Abstract
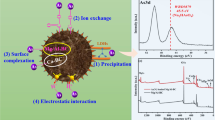
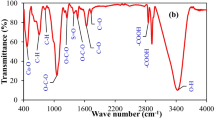
This study was aimed to enhance the phosphorus (P) sorption capability of biochar through embedding of Mg-Fe layered double hydroxide (LDH) particles within its matrix. The structure, morphology, and surface chemistry of the prepared LDH/biochar composite were investigated via X-ray diffraction (XRD), scanning electron microscopy (SEM), energy dispersive spectroscopy (EDS), and Fourier transform infrared (FTIR) analysis. The P sorption behavior of LDH/biochar composite was assessed in comparison with the raw biochar under batch conditions. The effects of initial P concentrations, pH levels, and contact times on P sorption were examined. Results showed that the P sorption on LDH/biochar composite was pH-dependent, and the maximum P sorption was found at the pH range of 2–4. The sorption isotherm and kinetic data were best fitted with the Langmuir and pseudo-second-order models, respectively. The maximum P sorption capacity (Qmax) improved from 1.39 mg g−1 for raw biochar to 17.46 mg g−1 for LDH/biochar composite, respectively. Also, the equilibrium contact time decreased from 8 h for raw biochar to 1 h for LDH/biochar composite, respectively. The sorption process followed electrostatic attraction, ligand exchange, and surface inner-sphere complex formation mechanisms. Overall, the results of the present study revealed that the synthesized LDH/biochar composite can be potentially used as a carbon-based sorbent for the removal of phosphorus from aqueous solutions.









Similar content being viewed by others
References
Ahmad M, Rajapaksha AU, Lim JE, Zhang M, Bolan N, Mohan D, Vithanage M, Lee SS, Ok YS (2014) Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere 99:19–33. https://doi.org/10.1016/j.chemosphere.2013.10.071
Bharali RK, Bhattacharyya KG (2015) Biosorption of fluoride on Neem (Azadirachta indica) leaf powder. J Environ Chem Eng 3:662–669. https://doi.org/10.1016/j.jece.2015.02.007
Cabrera A, Cox L, Spokas KA, Celis R, Hermosín MC, Cornejo J, Koskinen WC (2011) Comparative sorption and leaching study of the herbicides fluometuron and 4-chloro-2-methylphenoxyacetic acid (MCPA) in a soil amended with biochars and other sorbents. J Agric Food Chem 59:12550–12560. https://doi.org/10.1021/jf202713q
Chen B, Chen Z, Lv S (2011) A novel magnetic biochar efficiently sorbs organic pollutants and phosphate. Bioresour Technol 102:716–723. https://doi.org/10.1016/j.biortech.2010.08.067
Chitrakar R, Tezuka S, Sonoda A, Sakane K, Ooi K, Hirotsu T (2005) Adsorption of phosphate from seawater on calcined MgMn-layered double hydroxides. J Colloid Interface Sci 290:45–51. https://doi.org/10.1016/j.jcis.2005.04.025
Chowdhury SR, Yanful EK (2010) Arsenic and chromium removal by mixed magnetite-maghemite nanoparticles and the effect of phosphate on removal. J Environ Manag 91:2238–2247. https://doi.org/10.1016/j.jenvman.2010.06.003
Chubar N et al (2005) Adsorption of phosphate ions on novel inorganic ion exchangers. Colloids Surf A Physicochem Eng Asp 255:55–63. https://doi.org/10.1016/j.colsurfa.2004.12.015
Das J, Patra BS, Baliarsingh N, Parida KM (2006) Adsorption of phosphate by layered double hydroxides in aqueous solutions. Appl Clay Sci 32:252–260. https://doi.org/10.1016/j.clay.2006.02.005
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156:2–10. https://doi.org/10.1016/j.cej.2009.09.013
Ghosal PS, Gupta AK (2017) Determination of thermodynamic parameters from Langmuir isotherm constant-revisited. J Mol Liq 225:137–146. https://doi.org/10.1016/j.molliq.2016.11.058
Goh KH, Lim TT, Dong Z (2008) Application of layered double hydroxides for removal of oxyanions: a review. Water Res 42:1343–1368. https://doi.org/10.1016/j.watres.2007.10.043
Ho YS (2006) Second-order kinetic model for the sorption of cadmium onto tree fern: a comparison of linear and non-linear methods. Water Res 40:119–125. https://doi.org/10.1016/j.watres.2005.10.040
Jung KW, Hwang MJ, Ahn KH, Ok YS (2015a) Kinetic study on phosphate removal from aqueous solution by biochar derived from peanut shell as renewable adsorptive media. Int J Environ Sci Technol 12:3363–3372. https://doi.org/10.1007/s13762-015-0766-5
Jung KW, Hwang MJ, Jeong TU, Ahn KH (2015b) A novel approach for preparation of modified-biochar derived from marine macroalgae: dual purpose electro-modification for improvement of surface area and metal impregnation. Bioresour Technol 191:342–345. https://doi.org/10.1016/j.biortech.2015.05.052
Jung KW, Jeong TU, Hwang MJ, Kim K, Ahn KH (2015c) Phosphate adsorption ability of biochar/Mg-Al assembled nanocomposites prepared by aluminum-electrode based electro-assisted modification method with MgCl2 as electrolyte. Bioresour Technol 198:603–610. https://doi.org/10.1016/j.biortech.2015.09.068
Lalley J, Han C, Li X, Dionysiou DD, Nadagouda MN (2016) Phosphate adsorption using modified iron oxide-based sorbents in lake water: kinetics, equilibrium, and column tests. Chem Eng J 284:1386–1396. https://doi.org/10.1016/j.biortech.2015.09.068
Lee JW, Kidder M, Evans BR, Paik S, Buchanan Iii AC, Garten CT, Brown RC (2010) Characterization of biochars produced from cornstovers for soil amendment. Environ Sci Technol 44:7970–7974. https://doi.org/10.1021/es101337x
Li W, Pierre-Louis AM, Kwon KD, Kubicki JD, Strongin DR, Phillips BL (2013) Molecular level investigations of phosphate sorption on corundum (α-Al2O3) by 31 P solid state NMR, ATR-FTIR and quantum chemical calculation. Geochim Cosmochim Acta 107:252–266. https://doi.org/10.1016/j.gca.2013.01.007
Li R, Wang JJ, Zhou B, Awasthi MK, Ali A, Zhang Z, Gaston LA, Lahori AH, Mahar A (2016) Enhancing phosphate adsorption by Mg/Al layered double hydroxide functionalized biochar with different Mg/Al ratios. Sci Total Environ 559:121–129. https://doi.org/10.1016/j.scitotenv.2016.03.151
Liu H, Sun X, Yin C, Hu C (2008) Removal of phosphate by mesoporous ZrO2. J Hazard Mater 151:616–622. https://doi.org/10.1016/j.jhazmat.2007.06.033
Lǚ J, Liu H, Liu R, Zhao X, Sun L, Qu J (2013) Adsorptive removal of phosphate by a nanostructured Fe-Al-Mn trimetal oxide adsorbent. Powder Technol 233:146–154. https://doi.org/10.1016/j.powtec.2012.08.024
Luengo CV, Volpe MA, Avena MJ (2017) High sorption of phosphate on Mg-Al layered double hydroxides: Kinetics and equilibrium. J Environ Chem Eng 5:4656–4662. https://doi.org/10.1016/j.jece.2017.08.051
Lung YF, Sun YS, Lin CK, Uan JY, Huang HH (2016) Synthesis of Mg-Fe-Cl hydrotalcite-like nanoplatelets as an oral phosphate binder: evaluations of phosphorus intercalation activity and cellular cytotoxicity. Sci Rep 6:32458. https://doi.org/10.1038/srep32458
Mohan D, Sarswat A, Singh VK, Alexandre-Franco M, Pittman CU (2011) Development of magnetic activated carbon from almond shells for trinitrophenol removal from water. Chem Eng J 172:1111–1125. https://doi.org/10.1016/j.cej.2011.06.054
Mohan D, Sarswat A, Ok YS, Pittman CU (2014) Organic and inorganic contaminants removal from water with biochar, a renewable, low cost and sustainable adsorbent A critical review. Bioresour Technol 160:191–202. https://doi.org/10.1016/j.biortech.2014.01.120
Nalawade P, Aware B, Kadam V, Hirlekar R (2009) Layered double hydroxides: A review. J Sci Ind Res 68:268–272
Novais SV, Zenero MDO, Tronto J, Conz RF, Cerri CEP (2018) Poultry manure and sugarcane straw biochars modified with MgCl2 for phosphorus adsorption. J Environ Manag 214:36–44. https://doi.org/10.1016/j.jenvman.2018.02.088
Oliveira FR, Patel AK, Jaisi DP, Adhikari S, Lu H, Khanal SK (2017) Environmental application of biochar: current status and perspectives. Bioresour Technol 246:110–122. https://doi.org/10.1016/j.biortech.2017.08.122
Park JH, Ok YS, Kim SH, Cho JS, Heo JS, Delaune RD, Seo DC (2015) Evaluation of phosphorus adsorption capacity of sesame straw biochar on aqueous solution: influence of activation methods and pyrolysis temperatures. Environ Geochem Health 37:969–983. https://doi.org/10.1007/s10653-015-9709-9
Peleka E, Deliyanni E (2009) Adsorptive removal of phosphates from aqueous solutions. Desalination 245:357–371. https://doi.org/10.1016/j.desal.2008.04.050
Rahmani A, Mousavi HZ, Fazli M (2010) Effect of nanostructure alumina on adsorption of heavy metals. Desalination 253:94–100. https://doi.org/10.1016/j.desal.2009.11.027
Ren J, Li N, Li L, An JK, Zhao L, Ren NQ (2015) Granulation and ferric oxides loading enable biochar derived from cotton stalk to remove phosphate from water. Bioresour Technol 178:119–125. https://doi.org/10.1016/j.biortech.2014.09.071
Rice E, Bridgewater L (2012) Association APH Standard methods for the examination of water and wastewater. American Public Health Association, Washington DC
Seida Y, Nakano Y (2002) Removal of phosphate by layered double hydroxides containing iron. Water Res 36:1306–1312. https://doi.org/10.1016/S0043-1354(01)00340-2
Sizmur T, Fresno T, Akgül G, Frost H, Moreno-Jiménez E (2017) Biochar modification to enhance sorption of inorganics from water. Bioresour Technol 246:34–47. https://doi.org/10.1016/j.biortech.2017.07.082
Soliemanzadeh A, Fekri M (2017) Synthesis of clay-supported nanoscale zero-valent iron using green tea extract for the removal of phosphorus from aqueous solutions. Chin J Chem Eng 25:924–930. https://doi.org/10.1016/j.cjche.2016.12.006
Soliemanzadeh A, Fekri M, Bakhtiary S, Mehrizi MH (2016) Biosynthesis of iron nanoparticles and their application in removing phosphorus from aqueous solutions. Chem Ecol 32:286–300. https://doi.org/10.1080/02757540.2016.1139091
Sparks DL (2003) Environmental soil chemistry. Academic Press, Elsevier
Triantafyllidis KS, Peleka EN, Komvokis VG, Mavros PP (2010) Iron-modified hydrotalcite-like materials as highly efficient phosphate sorbents. J Colloid Interface Sci 342:427–436. https://doi.org/10.1016/j.jcis.2009.10.063
Vaccari A (1998) Preparation and catalytic properties of cationic and anionic clays. Catal Today 41:53–71. https://doi.org/10.1016/S0920-5861(98)00038-8
Wan D, Liu H, Liu R, Qu J, Li S, Zhang J (2012) Adsorption of nitrate and nitrite from aqueous solution onto calcined (Mg-Al) hydrotalcite of different Mg/Al ratio. Chem Eng J 195-196:241–247. https://doi.org/10.1016/j.cej.2012.04.088
Wang Z, Nie E, Li J, Yang M, Zhao Y, Luo X, Zheng Z (2012) Equilibrium and kinetics of adsorption of phosphate onto iron-doped activated carbon. Environ Sci Pollut Res 19:2908–2917. https://doi.org/10.1007/s11356-012-0799-y
Wang Z, Shi M, Li J, Zheng Z (2014) Influence of moderate pre-oxidation treatment on the physical, chemical and phosphate adsorption properties of iron-containing activated carbon. J Environ Sci 26:519–528. https://doi.org/10.1016/S1001-0742(13)60440-4
Wang Z, Shen D, Shen F, Li T (2016) Phosphate adsorption on lanthanum loaded biochar. Chemosphere 150:1–7. https://doi.org/10.1016/j.chemosphere.2016.02.004
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89:31–60
Xu Y, Dai Y, Zhou J, Xu ZP, Qian G, Lu GQM (2010) Removal efficiency of arsenate and phosphate from aqueous solution using layered double hydroxide materials: intercalation vs. precipitation. J Mater Chem 20:4684–4691. https://doi.org/10.1039/B926239C
Yang M, Lin J, Zhan Y, Zhang H (2014) Adsorption of phosphate from water on lake sediments amended with zirconium-modified zeolites in batch mode. Ecol Eng 71:223–233. https://doi.org/10.1016/j.ecoleng.2014.07.035
Yang F, Zhang S, Sun Y, Tsang DCW, Cheng K, Ok YS (2019) Assembling biochar with various layered double hydroxides for enhancement of phosphorus recovery. J Hazard Mater 365:665–673. https://doi.org/10.1016/j.jhazmat.2018.11.047
Yao Y, Gao B, Inyang M, Zimmerman AR, Cao X, Pullammanappallil P, Yang L (2011) Biochar derived from anaerobically digested sugar beet tailings: characterization and phosphate removal potential. Bioresour Technol 102:6273–6278. https://doi.org/10.1016/j.biortech.2011.03.006
Yao Y, Gao B, Zhang M, Inyang M, Zimmerman AR (2012) Effect of biochar amendment on sorption and leaching of nitrate, ammonium, and phosphate in a sandy soil. Chemosphere 89:1467–1471. https://doi.org/10.1016/j.chemosphere.2012.06.002
Zhang G, Liu H, Liu R, Qu J (2009) Removal of phosphate from water by a Fe-Mn binary oxide adsorbent. J Colloid Interface Sci 335:168–174. https://doi.org/10.1016/j.jcis.2009.03.019
Zhang M, Gao B, Yao Y, Inyang M (2013) Phosphate removal ability of biochar/MgAl-LDH ultra-fine composites prepared by liquid-phase deposition. Chemosphere 92:1042–1047. https://doi.org/10.1016/j.chemosphere.2013.02.050
Zhang MM, Liu YG, Li TT, Xu WH, Zheng BH, Tan XF, Wang H, Guo YM, Guo FY, Wang SF (2015) Chitosan modification of magnetic biochar produced from Eichhornia crassipes for enhanced sorption of Cr(vi) from aqueous solution. RSC Adv 5:46955–46964. https://doi.org/10.1039/C5RA02388B
Zheng W, Guo M, Chow T, Bennett DN, Rajagopalan N (2010) Sorption properties of greenwaste biochar for two triazine pesticides. J Hazard Mater 181:121–126. https://doi.org/10.1016/j.jhazmat.2010.04.103
Author information
Authors and Affiliations
Corresponding author
Additional information
Editorial handling: Broder J. Merkel
Rights and permissions
About this article
Cite this article
Bolbol, H., Fekri, M. & Hejazi-Mehrizi, M. Layered double hydroxide–loaded biochar as a sorbent for the removal of aquatic phosphorus: behavior and mechanism insights. Arab J Geosci 12, 503 (2019). https://doi.org/10.1007/s12517-019-4694-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12517-019-4694-4