Abstract
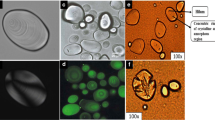
Starch is a major source of our daily diet and it is important to understand the molecular structure that plays a significant role in its wide number of applications. In this review article, microscopic structures of starch granules from potato, corn, rice canna, tania, wheat, sweet potato, and cassava are revealed using advanced microscopic techniques. Optical microscopy depicts the size and shape, polarization microscopy shows the anisotropy properties of starch granules, scanning electron microscopy (SEM) displays surface topography, and confocal microscopy is used to observe the three-dimensional internal structure of starch granules. The crystallinity of starch granules is revealed by second harmonic generation (SHG) microscopy and atomic force microscopy (AFM) provides mechanical properties including strength, texture, and elasticity. These properties play an important role in understanding the stability of starch granules under various processing conditions like heating, enzyme degradation, and hydration and determining its applications in various industries such as food packaging and textile industries.

















Similar content being viewed by others
References
Alcázar-Alay SC, Meireles MAA (2015) Physicochemical properties, modifications and applications of starches from different botanical sources. Food Sci Technol 35:215–236. https://doi.org/10.1590/1678-457X.6749
Ando T (2018) High-speed atomic force microscopy and its future prospects. Biophys Rev 10:285–292. https://doi.org/10.1007/s12551-017-0356-5
Ayoub A, Ohtani T, Sugiyama S (2006) Atomic force microscopy investigation of disorder process on rice starch granule surface. Starch-Stärke 58:475–479. https://doi.org/10.1002/star.200500471
Baldwin PM, Adler J, Davies MC, Melia CD (1998) High resolution imaging of starch granule surfaces by atomic force microscopy. J Cereal Sci 27:255–265
Blennow A, Hansen M, Schulz A, Jørgensen K, Donald AM, Sanderson J (2003) The molecular deposition of transgenically modified starch in the starch granule as imaged by functional microscopy. J Struct Biol 143:229–241. https://doi.org/10.1016/j.jsb.2003.08.009
Buleon A, Colonna P, Planchot V, Ball S (1998) Starch granules: structure and biosynthesis. Int J Biol Macromol 23:85–112. https://doi.org/10.1016/s0141-8130(98)00040-3
Cai C, Wei C (2013) In situ observation of crystallinity disruption patterns during starch gelatinization. Carbohydr Polym 92:469–478. https://doi.org/10.1016/j.carbpol.2012.09.073
Carlton RA (2011) Polarized light microscopy. In Pharmaceutical microscopy 7-64. https://doi.org/10.1007/978-1-4419-8831-7_2
Chang KC, Chiang YW, Yang CH, Liou JW (2012) Atomic force microscopy in biology and biomedicine. Tzu Chi Med J 24:162–169. https://doi.org/10.1016/j.tcmj.2012.08.002
Chen P, Yu L, Simon GP, Liu X, Dean K, Chen L (2011a) Internal structures and phase-transitions of starch granules during gelatinization. Carbohydr Polym 83:1975–1983. https://doi.org/10.1155/2015/397128
Chen Y, Huang S, Tang Z, Chen X, Zhang Z (2011b) Structural changes of cassava starch granules hydrolyzed by a mixture of α-amylase and glucoamylase. Carbohydr Polym 85:272–275. https://doi.org/10.1016/2Fj.carbpol.2011.01.047
Cox G (2011) Biological applications of second harmonic imaging. Biophys Rev 3:131. https://doi.org/10.1007/2Fs12551-011-0052-9
Demirkan ES, Mikami B, Adachi M, Higasa T, Utsumi S (2005) α-Amylase from B. amyloliquefaciens: purification, characterization, raw starch degradation and expression in E. coli. Process Biochem 40:2629–2636. https://doi.org/10.1016/j.procbio.2004.08.015
Egerton RF (2005) Physical principles of electron microscopy: 41. Springer, New York. https://doi.org/10.1007/b136495
Engel A, Lyubchenko Y, Müller D (1999) Atomic force microscopy: a powerful tool to observe biomolecules at work. Trends Cell Biol 9:77–80. https://doi.org/10.1016/S0962-8924(98)01415-9
Fannon JE, Gray JA, Gunawan N, Huber KC, BeMiller JN (2004) Heterogeneity of starch granules and the effect of granule channelization on starch modification. Cellulose 11:247–254. https://doi.org/10.1023/B:CELL.0000025399.66700.d7
Fonseca-Florido HA, Hernández-Ávilab J, Rodríguez-Hernández AI, Castro-Rosas J, Acevedo-Sandoval OA, Chavarria-Hernández N, Gómez-Aldapa CA (2017) Thermal, rheological, and mechanical properties of normal corn and potato starch blends. Int J Food Prop 20:611–622. https://doi.org/10.1080/10942912.2016.1171779
Gallant DJ, Bouchet B, Baldwin PM (1997) Microscopy of starch: evidence of a new level of granule organization. Carbohydr Polym 32:177–191. https://doi.org/10.1016/S0144-8617(97)00008-8
Gallant DJ, Bouchet B, Buleon A, Perez S (1992) Physical characteristics of starch granules and susceptibility to enzymatic degradation. Eur J Clin Nutr 46:3–16
Glaring MA, Koch CB, Blennow A (2006) Genotype-specific spatial distribution of starch molecules in the starch granule: a combined CLSM and SEM approach. Biomacromolecules 7:2310–2320. https://doi.org/10.1021/bm060216e
Jacobson MR, Obanni M, Bemiller JN (1997) Retrogradation of starches from different botanical sources. Cereal Chem 74:511–518. https://doi.org/10.1094/CCHEM.1997.74.5.511
Ji Y, Seetharaman K, White PJ (2004) Optimizing a small-scale corn-starch extraction method for use in the laboratory. Cereal Chem 81:55–58. https://doi.org/10.1094/CCHEM.2004.81.1.55
Li X, Gao W, Wang Y, Jiang Q, Huang L (2011) Granule structural, crystalline, and thermal changes in native Chinese yam starch after hydrolysis with two different enzymes—α-amylase and gluco-amylase. Starch-Stärke 63:75–82. https://doi.org/10.1002/star.201000104
Liu Y, Xu Y, Yan Y, Hu D, Yang L, Shen R (2015) Application of Raman spectroscopy in structure analysis and crystallinity calculation of corn starch. Starch-Stärke 67:612–619. https://doi.org/10.1002/star.201400246
Manca M, Woortman AJ, Loos K, Loi MA (2015) Imaging inclusion complex formation in starch granules using confocal laser scanning microscopy. Starch-Stärke 67:132–138. https://doi.org/10.1002/star.201400118
Mazumder N, Qiu J, Foreman MR, Romero CM, Török P, Kao FJ (2013) Stokes vector-based polarization resolved second harmonic microscopy of starch granules. Biomed Opt Express 4:538–547. https://doi.org/10.1364/BOE.4.000538
Mazumder N, Xiang L, Qiu J, Kao FJ (2017) Investigating starch gelatinization through Stokes vector resolved second harmonic generation microscopy. Sci Rep 7:45816. https://doi.org/10.1038/2Fsrep45816
Mazumder N, Yun-Xiang L, Qiu J, Kao FJ (2018) Revealing molecular structure of starch with Stokes-vector based second harmonic generation microscopy. J Opt 47:40–46. https://doi.org/10.1007/s12596-017-0419-9
Mehta SB, McQuilken M, La Riviere PJ, Occhipinti P, Verma A, Oldenbourg R, Tani T (2016) Dissection of molecular assembly dynamics by tracking orientation and position of single molecules in live cells. Proc Natl Acad Sci 113:E6352–E6361. https://doi.org/10.1073/pnas.1607674113
Mizutani G, Sonoda Y, Sano H, Sakamoto M, Takahashi T, Ushioda S (2000) Detection of starch granules in a living plant by optical second harmonic microscopy. J Lumin 87:824–826. https://doi.org/10.1016/S0022-2313(99)00428-7
Mu TH, Zhang M, Raad L, Sun HN, Wang C (2015) Effect of α-amylase degradation on physicochemical properties of pre-high hydrostatic pressure-treated potato starch. PloS One 10:e0143620. https://doi.org/10.1371/journal.pone.0143620
Murphy DB (2002) Fundamentals of light microscopy and electronic imaging. John Wiley & Sons
Ong MH, Blanshard JMV (1995) Texture determinants in cooked, parboiled rice. I: Rice starch amylose and the fine stucture of amylopectin. Journal of Cereal Science 21:251–260. https://doi.org/10.1006/jcrs.1995.0028
Oostergetel GT, Van Bruggen EF (1993) The crystalline domains in potato starch granules are arranged in a helical fashion. Carbohydrate Polymers 21:7–12. https://doi.org/10.1016/0144-8617(93)90110-P
Pérez S, Bertoft E (2010) The molecular structures of starch components and their contribution to the architecture of starch granules: a comprehensive review. Starch-Stärke 62:389–420. https://doi.org/10.1002/star.201000013
Perez-Rea D, Rojas C, Carballo S, Aguilar W, Bergenståhl B, Nilsson L (2013) Enzymatic hydrolysis of Canna indica, Manihot esculenta and Xanthosoma sagittifolium native starches below the gelatinization temperature. Starch-Stärke 65:151–161. https://doi.org/10.1002/star.201200103
Peroni-Okita FH, Gunning AP, Kirby A, Simão RA, Soares CA, Cordenunsi BR (2015) Visualization of internal structure of banana starch granule through AFM. Carbohydr Polym 128:32–40. https://doi.org/10.1016/j.carbpol.2015.04.019
Peroni-Okita FH, Cardoso MB, Agopian RG, Louro RP, Nascimento JR, Purgatto E, Cordenunsi BR (2013) The cold storage of green bananas affects the starch degradation during ripening at higher temperature. Carbohydrate Polymers 96:137–147. https://doi.org/10.1016/j.carbpol.2013.03.050
Pilling E, Smith AM (2003) Growth ring formation in the starch granules of potato tubers. Plant Physiol 132:365–371. https://doi.org/10.1104/pp.102.018044
Psilodimitrakopoulos S, Amat-Roldan I, Loza-Alvarez P, Artigas D (2010) Estimating the helical pitch angle of amylopectin in starch using polarization second harmonic generation microscopy. J Opt 12:084007. https://doi.org/10.1088/2040-8978/12/8/084007
Qin F, Man J, Xu B, Hu M, Gu M, Liu Q, Wei C (2011) Structural properties of hydrolyzed high-amylose rice starch by α-amylase from Bacillus licheniformis. J Agric Food Chem 59:12667–12673. https://doi.org/10.1021/jf203167f
Ridout MJ, Gunning AP, Parker ML, Wilson RH, Morris VJ (2002) Using AFM to image the internal structure of starch granules. Carbohydr Polym 50:123–132. https://doi.org/10.1016/S0144-8617(02)00021-8
Ridout MJ, Parker ML, Hedley CL, Bogracheva TY, Morris VJ (2003) Atomic force microscopy of pea starch granules: granule architecture of wild-type parent, r and rb single mutants, and the rrb double mutant. Carbohydr Res 338:2135–2147. https://doi.org/10.1016/s0008-6215(03)00309-4
Roy JK, Borah A, Mahanta CL, Mukherjee AK (2013) Cloning and overexpression of raw starch digesting α-amylase gene from Bacillus subtilis strain AS01a in Escherichia coli and application of the purified recombinant α-amylase (AmyBS-I) in raw starch digestion and baking industry. J Mol Catal B Enzym 97:118–129. https://doi.org/10.1016/j.molcatb.2013.07.019
Sarko A, Wu HC (1978) The crystal structures of A-, B-and C-polymorphs of amylose and starch. Starch-Stärke 30:73–78. https://doi.org/10.1002/star.19780300302
Shalin BM, Shribak M, Oldenbourg R (2013) Polarized light imaging of birefringence and diattenuation at high resolution and high sensitivity. J Optics 15:094007
Shaw SL (2006) Imaging the live plant cell. Plant J 45:573–598. https://doi.org/10.1111/j.1365-313X.2006.02653.x
Slepkov AD, Ridsdale A, Pegoraro AF, Moffatt DJ, Stolow A (2010) Multimodal CARS microscopy of structured carbohydrate biopolymers. Biomed Opt Express 1:1347–1357. https://doi.org/10.1364/BOE.1.001347
Souza PMD (2010) Application of microbial α-amylase in industry—a review. Braz J Microbiol 41:850–861. https://doi.org/10.1590/2FS1517-83822010000400004
Sujka M, Jamroz J (2009) α-Amylolysis of native potato and corn starches—SEM, AFM, nitrogen and iodine sorption investigations. LWT-Food Sci Technol 42:1219–1224. https://doi.org/10.1016/j.lwt.2009.01.016
Sundarram A, Murthy TPK (2014) α-Amylase production and applications: a review. J Appl Environ Microbiol 2:166–175. https://doi.org/10.12691/jaem-2-4-10
Tapia MS, Pérez E, Rodríguez PE, Guzmán R, Ducamp-Collin MN, Tran T, Rolland-Sabaté A (2012) Some properties of starch and starch edible films from under-utilized roots and tubers from the Venezuelan Amazons. J Cell Plast 48:526–544. https://doi.org/10.1177/2F0021955X12445291
Vahabi S, Salman BN, & Javanmard A (2013) Atomic force microscopy application in biological research: a review study. Iranian Journal of Medical Sciences 38(2):76
Velde FV, Riel J, Tromp RH (2002) Visualisation of starch granule morphologies using confocal scanning laser microscopy (CSLM). Journal of the Science of Food and Agriculture 82(13):1528–1536. https://doi.org/10.1002/jsfa.1165
Wang J, Guo K, Fan X, Feng G, Wei C (2018) Physicochemical properties of C-type starch from root tuber of Apios fortunei in comparison with maize, potato, and pea starches. Molecules 23:2132. https://doi.org/10.3390/2Fmolecules23092132
Yamada T, Hisamatsu M, Teranishi K, Katsuro K, Hasegawa N, Hayashi M (1995) Components of the porous maize starch granule prepared by amylase treatment. Starch-Stärke 47:358–361. https://doi.org/10.1002/star.19950470907
Yang R, Xi N, Fung CKM, Seiffert-Sinha K, Lai KWC, & Sinha AA (2011) The emergence of AFM applications to cell biology: How new technologies are facilitating investigation of human cells in health and disease at the nanoscale. Journal of Nanoscience letters 1(2):87.
Zhuo ZY, Liao CS, Huang CH, Yu JY, Tzeng YY, Lo W, Chu SW (2010) Second harmonic generation imaging—a new method for unravelling molecular information of starch. Journal of Structural Biology 171:88–94. https://doi.org/10.1016/j.jsb.2010.02.020
Acknowledgements
The authors thank Dr. K. Satyamoorthy, Director, Manipal School of Life Sciences (MSLS), MAHE, for his encouragement and Manipal Academy of Higher Education, Manipal, for providing the infrastructure and facilities. We thank Dr. K. K. Mahato, HoD, Department of Biophysics, MSLS, MAHE, for the fruitful discussion in preparation of this review article.
Funding
We thank Department of Biotechnology (DBT), Government of India, for the financial support (project number: BT/PR25099/NER/95/1014/2017).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chakraborty, I., Pallen, S., Shetty, Y. et al. Advanced microscopy techniques for revealing molecular structure of starch granules. Biophys Rev 12, 105–122 (2020). https://doi.org/10.1007/s12551-020-00614-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-020-00614-7