Abstract
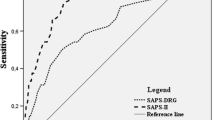
This study aimed to establish a new scoring model based on the early brain injury (EBI) indicators to predict the 90-day functional outcomes in patients with aneurysmal subarachnoid hemorrhage (aSAH). We retrospectively enrolled 825 patients and prospectively enrolled 108 patients with aSAH who underwent surgical clipping or endovascular coiling (derivation cohort = 640; validation cohort = 185; prospective cohort = 108) in our institute. We established a logistic regression model based on independent risk factors associated with 90-day unfavorable outcomes. The discrimination of the prognostic model was assessed by the area under the curve in a receiver operating characteristic curve analysis. The Hosmer–Lemeshow goodness-of-fit test and a calibration plot were used to evaluate the calibration of the prediction model. The developed scoring model named “TAPS” (total score, 0–7 points) included the following admission variables: age > 55 years old, WFNS grade of 4–5, mFS grade of 3–4, Graeb score of 5–12, white blood cell count > 11.28 × 109/L, and surgical clipping. The model showed good discrimination with the area under the curve in the derivation, validation, and prospective cohorts which were 0.816 (p < 0.001, 95%CI = 0.77–0.86), 0.810 (p < 0.001, 95%CI = 0.73–0.90), and 0.803 (p < 0.001, 95%CI = 0.70–0.91), respectively. The model also demonstrated good calibration (Hosmer–Lemeshow goodness-of-fit test: X2 = 1.75, df = 8, p = 0.988). Compared with other predictive models, TAPS is an easy handle tool for predicting the 90-day unfavorable outcomes of aSAH patients, which can help clinicians better understand the concept of EBI and quickly identify those patients at risk of poor prognosis, providing more positive treatment strategies. Trial registration: NCT04785976. Registered 5 March 2021-retrospectively registered, http://www.clinicaltrials.gov.


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Kusaka G, Ishikawa M, Nanda A, Granger DN, Zhang JH. Signaling pathways for early brain injury after subarachnoid hemorrhage. J Cereb Blood Flow Metab. 2004;24:916–25.
Tso MK, Macdonald RL. Subarachnoid hemorrhage: a review of experimental studies on the microcirculation and the neurovascular unit. Transl Stroke Res. 2014;5:174–89.
Claassen J, Carhuapoma JR, Kreiter KT, Du EY, Connolly ES, Mayer SA. Global cerebral edema after subarachnoid hemorrhage: frequency, predictors, and impact on outcome. Stroke. 2002;33:1225–32.
Fujii M, Yan J, Rolland WB, Soejima Y, Caner B, Zhang JH. Early brain injury, an evolving frontier in subarachnoid hemorrhage research. Transl Stroke Res. 2013;4:432–46.
Ray B, Pandav VM, Mathews EA, et al. Coated-platelet trends predict short-term clinical outcomeafter subarachnoid hemorrhage. Transl Stroke Res. 2018;9:459–70.
Rass V, Helbok R. Early brain injury after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep. 2019;19:78.
Sehba FA, Friedrich V. Early events after aneurysmal subarachnoid hemorrhage. Acta Neurochir Suppl. 2015;120:23–8.
Taki W, Sakai N, Suzuki H, et al. Determinants of poor outcome after aneurysmal subarachnoid hemorrhage when both clipping and coiling are available: Prospective Registry of Subarachnoid Aneurysms Treatment (PRESAT) in Japan. World Neurosurgery. 2011;76:437–45.
Fu C, Yu W, Sun L, Li D, Zhao C. Early cerebral infarction following aneurysmal subarachnoid hemorrhage: frequency, risk factors, patterns, and prognosis. Curr Neurovasc Res. 2013;10:316–24.
Radolf S, Smoll N, Drenckhahn C, Dreier JP, Vajkoczy P, Sarrafzadeh AS. Cerebral lactate correlates with early onset pneumonia after aneurysmal SAH. Transl Stroke Res. 2014;5:278–85.
Suzuki H, Sakurai M, Fujimoto M, Tsuchiya T, Sakaida H, Taki W. Complete recovery from aneurysmal subarachnoid hemorrhage associated with out-of-hospital cardiopulmonary arrest. Eur J Emerg Med. 2010;17:42–4.
Rass V, Helbok R. Early brain injury after poor-grade subarachnoid hemorrhage. Current Neurology and Neuroscience Reports. 2019 19.
Connolly ES Jr, Rabinstein AA, Carhuapoma JR, et al. Guidelines for the management of aneurysmal subarachnoid hemorrhage: a guideline for healthcare professionals from the American Heart Association/american Stroke Association. Stroke. 2012;43:1711–37.
Report of World Federation of Neurological Surgeons Committee on a universal subarachnoid hemorrhage grading scale. J Neurosurg. 1988 68: 985–986.
Claassen J, Bernardini GL, Kreiter K, et al. Effect of cisternal and ventricular blood on risk of delayed cerebral ischemia after subarachnoid hemorrhage: the Fisher scale revisited. Stroke. 2001;32:2012–20.
Graeb DA, Robertson WD, Lapointe JS, Nugent RA, Harrison PB. Computed tomographic diagnosis of intraventricular hemorrhage. Etiology and prognosis Radiology. 1982;143:91–6.
Ahn SH, Savarraj JP, Pervez M, et al. The subarachnoid hemorrhage early brain edema score predicts delayed cerebral ischemia and clinical outcomes. Neurosurgery. 2018;83:137–45.
Fujiki Y, Matano F, Mizunari T, et al. Serum glucose/potassium ratio as a clinical risk factor for aneurysmal subarachnoid hemorrhage. J Neurosurg. 2018;129:870–5.
Giede-Jeppe A, Reichl J, Sprügel M, et al. Neutrophil-to-lymphocyte ratio as an independent predictor for unfavorable functional outcome in aneurysmal subarachnoid hemorrhage. J Neurosurg. 2019;132:400–7.
Wang Z, Pei W, Chen L, Ning Y, Luo Y. Mean platelet volume/platelet count ratio is associated with poor clinical outcome after aneurysmal subarachnoid hemorrhage. J Stroke Cerebrovas Dis: Official J Nat Stroke Assoc. 2020;29:105208.
Yun S, Jun Yi H, Hoon Lee D, Hoon SJ. Clinical significance of platelet to neutrophil ratio and platelet to lymphocyte ratio in patients with aneurysmal subarachnoid hemorrhage. J Clin Neurosci Official J Neurosurg Soc Australasia. 2021;92:49–54.
Zhang P, Li Y, Zhang H, et al. Prognostic value of the systemic inflammation response index in patients with aneurismal subarachnoid hemorrhage and a Nomogram model construction. British journal of neurosurgery. 2020: 1–7. online ahead of print.
Sullivan LM, Massaro JM, D’Agostino RB Sr. Presentation of multivariate data for clinical use: the Framingham Study risk score functions. Stat Med. 2004;23:1631–60.
Time trends in outcome of subarachnoid hemorrhage. population-based study and systematic review. Neurology. 2010;74:1494–501.
The falling rates of hospital admission. case fatality, and population-based mortality for subarachnoid hemorrhage in England, 1999–2010. J Neurosurg. 2016;125:1–7.
Zhang JH. Vascular neural network in subarachnoid hemorrhage. Transl Stroke Res. 2014;5:423–8.
Sehba FA, Pluta RM, Zhang JH. Metamorphosis of subarachnoid hemorrhage research: from delayed vasospasm to early brain injury. Mol Neurobiol. 2011;43:27–40.
Yue Q, Liu Y, Leng B, et al. A prognostic model for early post-treatment outcome of elderly patients with aneurysmal subarachnoid hemorrhage. World Neurosurg. 2016;95:253–61.
Lai X, Zhang W, Ye M, Liu X, Luo X. Development and validation of a predictive model for the prognosis in aneurysmal subarachnoid hemorrhage. J Clin Lab Anal. 2020;34:e23542.
Jaja B, Saposnik G, Lingsma H, et al. Development and validation of outcome prediction models for aneurysmal subarachnoid haemorrhage: the SAHIT multinational cohort study. BMJ (Clinical research ed). 2018;360:j5745.
Lee V, Ouyang B, John S, et al. Risk stratification for the in-hospital mortality in subarachnoid hemorrhage: the HAIR score. Neurocrit Care. 2014;21:14–9.
Witsch J, Frey H, Patel S, et al. Prognostication of long-term outcomes after subarachnoid hemorrhage: the FRESH score. Ann Neurol. 2016;80:46–58.
Medzhitov R. Origin and physiological roles of inflammation. Nature. 2008;454:428–35.
Saand A, Yu F, Chen J, Chou S. Systemic inflammation in hemorrhagic strokes - a novel neurological sign and therapeutic target? Journal of cerebral blood flow and metabolism : official journal of the International Society of Cerebral Blood Flow and Metabolism. 2019;39:959–88.
Al-Mufti F, Misiolek K, Roh D, et al. White blood cell count improves prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurgery. 2019;84:397–403.
Ma X, Lan F, Zhang Y. Associations between C-reactive protein and white blood cell count, occurrence of delayed cerebral ischemia and poor outcome following aneurysmal subarachnoid hemorrhage: a systematic review and meta-analysis. Acta Neurol Belg. 2021;121:1311–24.
Mahta A, Azher A, Moody S, et al. Association of early white blood cell trend with outcomes in aneurysmal subarachnoid hemorrhage. World Neurosurgery. 2021;151:e803–9.
Hu P, Yang X, Li Y, et al. Predictive effects of admission white blood cell counts and hounsfield unit values on delayed cerebral ischemia after aneurysmal subarachnoid hemorrhage. Clin Neurol Neurosurg. 2022;212:107087.
Kasius K, Frijns C, Algra A, Rinkel G. Association of platelet and leukocyte counts with delayed cerebral ischemia in aneurysmal subarachnoid hemorrhage. Cerebrovascular Dis (Basel, Switzerland). 2010;29:576–83.
Li R, Lin F, Chen Y, et al. In-hospital complication-related risk factors for discharge and 90-day outcomes in patients with aneurysmal subarachnoid hemorrhage after surgical clipping and endovascular coiling: a propensity score-matched analysis. Journal of Neurosurgery. 2021: 1–12. online ahead of print.
Shen J, Yu J, Huang S, et al. Scoring model to predict functional outcome in poor-grade aneurysmal subarachnoid hemorrhage. Front Neurol. 2021;12:601996.
Takemoto Y, Hasegawa Y, Hashiguchi A, Moroki K, Tokuda H, Mukasa A. Predictors for functional outcome in patients with aneurysmal subarachnoid hemorrhage who completed in-hospital rehabilitation in a single institution. J Stroke Cerebrovasc Dis. 2019;28:1943–50.
McDougall CG, Spetzler RF, Zabramski JM, et al. The Barrow Ruptured Aneurysm Trial. J Neurosurg. 2012;116:135–44.
Molyneux AJ, Kerr RS, Yu LM, et al. International subarachnoid aneurysm trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised comparison of effects on survival, dependency, seizures, rebleeding, subgroups, and aneurysm occlusion. Lancet. 2005;366:809–17.
Stienen MN, Germans M, Burkhardt JK, et al. Predictors of in-hospital death after aneurysmal subarachnoid hemorrhage: analysis of a nationwide database (Swiss SOS [Swiss Study on Aneurysmal Subarachnoid Hemorrhage]). Stroke. 2018;49:333–40.
Jabbarli R, Reinhard M, Roelz R, et al. The predictors and clinical impact of intraventricular hemorrhage in patients with aneurysmal subarachnoid hemorrhage. International journal of stroke : official journal of the International Stroke Society. 2016;11:68–76.
Eagles ME, Jaja BNR, Macdonald RL. Incorporating a modified Graeb score to the modified fisher scale for improved risk prediction of delayed cerebral ischemia following aneurysmal subarachnoid hemorrhage. Neurosurgery. 2018;82:299–305.
Xu P, Tao C, Zhu Y, et al. TAK1 mediates neuronal pyroptosis in early brain injury after subarachnoid hemorrhage. J Neuroinflammation. 2021;18:188.
Deng HJ, Deji Q, Zhaba W, et al. A20 establishes negative feedback with TRAF6/NF-kappaB and attenuates early brain injury after experimental subarachnoid hemorrhage. Front Immunol. 2021;12:623256.
Kuang H, Wang T, Liu L, et al. Treatment of early brain injury after subarachnoid hemorrhage in the rat model by inhibiting p53-induced ferroptosis. Neurosci Lett. 2021;762:136134.
Dodd WS, Noda I, Martinez M, Hosaka K, Hoh BL. NLRP3 inhibition attenuates early brain injury and delayed cerebral vasospasm after subarachnoid hemorrhage. J Neuroinflammation. 2021;18:163.
Yuan JY, Chen Y, Kumar A, et al. Automated quantification of reduced sulcal volume identifies early brain injury after aneurysmal subarachnoid hemorrhage. Stroke. 2021;52:1380–9.
McIntyre MK, Halabi M, Li B, et al. Glycemic indices predict outcomes after aneurysmal subarachnoid hemorrhage: a retrospective single center comparative analysis. Sci Rep. 2021;11:158.
Wang Y, Tian Y, Wang D, et al. High angiopoietin-1 levels predict a good functional outcome within 72 h of an aneurysmal subarachnoid hemorrhage: a prospective study from a single center. J Neurol Sci. 2015;356:72–6.
Feghali J, Kim J, Gami A, et al. Monocyte-based inflammatory indices predict outcomes following aneurysmal subarachnoid hemorrhage. Neurosurg Rev. 2021;44:3499–507.
Zheng S, Wang H, Chen G, et al. Higher serum levels of lactate dehydrogenase before microsurgery predict poor outcome of aneurysmal subarachnoid hemorrhage. Front Neurol. 2021;12:720574.
Acknowledgements
We acknowledge the contribution of all staff who participated in the present study.
Funding
This study was supported by the National Key Research and Development Program of China (Grant Nos. 2021YFC2501101 and 2020YFC2004701) and the National Natural Science Foundation of China (Grant Nos. 82071296, 81671129, and 81471210).
Author information
Authors and Affiliations
Contributions
Runting Li: conceptualization (equal); data curation (lead); formal analysis (lead); investigation (lead); methodology (equal); project administration (equal); software (lead); validation (lead); visualization (lead); writing original draft (lead); and writing review and editing (equal). Fa Lin: data curation (equal); methodology (equal); and software (equal). Yu Chen: data curation (equal); methodology (equal); and software (equal). Junlin Lu: data curation (equal); methodology (equal); and software (equal). Heze Han: data curation (equal); methodology (equal); and software (equal). Li Ma: data curation (equal). Yahui Zhao: data curation (equal). Debin Yan: data curation (equal). Ruinan Li: data curation (equal). Jun Yang: data curation (equal). Shihao He: data curation (equal). Zhipeng Li: data curation (equal). Haibin Zhang: data curation (equal). Kexin Yuan: data curation (equal). Ke Wang: data curation (equal). Qiang Hao: data curation (equal) and supervision (equal). Xun Ye: data curation (equal) and supervision (equal). Hao Wang: data curation (equal) and supervision (equal). Hongliang Li: data curation (equal) and supervision (equal). Linlin Zhang: data curation (equal) and supervision (equal). Guangzhi Shi: data curation (equal) and supervision (equal). Jianxin Zhou: data curation (equal) and supervision (equal). Yang Zhao: data curation (equal); resources (equal); and supervision (equal). Yukun Zhang: data curation (equal); resources (equal); and supervision (equal). Youxiang Li: data curation (equal); resources (equal); and supervision (equal). Shuo Wang: conceptualization (equal); funding acquisition (equal); resources (equal); supervision (equal); and writing review and editing (equal). Xiaolin Chen: conceptualization (equal); funding acquisition (equal); resources (equal); supervision (equal); and writing review and editing (equal). Yuanli Zhao: conceptualization (equal); resources (equal); supervision (equal); and writing review and editing (equal).
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
This study was approved by the Institutional Review Board of the Beijing Tiantan Hospital (KY 2021–008-01). Informed consent was obtained from all individual participants or their authorized representatives included in the study. All the analyses were performed in accordance with the Declaration of Helsinki and the local ethics policies.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, R., Lin, F., Chen, Y. et al. A 90-Day Prognostic Model Based on the Early Brain Injury Indicators after Aneurysmal Subarachnoid Hemorrhage: the TAPS Score. Transl. Stroke Res. 14, 200–210 (2023). https://doi.org/10.1007/s12975-022-01033-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-022-01033-4