Abstract
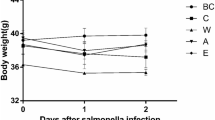
Lactic acid bacteria (LAB) are recognized as food-grade safe microorganisms and have many beneficial effects. LAB could maintain the host intestinal homeostasis and regulate intestinal microbial community to exert antibacterial effects. In this study, Lactiplantibacillus plantarum (L. plantarum, Lp01) strain isolated from pig intestine was orally administered to C57BL/6 mice, and mice were then infected with Salmonella typhimurium (ATCC14028). The protective effects of L. plantarum were evaluated by monitoring body weight loss, survival rates, bacterial loads in tissue, colon histopathology analysis, and cytokine secretion. 16S rRNA gene sequencing was also utilized to detect the dynamics of the blind gut microbial community in mice. We found that L. plantarum could significantly reduce the body weight loss and improve the survival rates. The survival rate in the L. P-Sty group was up to 67.5%, which was much higher than that in the STY group (25%). Counting of bacterial loads displayed that the colony-forming unit (CFU) of S. typhimurium in the spleen (p < 0.05) and the liver (p < 0.05) from L. P-Sty group both decreased, compared with STY group. Intestinal histopathology showed that it alleviated the intestinal injury caused by Salmonella, inhibited the secretion of pro-inflammatory cytokines, and promoted anti-inflammatory cytokines (p < 0. 01). In addition, L. plantarum also significantly ameliorated the intestinal gut microbiome disturbance caused by Salmonella. It displayed an obvious increase of beneficial bacteria including Lactobacillus and Bacteroidetes and reduction of pathogenic bacteria like Proteobacteria. In conclusion, L. plantarum could regulate microbial community to inhibit Salmonella typhimurium infection.











Similar content being viewed by others
References
Singh RK, Chang HW, Yan D et al (2017) Influence of diet on the gut microbiome and implications for human health. J Transl Med 15(1):73. https://doi.org/10.1186/s12967-017-1175-y
Nicholson JK, Holmes E, Kinross J et al (2012) Host-gut microbiota metabolic interactions. Science (New York, N.Y.) 336(6086):1262–1267. https://doi.org/10.1126/science.1223813
Kelly D, Conway S, Aminov R (2005) Commensal gut bacteria: mechanisms of immune modulation. Trends Immunol 26(6):326–333. https://doi.org/10.1016/j.it.2005.04.008
Nguyen T, Jin YY, Chung HJ et al (2017) Pharmabiotics as an emerging medication for metabolic syndrome and its related diseases. Molecules (Basel, Switzerland) 22(10):1795. https://doi.org/10.3390/molecules22101795
Quan J, Cai G, Yang M et al (2019) Exploring the fecal microbial composition and metagenomic functional capacities associated with feed efficiency in commercial DLY pigs. Front Microbiol 10:52. https://doi.org/10.3389/fmicb.2019.00052
Shah T, Baloch Z, Shah Z et al (2021) The intestinal microbiota: impacts of antibiotics therapy, colonization resistance, and diseases. Int J Mol Sci 22(12):6597. https://doi.org/10.3390/ijms22126597
Yang GY, Zhu YH, Zhang W et al (2016) Influence of orally fed a select mixture of Bacillus probiotics on intestinal T-cell migration in weaned MUC4 resistant pigs following Escherichia coli challenge. Vet Res 47(1):71. https://doi.org/10.1186/s13567-016-0355-8
Trevisi P, Latorre R, Priori D et al (2016) Effect of feed supplementation with live yeast on the intestinal transcriptome profile of weaning pigs orally challenged with Escherichia coli F4. Animal 47(1):71. https://doi.org/10.1017/S1751731116001178
Yang KM, Jiang ZY, Zheng CT et al (2014) Effect of Lactobacillus plantarum on diarrhea and intestinal barrier function of young piglets challenged with enterotoxigenic Escherichia coli K88. J Anim Sci 92(4):1496–1503. https://doi.org/10.2527/jas.2013-6619
Lashani E, Davoodabadi A, Soltan Dallal MM (2020) Some probiotic properties of Lactobacillus species isolated from honey and their antimicrobial activity against foodborne pathogens. Vet Res Forum 11(2):121–126. https://doi.org/10.30466/vrf.2018.90418.2188
Kemgang TS, Kapila S, Shanmugam VP et al (2016) Fermented milk with probiotic Lactobacillus rhamnosus S1K3 (MTCC5957) protects mice from salmonella by enhancing immune and nonimmune protection mechanisms at intestinal mucosal level. J Nutr Biochem 30:62–73. https://doi.org/10.1016/j.jnutbio.2015.11.018
Filannino P, De AM, Di CR et al (2018) How Lactobacillus plantarum shapes its transcriptome in response to contrasting habitats. Env Microbiol 20:3700–3716. https://doi.org/10.1111/1462-2920.14372
Wang J, Ji H, Wang S et al (2018) Probiotic Lactobacillus plantarum promotes intestinal barrier function by strengthening the epithelium and modulating gut microbiota. Front Microbiol 9:1953. https://doi.org/10.3389/fmicb.2018.01953
Kim AR, Ahn KB, Yun CH et al (2019) Lactobacillus plantarum lipoteichoic acid inhibits oral multispecies biofilm. J Endod 45:310–315. https://doi.org/10.1016/j.joen.2018.12.007
Tran T, Everaert N, Bindelle J (2018) Review on the effects of potential prebiotics on controlling intestinal enteropathogens Salmonella and Escherichia coli in pig production. J Anim Physiol Anim Nutr 102(1):17–32. https://doi.org/10.1111/jpn.12666
Lawley TD, Bouley DM, Hoy YE et al (2008) Host transmission of Salmonella enterica serovar Typhimurium is controlled by virulence factors and indigenous intestinal microbiota. Infect Immun 76(1):403–416. https://doi.org/10.1128/IAI.01189-07
Barman M, Unold D, Shifley K et al (2008) Enteric salmonellosis disrupts the microbial ecology of the murine gastrointestinal tract. Infect Immun 76(3):907–915. https://doi.org/10.1128/IAI.01432-07
Borewicz KA, Kim HB, Singer RS et al (2015) Changes in the porcine intestinal microbiome in response to infection with Salmonella enterica and Lawsonia intracellularis. PLoS ONE 10(10):e0139106. https://doi.org/10.1371/journal.pone.0139106
Shi SH, Yang WT, Huang KY et al (2016) β-glucans from coriolus versicolor protect mice against S. typhimurium challenge by activation of macrophages. Int J Biol Macromol 86:352–361. https://doi.org/10.1016/j.ijbiomac.2016.01.058
DeSantis TZ, Hugenholtz P, Larsen N et al (2006) Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl Environ Microbiol 72(7):5069–5072. https://doi.org/10.1128/AEM.03006-05
Bokulich NA, Dillon MR, Bolyen E et al (2018) q2-sample-classifier: machine-learning tools for microbiome classification and regression. J Open Res Softw 3(30):934. https://doi.org/10.21105/joss.00934
Dougan G, John V, Palmer S et al (2011) Immunity to salmonellosis. Immunol Rev 240(1):196–210. https://doi.org/10.1111/j.1600-065X.2010.00999.x
Riddle MS, Porter CK (2012) Detection bias and the association between inflammatory bowel disease and Salmonella and Campylobacter infection. Gut 61(4):635. https://doi.org/10.1136/gutjnl-2011-300617
Gut AM, Vasiljevic T, Yeager T et al (2018) Salmonella infection - prevention and treatment by antibiotics and probiotic yeasts: a review. Microbiology (Reading, England) 164(11):1327–1344. https://doi.org/10.1099/mic.0.000709
Su JH, Zhu YH, Ren TY et al (2018) Distribution and antimicrobial resistance of Salmonella isolated from pigs with diarrhea in China. Microorganisms 6(4):117. https://doi.org/10.3390/microorganisms6040117
Allen SJ, Martinez EG, Gregorio GV et al (2010) Probiotics for treating acute infectious diarrhoea. Cochrane Database Syst Rev 11:CD003048. https://doi.org/10.1002/14651858.CD003048.pub3
Wen L, Duffy A (2017) Factors influencing the gut microbiota, inflammation, and type 2 diabetes. J Nutr 147(7):1468S-1475S. https://doi.org/10.3945/jn.116.240754
Chen HL, Zhao XY, Zhao GX et al (2020) Dissection of the cecal microbial community in chickens after Eimeria tenella infection. Parasit Vectors 13(1):56. https://doi.org/10.1186/s13071-020-3897-6
Kuda T, Yokota Y, Haraguchi Y et al (2019) Susceptibility of gut indigenous lactic acid bacteria in BALB/c mice to oral administered Lactobacillus plantarum. Int J Food Sci Nutr 70(1):53–62. https://doi.org/10.1080/09637486.2018.1471590
Grassl GA, Valdez Y, Bergstrom KS et al (2008) Chronic enteric salmonella infection in mice leads to severe and persistent intestinal fibrosis. Gastroenterology 134(3):768–780. https://doi.org/10.1053/j.gastro.2007.12.043
Yu Q, Liu X, Liu Y et al (2016) Defective small intestinal anion secretion, dipeptide absorption, and intestinal failure in suckling NBCe1-deficient mice. Pflugers Arch 468(8):1419–1432. https://doi.org/10.1007/s00424-016-1836-3
Wu H, Ye L, Lu X et al (2018) Lactobacillus acidophilus alleviated Salmonella-induced goblet cells loss and colitis by Notch pathway. Mol Nutr Food Res 62(22):e1800552. https://doi.org/10.1002/mnfr.201800552
Kimura N, Mimura F, Nishida S et al (1976) Studies on the relationship between intestinal gut microbiome and cecal coccidiosis in chicken. Poult Sci 55(4):1375–1383. https://doi.org/10.3382/ps.0551375
Arango Duque G, Descoteaux A (2014) Macrophage cytokines: involvement in immunity and infectious diseases. Front Immunol 5:491. https://doi.org/10.3389/fimmu.2014.00491
Anderson CJ, Kendall MM (2017) Salmonella enterica serovar Typhimurium strategies for host adaptation. Front Microbiol 8:1983. https://doi.org/10.3389/fmicb.2017.01983
Santos TT, Ornellas R, Acurcio LB et al (2021) Differential immune response of Lactobacillus plantarum 286 against Salmonella typhimurium infection in conventional and germ-free mice. Adv Exp Med Biol 1323:1–17. https://doi.org/10.1007/5584_2020_544
Santos RL (2014) Pathobiology of salmonella, intestinal microbiota, and the host innate immune response. Front Immunol 5:252. https://doi.org/10.3389/fimmu.2014.00252
Monack DM (2012) Salmonella persistence and transmission strategies. Curr Opin Microbiol 15(1):100–107. https://doi.org/10.1016/j.mib.2011.10.013
Wolter M, Steimle A, Parrish A et al (2021) Dietary modulation alters susceptibility to Listeria monocytogenes and Salmonella typhimurium with or without a gut microbiota. mSystems 6(6):e0071721. https://doi.org/10.1128/mSystems.00717-21
Saad N, Delattre C, Urdaci M et al (2013) An overview of the last advances in probiotic and prebiotic field. LWT - Food Sci Technol 50(1)
Kim YG, Sakamoto K, Seo SU et al (2017) Neonatal acquisition of Clostridia species protects against colonization by bacterial pathogens. Science (New York, N.Y.) 356(6335):315–319. https://doi.org/10.1126/science.aag2029
Toscano M, De Grandi R, Stronati L et al (2017) Effect of Lactobacillus rhamnosus HN001 and Bifidobacterium longum BB536 on the healthy gut microbiota composition at phyla and species level: a preliminary study. World J Gastroenterol 23(15):2696–2704. https://doi.org/10.3748/wjg.v23.i15.2696
Hills RD Jr, Pontefract BA, Mishcon HR et al (2019) Gut microbiome: profound implications for diet and disease. Nutrients 11(7):1613. https://doi.org/10.3390/nu11071613
Shin NR, Whon TW, Bae JW (2015) Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol 33(9):496–503. https://doi.org/10.1016/j.tibtech.2015.06.011
Yue Y, He Z, Zhou Y et al (2020) Lactobacillus plantarum relieves diarrhea caused by enterotoxin-producing Escherichia coli through inflammation modulation and gut microbiota regulation. Food Funct 11(12):10362–10374. https://doi.org/10.1039/d0fo02670k
Hakansson A, Molin G (2011) Gut microbiota and inflammation. Nutrients 3(6):637–682. https://doi.org/10.3390/nu3060637
Armstrong H, Bording-Jorgensen M, Dijk S et al (2018) The complex interplay between chronic inflammation, the microbiome, and cancer: understanding disease progression and what we can do to prevent it. Cancers 10(3):83. https://doi.org/10.3390/cancers10030083
Al Bander Z, Nitert MD, Mousa A et al (2020) The gut microbiota and inflammation: an overview. Int J Environ Res Public Health 17(20):7618. https://doi.org/10.3390/ijerph17207618
Yan Y, Zhou X, Guo K et al (2020) Chlorogenic acid protects against indomethacin-induced inflammation and mucosa damage by decreasing Bacteroides-derived LPS. Front Immunol 11:1125. https://doi.org/10.3389/fimmu.2020.01125
Dong N, Li X, Xue C et al (2019) Astragalus polysaccharides attenuated inflammation and balanced the gut microgut microbiome in mice challenged with Salmonella typhimurium. Int Immunopharmacol 74:105681. https://doi.org/10.1016/j.intimp.2019.105681
Funding
This work was supported by the Jilin Province Science and Technology Project (20200201109JC) and the National Natural Science Foundation of China (31302061).
Author information
Authors and Affiliations
Contributions
Jingtao Hu was corresponding authors to design the experiments. Rui-Han Liu and Ye Liao completed the experiments and wrote the paper. An-Qi Sun analyzed the data. All authors reviewed the results and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, RH., Sun, AQ., Liao, Y. et al. Lactiplantibacillus plantarum Regulated Intestinal Microbial Community and Cytokines to Inhibit Salmonella typhimurium Infection. Probiotics & Antimicro. Prot. 15, 1355–1370 (2023). https://doi.org/10.1007/s12602-022-09987-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12602-022-09987-5