241. https://doi.org/10.1016/S0277-3791(01)00103-2
Jiang, G. C., Chan, T. Y., & Shih, T. W. (2014). Morphology of the first zoeal stage of three deep-water pandalid shrimps, Heterocarpus abulbus Yang, Chan & Chu, 2010, H. hayashii Crosnier, 1988 and H. sibogae De Man, 1917 (Crustacea: Decapoda: Caridea). Zootaxa, 3768, 428–436. https://doi.org/10.11646/zootaxa.3768.4.2
Jiang, G. C., Landeira, J. M., Shih, T. W., & Chan, T. Y. (2016). Larval development to the ninth zoeal stage of Heterocarpus abulbus Yang, Chan and Chu, 2010 (Decapoda: Caridea: Pandalidae), a deep-water shrimp with high fishery potential. Journal of Crustacean Biology, 36, 310–328. https://doi.org/10.1163/1937240X-00002423
Jorde, P. E., Sovik, G., Wesrgaard, J. I., Albertsen, J., Andre, C., Hvingel, C., Johansen, T., Sandvik, A. D., Kingsley, M., & Jordstad, K. E. (2015). Genetically distinct populations of northern shrimp, Pandalus borealis, in the North Atlantic: Adaptation to different temperatures as an isolation factor. Molecular Ecology, 24, 1742–1757. https://doi.org/10.1111/mec.13158
Kaiser, J., Lamy, F., & Hebbeln, D. (2005). A 70-kyr sea surface temperature record off southern Chile (ocean drilling program site 1233). Paleoceanography, 20, 1–15. https://doi.org/10.1029/2005PA001146
Kaiser, J., Schefuß, E., Lamy, F., Mohtadi, M., & Hebbeln, D. (2008). Glacial to Holocene changes in sea surface temperature and coastal vegetation in north central Chile: high versus low latitude forcing. Quaternary Science Reviews, 27(21–22), 2064–2075.
Kass, R. E., & Raftery, A. E. (1995). Bayes factors. Journal of the American Statistical Association, 90, 773–795. https://doi.org/10.1080/01621459.1995.10476572
Kearse, M., Moir, R., Wilson, A., Stones-Havas, S., Cheung, M., Sturrock, S., Buxton, S., Cooper, A., Markowitz, S., Duran, C., Thierer, T., Ashton, B., Meintjes, P., & Drummond, A. (2012). Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics, 28, 1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Kehdkar, G. D., Reddy, A. C., Ron, T. B., & Haymer, D. (2013). High levels of genetic diversity in Penaeus monodon populations from the east coast of India. Springerplus, 2, 671. https://doi.org/10.1186/2193-1801-2-671
Kelly, R. P., & Palumbi, S. R. (2010). Genetic structure among 50 species of the Northeastern Pacific rocky intertidal community. PLoS One, 5, e8594. https://doi.org/10.1371/journal.pone.0008594
Kilada, R., & Acuña, E. (2015). Direct age determination by growth band counts of three commercially important crustacean species in Chile. Fisheries Research, 170, 134–143. https://doi.org/10.1016/j.fishres.2015.05.026
Knutsen, H., Jorde, P. E., Gonzalez, E. B., Eigaard, O. R., Pereyra, R. T., Sannaes, H., Dahl, M., André, C., & Sovik, G. (2015). Does population genetic structure support present management regulations of the northern shrimp (Pandalus borealis) in Skagerrak and the North Sea? ICES Journal of Marine Science, 72, 863–871. https://doi.org/10.1093/icesjms/fsu204
Kong, X. Y., Li, Y. L., Shi, W., & Kong, J. (2010). Genetic variation and evolutionary demography of Fenneropenaeus chinensis populations, as revealed by the analysis of mitochondrial control region sequences. Genetics and Molecular Biology, 33, 379–389. https://doi.org/10.1590/S1415-47572010005000019
Landeira, J. M., Lozano-Soldevilla, F., Almansa, E., & González-Gordillo, J. I. (2010). Early larval morphology of the armed nylon shrimp Heterocarpus ensifer ensifer A. Milne-Edwards, 1881 (Decapoda, Caridea, Pandalidae) from laboratory culture. Zootaxa, 2427, 1–14. https://doi.org/10.11646/zootaxa.2427.1.1
Legendre, P., & Gallagher, E. D. (2001). Ecologically meaningful transformations for ordination of species data. Oecologia, 129, 271–280. https://doi.org/10.1007/s004420100716
Lessios, H. A. (2008). The great American Schism: Divergence of marine organisms after the rise of the Central American Isthmus. Annual Review of Ecology, Evolution and Systematics, 39, 63–91. https://doi.org/10.1146/annurev.ecolsys.38.091206.095815
Liao, Y., Ma, K. Y., De Grave, S., Komai, T., & Chan, T.-Y. (2019). Systematic analysis of the caridean shrimp superfamily Pandaloidea (Crustacea: Decapoda) based on molecular and morphological evidence. Molecular Phylogenetics and Evolution, 134, 200–210. https://doi.org/10.1016/j.ympev.2019.02.006
Librado, P., & Rozas, J. (2009). DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics, 25, 1451–1452. https://doi.org/10.1093/bioinformatics/btp187
Lipcius, R. N., Eggleston, D. B., Schreiber, S. J., Seitz, R. D., Shen, J., Sisson, M., Stockhausen, W. T., & Wang, H. V. (2008). Importance of metapopulation connectivity to restocking and restoration of marine species. Reviews in Fisheries Science, 16, 101–110. https://doi.org/10.1080/10641260701812574
Marra, A., Mona, S., Sa, R. M., D’Onghia, G., & Maiorano, P. (2015). Population genetic history of Aristeus antennatus (Crustacea: Decapoda) in the Western and Central Mediterranean Sea. PLoS One, 10, e0117272. https://doi.org/10.1371/journal.pone.0117272
Martinez, I., Aschan, M., Skjerdal, T., & Aljanabi, S. M. (2006). The genetic structure of Pandalus borealis in the Northeast Atlantic determined by RAPD analysis. ICES Journal of Marine Science, 63, 840–850. https://doi.org/10.1016/j.icesjms.2006.03.006
McCulloch, R. D., Bentley, M. J., Purves, R. S., Hulton, N. R. J., Sugden, D. E., & Clapperton, C. M. (2000). Climatic inferences from glacial and palaeoecological evidence at the last glacial termination, southern South America. Journal of Quaternary Science, 15, 409–417. https://doi.org/10.1002/1099-1417(200005)15:4%3c409::AID-JQS539%3e3.0.CO;2-%23
McMillen-Jackson, A. L., & Bert, T. M. (2003). Disparate patterns of population genetic structure and population history in two sympatric penaeid shrimp species (Farfantepenaeus aztecus and Litopenaeus setiferus) in the eastern United States. Molecular Ecology, 12, 2895–2905. https://doi.org/10.1046/j.1365-294X.2003.01955.x
McMillen-Jackson, A. L., & Bert, T. M. (2004). Genetic diversity in the mtDNA control region and population structure in the pink shrimp Farfantepenaeus duorarum. Journal of Crustacean Biology, 24, 101–109. https://doi.org/10.1651/C-2372
Mujica, A., Acuña, E., & Nava, M. L. (2011). Distribución y abundancia de larvas de Heterocarpus reedi Bahamonde, 1955, Cervimunida johni Porter, 1903 y Pleuroncodes monodon (H. Milne Edwards, 1837), frente a Coquimbo y Caldera, Chile. Latin American Journal of Aquatic Research, 39, 138–150. https://doi.org/10.3856/vol39-issuel-fulltext-13
Oksanen, J., Kindt, R., Legendre, P., O’Hara, B., Stevens, M. H. H., Oksanen, M. J., & Suggests, M. A. S. S. (2017). VEGAN: Community Ecology Package. R package version 2.4–3. https://CRAN.R-project.org/package=vegan
Palsboll, P. J., Bérubé, M., & Allendorf, F. W. (2006). Identification of management units using population genetic data. Trends in Ecology and Evolution, 22, 11–16. https://doi.org/10.1016/j.tree.2006.09.003
Pedersen, S. A., Storm, L., & Simonsen, C. S. (2002). Northern shrimp Pandalus borealis recruitment in West Greenland waters Part I Distribution of Pandalus shrimp larvae in relation to hydrography and plankton. Journal of Northwest Atlantic Fishery Science, 30, 47–60. https://doi.org/10.2960/J.v30.a3
Petit, R. J., Mousadik, A. E., & Pons, O. (1998). Identifying populations for conservation on the basis of genetic markers. Conservation Biology, 12, 844–855. https://doi.org/10.1111/j.1523-1739.1998.96489.x
Pineda, J., Hare, J. A., & Sponaugle, S. (2007). Larval transport and dispersal in the coastal ocean and consequences for population connectivity. Oceanography, 20, 22–39. https://doi.org/10.5670/oceanog.2007.27
Pinsky, M. L., & Palumbi, S. R. (2014). Meta-analysis reveals lower genetic diversity in overfished populations. Molecular Ecology, 23, 29–39. https://doi.org/10.1111/mec.12509
Purushothaman, P., Chakraborty, R. D., Gidda, M., & Ganesan, K. (2020). Investigation of genetic diversity and stock structure of Aristeus alcocki Ramadan, 1938 (Decapoda: Aristeidae) populations in the Indian coast with microsatellite markers. Fisheries Research, 227, 205550. https://doi.org/10.1016/j.fishres.2020.105550
R Development Core Team. (2022). R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org
Rambaut, A., & Drummond, A. J. (2007). Tracer v1.5. http://beast.bio.ed.ac.uk
Robainas-Barcia, A., Blanco, G., Sanchez, J. A., Monnerot, M., Solignac, M., & Garcia-Machdo, E. (2008). Spatiotemporal genetic differentiation of Cuban natural populations of the pink shrimp Farfantepenaeus notialis. Genetica, 133, 283–294. https://doi.org/10.1007/s10709-007-9212-z
Rogers, A. R., & Harpending, H. (1992). Population growth makes waves in the distribution of pairwise genetic differences. Molecular Biology and Evolution, 9, 552–569. https://doi.org/10.1093/oxfordjournals.molbev.a040727
Romero, O. E., Kim, J. H., & Hebbeln, D. (2006). Paleoproductivity evolution off central Chile from the last glacial maximum to the early Holocene. Quaternary Research, 65, 519–525. https://doi.org/10.1016/j.yqres.2005.07.003
Sarda, F., Roldan, M. I., Heras, S., & Maltagliati, F. (2010). Influence of the genetic structure of the red and blue shrimp, Aristeus antennatus (Risso, 1816), on the sustainability of a deep-sea population along a depth gradient in the western Mediterranean. Scientia Marina, 74, 569–575. https://doi.org/10.3989/scimar.2010.74n3569
Segovia, N. I., González-Wevar, C. A., Naretto, J., Rosenfeld, S., Brickle, P., Hüne, M., Bernal, V., Haye, P. A., & Poulin, E. (2022). The right tool for the right question: Contrasting biogeographic patterns in the notothenioid fish. Proceedings of the Royal Society B, 20212738. https://doi.org/10.1098/rspb.2021.2738
Shumway, S. E., Perkins, H. C., Schick, D. F., & Stickney, A. P. (1985). Synopsis of biological data on the pink shrimp, Pandalus borealis Krøyer 1838. FAO Fisheries Synopsis, 144. pp. 157.
Tajima, F. (1989). Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics, 123, 585–595.
Taylor, M. L., & Roterman, C. N. (2017). Invertebrate population genetics across Earth’s largest habitat: the deep-sea floor. Molecular Ecology, 26, 4872–4896. https://doi.org/10.1111/mec.14237
Teodoro, S. S. A., da Silva Cortinhas, M. C., Proietti, M. C., Costa, R. C., & Dumont, L. F. C. (2020). High genetic connectivity among pink shrimp Farfantepenaeus paulensis (Pérez-Farfante, 1967) groups along the south-southeastern coast of Brazil. Estuarine and Coastal Shelf Science, 232, 106488. https://doi.org/10.1016/j.ecss.2019.106488
Thatje, S., & Bacardit, R. (2000). Larval development of Austropandalus grayi (Cunningham, 1871) (Decapoda: Caridea: Pandalidae) from the southwestern Atlantic Ocean. Crustaceana, 73, 609–628. https://doi.org/10.1163/156854000504697
Thorpe, J. P., Solé-Cava, A. M., & Watts, P. C. (2000). Exploited marine invertebrates: Genetics and fisheries. Hydrobiologia, 420, 165–184. https://doi.org/10.1023/A:1003987117508
Vu, N. T. T., Zenger, K. R., Guppy, J. L., Sellars, M. J., Silva, C. N. S., Kjeldsen, S. R., & Jerry, D. R. (2020). Fine-scale population structure and evidence of local adaptation in Australian giant tiger shrimp (Penaeus monodon) using SNP analysis. BMC Genomics, 21, 669. https://doi.org/10.1186/s12864-020-07084-x
Wang, L., Yang, J., Sun, M., Yang, C., Cui, Z., Jang, I. K., & Song, L. (2014). The genetic diversity and differentiation of shrimp Fenneropenaeus chinensis in the Yellow Sea revealed by polymorphism in control region of mitochondrial DNA. Invertebrate Survival Journal, 11, 309–318. http://www.isj.unimo.it/index.php/ISJ/article/view/331
Waqairatu, S. S., Dierens, L., Cowley, J. A., Dixon, T. J., Johnson, K. N., Barnes, A. C., & Li, Y. T. (2012). Genetic analysis of Black Tiger shrimp (Penaeus monodon) across its natural distribution range reveals more recent colonization of Fiji and other South Pacific islands. Ecology and Evolution, 8, 2057–2071. https://doi.org/10.1002/ece3.316
Weersing, K., & Toonen, R. J. (2009). Population genetics, larval dispersal, and connectivity in marine systems. Marine Ecology Progress Series, 393, 1–12. https://doi.org/10.3354/meps08287
Wehrtmann, I. S., Arana, P., Barriga, E., Gracia, A., & Pezzuto, P. R. (2012). Deep-water shrimp fisheries in Latin America: a review. Latin American Journal of Aquatic Research, 40, 497–535.
Weir, B. S., & Cockerham, C. C. (1984). Estimating F-Statistics for the analysis of population structure. Evolution, 38, 1358–1370. https://doi.org/10.1111/j.1558-5646.1984.tb05657.x
Willi, Y., Van Buskirk, J., & Hoffmann, A. A. (2006). Limits to the adaptive potential of small populations. Annual Review of Ecology, Evolution, and Systematics, 37, 433–438. https://www.jstor.org/stable/30033839
Yannicelli, B., & Castro, L. (2013). Ecophysiological constrains on the larvae of Pleuroncodes monodon and the implications for its reproductive strategy in poorly oxygenated waters of the Chile-Peru undercurrent. Journal of Plankton Research, 35, 566–581. https://doi.org/10.1093/plankt/fbs090
Yáñez, E. (1974). Distribución y abundancia relativa estacional de los recursos disponibles a un arte de arrastre camaronero frente a la costa de Valparaíso (invierno y primavera 1972). Investigaciones Marinas, 5, 125–138.
Yáñez, E., & Barbieri, M. A. (1974). Distribución y abundancia relativa de los recursos disponibles a un arte de arrastre camaronero frente a la costa de Valparaíso (invierno 1973). Investigaciones Marinas, 5, 137–156.
Acknowledgements
We thank Alex Cortés and Christian Véliz for sample collection and Cecilia Gatica for sample processing. This study was funded by Universidad Católica del Norte, IFOP Grant P-682058, and the Millennium Science Initiative Program - ICN2019_015, Chile.
Funding
This study was funded by Universidad Católica del Norte, IFOP Grant P-682058 and the Millennium Science Initiative Program-ICN2019_015, Chile.
Author information
Authors and Affiliations
Contributions
PAH, EA, and NIS conceived the idea, designed the study, contributed with acquisition of data, conceived, designed, performed the analyses, and wrote the manuscript. RV contributed with laboratory work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Haye, P.A., Segovia, N.I., Vera, R. et al. Phylogeography reveals a panmictic population of the Chilean nylon shrimp along its exploitation range in the southeast Pacific Ocean. Org Divers Evol 23, 125–138 (2023). https://doi.org/10.1007/s13127-022-00589-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13127-022-00589-1