Abstract
Background
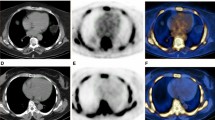
Previous studies have suggested the role of microcalcifications in plaque vulnerability. This exploratory study sought to assess the potential of hybrid positron-emission tomography (PET)/magnetic resonance imaging (MRI) using 18F-sodium fluoride (18F-NaF) to check simultaneously 18F-NaF uptake, a marker of microcalcifications, and morphological criteria of vulnerability.
Methods and results
We included 12 patients with either recently symptomatic or asymptomatic carotid stenosis. All patients underwent 18F-NaF PET/MRI. 18F-NaF target-to-background ratio (TBR) was measured in culprit and nonculprit (including contralateral plaques of symptomatic patients) plaques as well as in other arterial walls. Morphological criteria of vulnerability were assessed on MRI. Mineral metabolism markers were also collected. 18F-NaF uptake was higher in culprit compared to nonculprit plaques (median TBR 2.6 [2.2-2.8] vs 1.7 [1.3-2.2]; P = 0.03) but was not associated with morphological criteria of vulnerability on MRI. We found a positive correlation between 18F-NaF uptake and calcium plaque volume and ratio but not with circulating tissue-nonspecific alkaline phosphatase (TNAP) activity and inorganic pyrophosphate (PPi) levels. 18F-NaF uptake in the other arterial walls did not differ between symptomatic and asymptomatic patients.
Conclusions
18F-NaF PET/MRI may be a promising tool for providing additional insights into the plaque vulnerability.




Similar content being viewed by others
Abbreviations
- 18F-NaF:
-
18F-sodium fluoride
- AP:
-
Alkaline phosphatase
- BMI:
-
Body mass index
- BW:
-
Bandwidth
- CE-MRA:
-
Contrast-enhanced
- CRP:
-
C-reactive protein
- CT:
-
Computed tomography
- DUS:
-
Doppler ultrasound
- FC:
-
Fibrous cap
- FLASH:
-
Fast-low angle shot
- FOV:
-
Field of view
- IPH:
-
Intraplaque hemorrhage
- IQR:
-
Interquartile range
- LDL:
-
Low-density lipoprotein
- LRNC:
-
Lipid-rich necrotic core
- MR:
-
Angiography
- MRI:
-
Magnetic resonance imaging
- NA:
-
Number of averages
- NASCET:
-
North American Symptomatic Carotid Endarterectomy Trial
- NIHSS:
-
National institutes of health stroke scale
- PET:
-
Positron-emission tomography
- PPi:
-
Inorganic pyrophosphate
- ROI:
-
Region of interest
- SD:
-
Standard Deviation
- SPAIR:
-
Spectral attenuated inversion recovery
- SUVmax:
-
Maximum standard uptake value
- TE:
-
Echo time
- TBR:
-
Target-to-background ratio
- TIA:
-
Transient ischemic attack
- TNAP:
-
Tissue-nonspecific alkaline phosphatase
- TOF:
-
Time-of-flight angiography
- TR:
-
Repetition time
- VSMC:
-
Vascular smooth muscle cell
References
Petty GW, Brown RD, Whisnant JP, Sicks JD, O’Fallon WM, Wiebers DO. Ischemic stroke subtypes: A population-based study of incidence and risk factors. Stroke. 1999;30:2513–6.
Stary HC, Chandler AB, Dinsmore RE, Fuster V, Glagov S, Insull W, et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis: A report from the committee on vascular lesions of the council on arteriosclerosis. Am Heart Assoc. Circ. 1995;92:1355–74.
Nighoghossian N, Derex L, Douek P. The vulnerable carotid artery plaque: Current imaging methods and new perspectives. Stroke. 2005;36:2764–72.
Rudd JHF, Warburton EA, Fryer TD, Jones HA, Clark JC, Antoun N, et al. Imaging atherosclerotic plaque inflammation with [18 F]-fluorodeoxyglucose positron emission tomography. Circulation. 2002;105:2708–11.
Davies JR, Rudd JHF, Fryer TD, Graves MJ, Clark JC, Kirkpatrick PJ, et al. Identification of culprit lesions after transient ischemic attack by combined 18 F fluorodeoxyglucose positron-emission tomography and high-resolution magnetic resonance imaging. Stroke. 2005;36:2642–7.
Tawakol A, Migrino RQ, Bashian GG, Bedri S, Vermylen D, Cury RC, et al. In vivo 18F-fluorodeoxyglucose positron emission tomography imaging provides a noninvasive measure of carotid plaque inflammation in patients. J Am Coll Cardiol. 2006;48:1818–24.
Marnane M, Merwick A, Sheehan OC, Hannon N, Foran P, Grant T, et al. Carotid plaque inflammation on 18F-fluorodeoxyglucose positron emission tomography predicts early stroke recurrence. Ann Neurol. 2012;71:709–18.
Taqueti VR, Di Carli MF, Jerosch-Herold M, Sukhova GK, Murthy VL, Folco EJ, et al. Increased microvascularization and vessel permeability associate with active inflammation in human atheromata. Circ Cardiovasc Imaging. 2014;7:920–9.
Chowdhury MM, Tarkin JM, Evans NR, Le E, Warburton EA, Hayes PD, et al. 18 F-FDG uptake on PET/CT in symptomatic versus asymptomatic carotid disease: A meta-analysis. Eur J Vasc Endovasc Surg. 2018;56:172–9.
Irkle A, Vesey AT, Lewis DY, Skepper JN, Bird JLE, Dweck MR, et al. Identifying active vascular microcalcification by 18F-sodium fluoride positron emission tomography. Nat Commun. 2015;6:7495.
Vesey AT, Jenkins WSA, Irkle A, Moss A, Sng G, Forsythe RO, et al. 18 F-Fluoride and 18 F-fluorodeoxyglucose positron emission tomography after transient ischemic attack or minor ischemic stroke: Case–control study. Circ Cardiovasc Imaging. 2017;10:e004976.
Derlin T, Toth Z, Papp L, Wisotzki C, Apostolova I, Habermann CR, et al. Correlation of inflammation assessed by 18F-FDG PET, active mineral deposition assessed by 18F-fluoride PET, and vascular calcification in atherosclerotic plaque: A dual-tracer PET/CT study. J Nucl Med. 2011;52:1020–7.
Li X, Heber D, Gonzalez JC, Karanikas G, Mayerhoefer ME, Rasul S, et al. Association between osteogenesis and inflammation during the progression of calcified plaque evaluated by 18 F-fluoride and 18 F-FDG. J Nucl Med. 2017;58:968–74.
Joshi NV, Vesey AT, Williams MC, Shah ASV, Calvert PA, Craighead FHM, et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: A prospective clinical trial. The Lancet. 2014;383:705–13.
Adamson PD, Vesey AT, Joshi NV, Newby DE, Dweck MR. Salt in the wound: (18)F-fluoride positron emission tomography for identification of vulnerable coronary plaques. Cardiovasc Diagn Ther. 2015;5:150–5.
Lee JM, Bang J-I, Koo B-K, Hwang D, Park J, Zhang J, et al. Clinical relevance of 18 F-sodium fluoride positron-emission tomography in noninvasive identification of high-risk plaque in patients with coronary artery disease. Circ Cardiovasc Imaging. 2017;10:e006704.
Li L, Li X, Jia Y, Fan J, Wang H, Fan C, et al. Sodium-fluoride PET-CT for the non-invasive evaluation of coronary plaques in symptomatic patients with coronary artery disease: a cross-correlation study with intravascular ultrasound. Eur J Nucl Med Mol Imaging. 2018;45:2181–9.
Quirce R, Martínez-Rodríguez I, De Arcocha Torres M, Jiménez-Bonilla JF, Banzo I, Rebollo M, et al. Contribution of 18F-sodium fluoride PET/CT to the study of the carotid atheroma calcification. Rev Esp Med Nucl E Imagen Mol. 2013;32:22–5.
Cocker MS, Spence JD, Hammond R, Wells G, deKemp RA, Lum C, et al. [18 F]-NaF PET/CT identifies active calcification in carotid plaque. JACC Cardiovasc Imaging. 2017;10:486–8.
Zhang Y, Li H, Jia Y, Yang P, Zhao F, Wang W, et al. Noninvasive assessment of carotid plaques calcification by 18 F-sodium fluoride accumulation: Correlation with pathology. J Stroke Cerebrovasc Dis. 2018;27:1796–801.
Hop H, de Boer SA, Reijrink M, Kamphuisen PW, de Borst MH, Pol RA, et al. 18F-sodium fluoride positron emission tomography assessed microcalcifications in culprit and non-culprit human carotid plaques. J Nucl Cardiol. 2019;26:1064–75.
den Hartog AG, Bovens SM, Koning W, Hendrikse J, Luijten PR, Moll FL, et al. Current status of clinical magnetic resonance imaging for plaque characterisation in patients with carotid artery stenosis. Eur J Vasc Endovasc Surg. 2013;45:7–21.
Saba L, Saam T, Jäger HR, Yuan C, Hatsukami TS, Saloner D, et al. Imaging biomarkers of vulnerable carotid plaques for stroke risk prediction and their potential clinical implications. Lancet Neurol. 2019;18:559–72.
Orimo H. The mechanism of mineralization and the role of alkaline phosphatase in health and disease. J Nippon Med Sch Nippon Ika Daigaku Zasshi. 2010;77:4–12.
Ryu W-S, Lee S-H, Kim CK, Kim BJ, Yoon B-W. Increased serum alkaline phosphatase as a predictor of long-term mortality after stroke. Neurology. 2010;75:1995–2002.
Tonelli M, Curhan G, Pfeffer M, Sacks F, Thadhani R, Melamed ML, et al. Relation between alkaline phosphatase, serum phosphate, and all-cause or cardiovascular mortality. Circulation. 2009;120:1784–92.
Barnett HJM. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med. 1991;325:445–53.
Kernan WN, Ovbiagele B, Black HR, Bravata DM, Chimowitz MI, Ezekowitz MD, et al. Guidelines for the prevention of stroke in patients with stroke and transient ischemic attack: A guideline for healthcare professionals from the american heart association/american stroke Association. Stroke. 2014;45:2160–236.
Fujimoto K, Norikane T, Yamamoto Y, Takami Y, Mitamura K, Okada M, et al. Association between carotid 18F-NaF and 18F-FDG uptake on PET/CT with ischemic vascular brain disease on MRI in patients with carotid artery disease. Ann Nucl Med. 2019;33:907–15.
Norikane T, Yamamoto Y, Maeda Y, Okada M, Nishiyama Y. Correlation of noninvasive imaging of vulnerable carotid artery plaque using NaF and FDG PET/CT and black-blood MRI with cerebral ischemia on brain MRI. J Nucl Med. 2018;59:40–40.
Vengrenyuk Y, Carlier S, Xanthos S, Cardoso L, Ganatos P, Virmani R, et al. A hypothesis for vulnerable plaque rupture due to stress-induced debonding around cellular microcalcifications in thin fibrous caps. Proc Natl Acad Sci. 2006;103:14678–83.
Kelly-Arnold A, Maldonado N, Laudier D, Aikawa E, Cardoso L, Weinbaum S. Revised microcalcification hypothesis for fibrous cap rupture in human coronary arteries. Proc Natl Acad Sci. 2013;110:10741–6.
Roijers RB, Debernardi N, Cleutjens JPM, Schurgers LJ, Mutsaers PHA, van der Vusse GJ. Microcalcifications in early intimal lesions of atherosclerotic human coronary arteries. Am J Pathol. 2011;178:2879–87.
den Harder AM, Wolterink JM, Bartstra JW, Spiering W, Zwakenberg SR, Beulens JW, et al. Vascular uptake on 18F-sodium fluoride positron emission tomography: Precursor of vascular calcification? J Nucl Cardiol. 2020. https://doi.org/10.1007/s12350-020-02031-5.
Murshed M, Harmey D, Millán JL, McKee MD, Karsenty G. Unique coexpression in osteoblasts of broadly expressed genes accounts for the spatial restriction of ECM mineralization to bone. Genes Dev. 2005;19:1093–104.
Sheen CR, Kuss P, Narisawa S, Yadav MC, Nigro J, Wang W, et al. Pathophysiological role of vascular smooth muscle alkaline phosphatase in medial artery calcification: Role of TNAP in medial vascular calcification. J Bone Miner Res. 2015;30:824–36.
Libby P. Inflammation in atherosclerosis. Arterioscler Thromb Vasc Biol. 2012;32:2045–51.
Arbab-Zadeh A, Fuster V. The Myth of the “Vulnerable Plaque”. J Am Coll Cardiol. 2015;65:846–55.
Chung AG, Frye JB, Zbesko JC, Constantopoulos E, Hayes M, Figueroa AG, et al. Liquefaction of the brain following stroke shares a similar molecular and morphological profile with atherosclerosis and mediates secondary neurodegeneration in an osteopontin-dependent mechanism. Eneuro. 2018;5:ENEURO.0076.
Lee YS, Kim JS, Kim KM, Kang JH, Lim SM, Kim H-J. Performance measurement of PSF modeling reconstruction (True X) on siemens biograph TruePoint TrueV PET/CT. Ann Nucl Med. 2014;28:340–8.
Schramm G, Maus J, Hofheinz F, Petr J, Lougovski A, Beuthien-Baumann B, et al. Correction of quantification errors in pelvic and spinal lesions caused by ignoring higher photon attenuation of bone in [18 F]NaF PET/MR. Med Phys. 2015;42:6468–76.
Kelly PJ, Camps-Renom P, Giannotti N, Martí-Fàbregas J, McNulty JP, Baron J-C, et al. A risk score including carotid plaque inflammation and stenosis severity improves identification of recurrent stroke. Stroke. 2020;51:838–45.
Debray J, Chang L, Marquès S, Pellet-Rostaing S, Le Duy D, Mebarek S, et al. Inhibitors of tissue-nonspecific alkaline phosphatase: Design, synthesis, kinetics, biomineralization and cellular tests. Bioorg Med Chem. 2013;21:7981–7.
Acknowledgements
We thank Thomas Troalen, application engineer Siemens Healthcare France, for his assistance in the development of the sequences used in this study, Curium for providing 18F-NaF (CISNAF 100 MBq/mL solution for injection, Cis bio international), Karen Reilly for proofreading the English, Morgane Bouin (Cellule Recherche Imagerie) for data collection and the French National ‘invest for the future’ programs: LILI – Lyon Integrated Life Imaging: hybrid MR-PET ANR-11-EQPX-0026 and CESAME - Brain and Mental Health ANR-10-IBHU-0003. Human biological samples and associated data were obtained from NeuroBioTec (CRB-HCL, Lyon France, Biobank BB-0033-00046).
Disclosure
The authors declare that they have no conflict of interest.
Funding
CREATIS CNRS Lab grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors of this article have provided a PowerPoint file, available for download at SpringerLink, which summarises the contents of the paper and is free for re-use at meetings and presentations. Search for the article DOI on SpringerLink.com.
The authors have also provided an audio summary of the article, which is available to download as ESM, or to listen to via the JNC/ASNC Podcast.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mechtouff, L., Sigovan, M., Douek, P. et al. Simultaneous assessment of microcalcifications and morphological criteria of vulnerability in carotid artery plaque using hybrid 18F-NaF PET/MRI. J. Nucl. Cardiol. 29, 1064–1074 (2022). https://doi.org/10.1007/s12350-020-02400-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-020-02400-0