Abstract
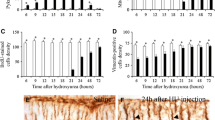
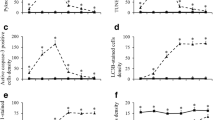
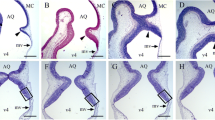
The current paper presents a histological analysis of the cell death in the cerebellar external granular layer (EGL) following the treatment with a single dose (2 mg/g) of hydroxyurea (HU). The rats were examined at postnatal days (P) 5, 10, and 15, and sacrificed at appropriate times ranging from 6 to 48 h after treatment administration. Studies were done in each cortical lobe (anterior, central, posterior, and inferior). The quantification of several parameters, such as density of 5-bromo-2′-deoxyuridine, TUNEL, vimentin, and tomato lectin-stained cells, revealed that HU compromises the viability of EGL cells. Our results indicate that P10 is a time of high vulnerability to injury. We also show here that the anterior and central lobes are the cortical regions most susceptible to the action of the HU. Additionally, our data also indicate that from 6 to 24 h after HU-exposure is a time-window of high sensibility to this agent. On the other hand, our ultrastructural analysis confirmed that HU administration produces the activation of apoptotic cellular events in the EGL, resulting in a substantial number of dying cells. Different stages of apoptosis can be observed in all cortical lobes at all investigated postnatal ages and survival times. Moreover, we observed that dying neuroblasts were covered by laminar processes of Bergmann glia, and that these unipolar astrocytes presented cytological features of phagocytes engulfing apoptotic bodies and cell debris. The electron microscopy study also revealed the participation of ameboid microglial cells in the phagocytosis of apoptotic cells in the regions of the EGL with extensive cell death.











Similar content being viewed by others
References
Allin MP (2016) Novel insights from quantitative imaging of the developing cerebellum. Semin Fetal Neonatal Med 21(5):333–338
Altman J, Bayer SA (1997) Development of the cerebellar system: in relation to its evolution, structure and functions. CRC Press, Inc., Boca Raton
Ashwell K (1990) Microglial and cell death in the developing mouse cerebellum. Dev Brain Res 55:219–230
Avella D, Pisu MB, Roda E, Gravati M, Bernocchi G (2006) Reorganization of the rat cerebellar cortex during postnatal development following cisplatin treatment. Exp Neurol 201:131–143
Bernocchi G, Bottone MG, Piccolini VM, Dal Bo V, Santin G, De Pascali SA, Migoni D, Fanizzi FP (2011) Developing central nervous system and vulnerability to platinum compounds. Chemther Res Pract 315418
Bottone MG, Veronica DB, Piccolini VM, Bottiroli G, De Pascali SA, Fanizzi FP, Bernocchi G (2012) Developmental expression of cellular prion protein and apoptotic molecules in the rat cerebellum: effects of platinum compounds. J Chem Neuroanat 46:19–29
Bovolenta P, Liem RK, Mason CA (1984) Development of cerebellar astroglia: transitions in form and cytoskeletal content. Dev Biol 102:248–259
Buffo A, Rossi F (2013) Origin, lineage and function of cerebellar glia. Prog Neurobiol 109:42–63
Campuzano O, Castillo-Ruiz MM, Acarín L, González B, Castellano B (2011) Decreased myeloperoxidase expressing cell in the aged rat brain after excitotoxic damage. Exp Gerontol 46:723–730
Chedotal A (2010) Should I stay or should I go? Becoming a granule cell. Trends Neurosci 33:163–172
Dahmane N, Ruiz i Altaba A (1999) Sonic hedgehog regulates the growth and patterning of the cerebellum. Development 126:3089–3100
Davies BW, Kohanski MA, Simmons LA, Winkler JA, Collins JJ, Walker GC (2009) Hydroxyurea induces hydroxyl radical-mediated cell death in Escherichia coli. Mol Cell 36:845–860
de Montalembert M, Belloy M, Bernaudin F, Gouraud F, Capdeville R, Mardini R, Philippe N, Jais JP, Bardakdjian J, Ducrocq R, Maier-Redelsperger M, Elion J, Labie D, Girot R (1997) Three-year follow-up of hydroxyurea treatment in severely ill children with sickle cell disease. The French Study Group on Sickle Cell Disease. J Pediatr Hematol Oncol 19:313–318
Doi K (2011) Mechanisms of neurotoxicity induced in the developing brain of mice and rats by DNA-damaging chemicals. J Toxicol Sci 36(6):695–712
Ebels EJ, Peters I, Thijs A (1975) Studies on ectopic granule cells in the cerebellar cortex. III. An investigation into the restoration of the external granular layer after partial destruction. Acta Neuropathol 31:103–107
Eroglu M, Derry WB (2016) Your neighbours matter - non-autonomous control of apoptosis in development and disease. Cell Death Differ 23:1110–1118
Goldowitz D, Hamre K (1998) The cells and molecules that make a cerebellum. Trends Neurosci 21:375–382
Hachem S, Laurenson AS, Hugnot JP, Legravered C (2007) Expression of S100B during embryonic development of the mouse cerebellum. BMC Dev Biol 7:17. https://doi.org/10.1186/1471-213X-7-17
Hamre KM, Goldowitz D (1996) Analysis of gene action in the meander tail mutant mouse: examination of cerebellar phenotype and mitotic activity of granule cell neuroblasts. J Comp Neurol 368:304–315
Hankins JS, Ware RE, Rogers ZR, Wynn LW, Lane PA, Scott JP, Wang WC (2005) Long-term hydroxyurea therapy for infants with sickle cell anemia: the HUSOFT extension study. Blood 106:2269–2275
Komuro H, Yacubova E, Yacubova E, Rakic P (2001) Mode and tempo of tangencial cell migration in the cerebellar external granular layer. J Neurosci 21:527–540
Koppel H, Lewis PD, Padel AJ (1983) Cell death in the external granular layer of normal and undernourished rats: futher observations, including estimates of rate of cell loss. Cell Tissue Kinet 16:99–106
Lafarga M, Andres MA, Calle E, Berciano MT (1998) Reactive gliosis of immature Bergmann glia and microglial activation in response to cell death of granule cell precursors induced by methylazoxymethanol treatment in developing rat cerebellum. Anat Embryol 198:111–122
Landis DM, Sidman RL (1978) Electron microscopic analysis of postnatal histogenesis in the cerebellar cortex of staggerer mutant mice. J Comp Neurol 179: 831–863
Lupo M, Olivito G, Siciliano L, Masciullo M, Bozzali M, Molinari M, Leggio M (2018) Development of a psychiatric disorder linked to cerebellar lesions. Cerebellum 17:438–446
Martí J, Santa-Cruz MC, Serra R, Hervás JP (2015) Systematic differences in time of cerebellar-neuron origin derived from bromodeoxyuridine immunoperoxidase staining protocols and tritiated thymidine autoradiographic: a comparative study. Int J Dev Neurosci 47:216–228
Martí J, Santa-Cruz MC, Serra R, Hervás JP (2016) Hydroxyurea treatment and development of the rat cerebellum: effects on the neurogenetic profiles and settled patterns of Purkinje cells and deep cerebellar nuclei neurons. Neurotox Res 30:563–580
Martí J, Molina V, Santa-Cruz MC, Hervás JP (2017) Developmental injury to the cerebellar cortex following hydroxyurea treatment in early postnatal life: an immunohistochemical and electron microscopic study. Neurotox Res 31:187–203
Moberget T, Ivry RB (2016) Cerebellar contributions to motor control and language comprehension: searching for common computational principles. Ann N Y Acad Sci 1369:154–171
Mousa AM, Al-Fadhli AS, Rao MS, Kilarkaje N (2015) Gestational lead exposure induces developmental abnormalities and up-regulates apoptosis of fetal cerebellar cells in rats. Drug Chem Toxicol 38:73–83
Navarra P, Preziosi P (1999) Hydroxyurea: new insights on an old drug. Crit Rev Oncol Hematol 29:249–255
Newton HB (2007) Hydroxyurea chemotherapy in the treatment of meningiomas. Neurosurg Focus 23(4):E11
Patxinos G, Watson C (1998) The rat brain in stereotaxic coordinates, 4th edn. Academic Press, San Diego
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Development Core Team (2017) Nlme: linear and nonlinear mixed effects models. R package version 3.1–131
Pisu MB, Roda E, Guioli S, Avella D, Bottone MG, Bernocchi G (2005) Proliferation and migration of granule cells in the developing rat cerebellum: cisplatin effects. Anat Rec 287:1226–1235
Rodríguez-Vázquez L, Martí J (2017) Effects of hydroxyurea exposure on the rat cerebellar neuroepithelium: an immunohistochemical and electron microscopic study along the anteroposterior and mediolateral axes. Neurotox Res 32:671–682
Saban N, Bujak M (2009) Hydroxyurea and hydroxamic acid derivatives as antitumor drugs. Cancer Chemother Pharmacol 64:213–221
Sampson M, Archibong AE, Powell A, Strange B, Roberson S, Hills ER, Bourne P (2010) Perturbation of the developmental potential of preimplantation mouse embryos by hydroxyurea. Int J Environ Res Public Health 7:2033–2044
Scheuer T, Sharkovska Y, Tarabykin V, Marggraf K, Brockmöller V, Bührer C, Endesfelder S, Schmitz T (2018) Neonatal hyperoxia perturbs neuronal development in the cerebellum. Mol Neurobiol 55:3901–3915
Schilling K (2018) Moving into shape: cell migration during the development and histogenesis of the cerebellum. Histochem Cell Biol 150:13–36
Schilling K, Oberdick J, Rossi F, Baader SL (2008) Besides Purkinje cells and granule neurons: an appraisal of the cell biology of the interneurons of the cerebellar cortex. Histochem Cell Biol 130:601–615
Schlisser AE, Hales BF (2013) Deprenyl enhances the teratogenicity of hydroxyurea in organogenesis stage mouse embryos. Toxicol Sci 134:391–399
Schultze B, Korr H (1981) Cell kinetic studies of different cell types in the developing and adult brain of the rat and the mouse: a review. Cell Tissue Kinet 14:309–325
Taranukhin AG, Taranukhina EY, Saransaari P, Pelto-Huikko M, Podkletnova IM, Oja SS (2012) Taurine protects cerebellar neurons of the external granular layer against ethanol-induced apoptosis in 7-day-old mice. Amino Acids 43:1705–1711
Ware RE, Despotovic JM, Mortier NA, Flanagan JM, He J, Smeltzer MP, Kimble AC, Aygun B, Wu S, Howard T, Sparreboom A (2011) Pharmacokinetics, pharmacodynamics, and pharmacogenetics of hydroxyurea treatment for children with sickle cell anemia. Blood 118:4985–4991
Woo GH, Katayama K, Jung JY, Uetsuka K, Bak EJ, Nakayama H, Doi K (2003) Hydroxyurea (HU)-induced apoptosis in the mouse fetal tissues. Histol Histopathol 18:387–392
Woo GH, Katayama K, Bak EJ, Ueno H, Tamauchi H, Uetsuka K, Nakayama H, Doi K (2004) Effects of prenatal hydroxyurea-treatment on mouse offspring. Exp Toxicol Pathol 56(1–2):1–7
Woo GH, Bak E-J, Nakayama H, Doi K (2005) Hydroxyurea (HU)-induced apoptosis in the mouse fetal lung. Exp Mol Pathol 79:59–67
Woo GH, Bak EJ, Katayama K, Doi K (2006) Molecular mechanisms of hydroxyurea (HU)-induced apoptosis in the mouse fetal brain. Neurotocol Teratol 28:125–134
World Health Organization. Model List of Essential Medicines. 2015
Wyss-Coray T, Mucke L (2002) Inflammation in neurodegenerative disease-a double-edged sword. Neuron 35:419–432
Xu H, Yang Y, Tang X, Zhao M, Liang F, Xu P, Hou B, Xing Y, Bao X, Fan X (2013) Bergman glia function in granule cell migration during cerebellum development. Mol Neurobiol 47:833–844
Yamada K, Watanabe M (2002) Cytodifferentiation of Bergmann glia and its relationship with Purkinje cells. Anat Sci Int 77:94–108
Zhou K, Bostrom M, Ek CJ, Li T, Xie C, Xu Y, Sun Y, Blomgren K, Zhu C (2017) Radiation induces progenitor cell death, microglia activation, and blood-brain barrier damage in the juvenile rat cerebellum. Sci Rep 7:46181
Acknowledgments
The authors are very grateful to Drs. María del Carmen Santa-Cruz and José Pablo Hervás for providing the animals.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Rodríguez-Vázquez, L., Vons, O., Valero, O. et al. Hydroxyurea Exposure and Development of the Cerebellar External Granular Layer: Effects on Granule Cell Precursors, Bergmann Glial and Microglial Cells. Neurotox Res 35, 387–400 (2019). https://doi.org/10.1007/s12640-018-9964-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-018-9964-5