Abstract
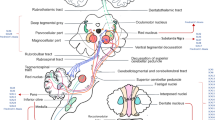
Ataxia-telangiectasia is the second most common autosomal recessive hereditary ataxia, with an estimated incidence of 1 in 100,000 births. Besides ataxia and ocular telangiectasias, eye movement abnormalities have long been associated with this disorder and is frequently present in almost all patients. A handful of studies have described the phenomenology of ocular motor deficits in ataxia-telangiectasia. Contemporary literature linked their physiology to cerebellar dysfunction and secondary abnormalities at the level of brainstem. These studies, while providing a proof of concept of ocular motor physiology in disease, i.e., ataxia-telangiectasia, also advanced our understanding of how the cerebellum works. Here, we will summarize the clinical abnormalities seen with ataxia-telangiectasia in each subtype of eye movements and subsequently describe the underlying pathophysiology. Finally, we will review how these deficits are linked to abnormal cerebellar function and how it allows better understanding of the cerebellar physiology.






Similar content being viewed by others
References
Swift M, Morrell D, Cromartie E, Chamberlin AR, Skolnick MH, Bishop DT. The incidence and gene frequency of ataxia-telangiectasia A-T in the United States. Am J Hum Genet. 1986;39(5):573–83.
Jayadev S, Bird TD. Hereditary ataxias: overview. Genet Med. 2013;15(9):673–83.
Boder E, Sedgwick RP. Ataxia-Telangiectasia A-T: a familial syndrome of progressive cerebellar ataxia, oculocutaneous telangiectasia and frequent pulmonary infection. Pediatrics. 1958;21(4):526–54.
McFarlin DE, Strober W, Waldmann TA. Ataxia-telangiectasia A-T. Medicine (Baltimore). 1972;51(4):281–314.
Biemond A. Paleo cerebellar atrophy with extrapyramidal manifestations in association with bronchiectasis and telangiectasia of the conjunctiva bulbi as a familial syndrome. Van Bogaert Radermecker J Eds Proc First Int Congr. 1957;206.
Savitsky K, Bar-Shira A, Gilad S, Rotman G, Ziv Y, Vanagaite L, et al. A single ataxia telangiectasia gene with a product similar to PI-3 kinase. Science. 1995;268(5218):1749–53.
Byrd PJ, McConville CM, Cooper P, Parkhill J, Stankovic T, McGuire GM, et al. Mutations revealed by sequencing the 5′ half of the gene for ataxia telangiectasia. Hum Mol Genet. 1996;5(1):145–9.
Moin M, Aghamohammadi A, Kouhi A, Tavassoli S, Rezaei N, Ghaffari S-R, et al. Ataxia-telangiectasia A-T in Iran: clinical and laboratory features of 104 patients. Pediatr Neurol. 2007;37(1):21–8.
Smith JL, Cogan DG. Ataxia-Telangiectasia A-T. AMA Arch Ophthalmol. 1959;62(3):364–9.
Cogan DG, Chu FC, Reingold D, Barranger J. Ocular motor signs in some metabolic diseases. Arch Ophthalmol. 1981;99(10):1802–8.
Federighi P, Ramat S, Rosini F, Pretegiani E, Federico A, Rufa A. Characteristic eye movements in ataxia-telangiectasia-like disorder: an explanatory hypothesis. Front Neurol. 2017;8:596.
Hyams SW, Reisner SH, Neumann E. The eye signs in ataxia-telangiectasia A-T. Am J Ophthalmol. 62(6):1118–24.
Boder E, Sedgwick RP. Ataxia telangiectasia: a review of 150 cases. Intern Cong Ment Retard. 1964.
Lewis RF, Lederman HM, Crawford TO. Ocular motor abnormalities in ataxia telangiectasia. Ann Neurol. 1999;46(3):287–95.
Baloh RW, Yee RD, Boder E. Eye movements in ataxia-telangiectasia. Neurology. 1978;28(11):1099–104.
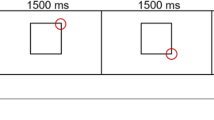
Harris CM, Shawkat F, Russell-Eggitt I, Wilson J, Taylor D. Intermittent horizontal saccade failure (‘ocular motor apraxia’) in children. Br J Ophthalmol. 1996;80(2):151–8.
Zee DS, Yee RD, Singer HS. Congenital ocular motor apraxia. Brain J Neurol. 1977;100(3):581–99.
Reed H, Israels S. Congenital ocular motor apraxia: a form of horizontal gaze palsy. Br J Ophthalmol. 1956;40(7):444–8.
Riopel DA. Congenital ocular motor apraxia* *from the division of ophthalmology, College of Medicine, University of Florida. Am J Ophthalmol. 1963;55(3):511–4.
ROBLES J. Congenital ocular motor apraxia in identical twins. Arch Ophthalmol. 1966;75(6):746–9.
Orrison WW, Robertson WC. Congenital ocular motor apraxia: a possible disconnection syndrome. Arch Neurol. 1979;36(1):29–31.
Leigh RJ, Zee DS. The neurology of eye movements: Oxford University Press; 2015. 1137 p
Van Gisbergen JA, Robinson DA, Gielen S. A quantitative analysis of generation of saccadic eye movements by burst neurons. J Neurophysiol. 1981;45(3):417–42.
Hepp K, Henn V. Spatio-temporal recoding of rapid eye movement signals in the monkey paramedian pontine reticular formation (PPRF). Exp Brain Res. 1983;52(1):105–20.
Henn V, Hepp K, Vilis T. Rapid eye movement generation in the primate. Physiology, pathophysiology, and clinical implications. Rev Neurol (Paris). 1989;145(8–9):540–5.
Ramat S, Leigh RJ, Zee DS, Optican LM. Ocular oscillations generated by coupling of brainstem excitatory and inhibitory saccadic burst neurons. Exp Brain Res. 2005;160(1):89–106.
Shaikh AG, Marti S, Tarnutzer AA, Palla A, Crawford T, Straumann D, et al. Gaze fixation deficits and their implication in ataxia-telangiectasia A-T. J Neurol Neurosurg Psychiatry. 2009;80(8):858–64.
Shook BL, Schlag-Rey M, Schlag J. Direct projection from the supplementary eye field to the nucleus raphe interpositus. Exp Brain Res. 1988;73(1):215–8.
Stanton GB, Goldberg ME, Bruce CJ. Frontal eye field efferents in the macaque monkey: I. subcortical pathways and topography of striatal and thalamic terminal fields. J Comp Neurol. 1988;271(4):473–92.
Noda H, Sugita S, Ikeda Y. Afferent and efferent connections of the oculomotor region of the fastigial nucleus in the macaque monkey. J Comp Neurol. 1990;302(2):330–48.
Büttner-Ennever JA, Horn AK, Henn V, Cohen B. Projections from the superior colliculus motor map to omnipause neurons in monkey. J Comp Neurol. 1999;413(1):55–67.
Gandhi NJ, Keller EL. Spatial distribution and discharge characteristics of superior colliculus neurons antidromically activated from the omnipause region in monkey. J Neurophysiol. 1997;78(4):2221–5.
Gandhi NJ, Keller EL. Activity of the brain stem omnipause neurons during saccades perturbed by stimulation of the primate superior colliculus. J Neurophysiol. 1999;82(6):3254–67.
Ohgaki T, Markham CH, Schneider JS, Curthoys IS. Anatomical evidence of the projection of pontine omnipause neurons to midbrain regions controlling vertical eye movements. J Comp Neurol. 1989;289(4):610–25.
Keller EL, Edelman JA. Use of interrupted saccade paradigm to study spatial and temporal dynamics of saccadic burst cells in superior colliculus in monkey. J Neurophysiol. 1994;72(6):2754–70.
Keller EL, Gandhi NJ, Shieh JM. Endpoint accuracy in saccades interrupted by stimulation in the omnipause region in monkey. Vis Neurosci. 1996;13(6):1059–67.
Munoz DP, Waitzman DM, Wurtz RH. Activity of neurons in monkey superior colliculus during interrupted saccades. J Neurophysiol. 1996;75(6):2562–80.
Schiller PH, True SD, Conway JL. Deficits in eye movements following frontal eye-field and superior colliculus ablations. J Neurophysiol. 1980;44(6):1175–89.
Schiller PH, Chou IH. The effects of frontal eye field and dorsomedial frontal cortex lesions on visually guided eye movements. Nat Neurosci. 1998;1(3):248–53.
Dias EC, Segraves MA. Muscimol-induced inactivation of monkey frontal eye field: effects on visually and memory-guided saccades. J Neurophysiol. 1999;81(5):2191–214.
Hanes DP, Smith MK, Optican LM, Wurtz RH. Recovery of saccadic dysmetria following localized lesions in monkey superior colliculus. Exp Brain Res. 2005;160(3):312–25.
Ohtsuka K, Noda H. Discharge properties of Purkinje cells in the oculomotor vermis during visually guided saccades in the macaque monkey. J Neurophysiol. 1995;74(5):1828–40.
Optican LM, Robinson DA. Cerebellar-dependent adaptive control of primate saccadic system. J Neurophysiol. 1980;44(6):1058–76.
Zee DS, Yamazaki A, Butler PH, Gücer G. Effects of ablation of flocculus and paraflocculus of eye movements in primate. J Neurophysiol. 1981;46(4):878–99.
Ohtsuka K, Noda H. Saccadic burst neurons in the oculomotor region of the fastigial nucleus of macaque monkeys. J Neurophysiol. 1991;65(6):1422–34.
Fuchs AF, Robinson FR, Straube A. Role of the caudal fastigial nucleus in saccade generation. I. Neuronal discharge pattern. J Neurophysiol. 1993;70(5):1723–40.
Helmchen C, Straube A, Büttner U. Saccade-related activity in the fastigial oculomotor region of the macaque monkey during spontaneous eye movements in light and darkness. Exp Brain Res. 1994;98(3):474–82.
Selhorst JB, Stark L, Ochs AL, Hoyt WF. Disorders in cerebellar ocular motor control. I. Saccadic overshoot dysmetria. An oculographic, control system and clinico-anatomical analysis. Brain J Neurol. 1976;99(3):497–508.
Selhorst JB, Stark L, Ochs AL, Hoyt WF. Disorders in cerebellar ocular motor control. II. Macrosaccadic oscillation. An oculographic, control system and clinico-anatomical analysis. Brain J Neurol. 1976;99(3):509–22.
Robinson FR, Straube A, Fuchs AF. Role of the caudal fastigial nucleus in saccade generation. II. Effects of muscimol inactivation. J Neurophysiol. 1993;70(5):1741–58.
Livingstone M, Hubel D. Segregation of form, color, movement, and depth: anatomy, physiology, and perception. Science. 1988;240(4853):740–9.
Glickstein M, Gerrits N, Kralj-Hans I, Mercier B, Stein J, Voogd J. Visual pontocerebellar projections in the macaque. J Comp Neurol. 1994;349(1):51–72.
Van Essen DC, Gallant JL. Neural mechanisms of form and motion processing in the primate visual system. Neuron. 1994;13(1):1–10.
Ilg UJ. Commentary: smooth pursuit eye movements: from low-level to high-level vision. Prog Brain Res. 2002;140:279–98.
Werner JS, Chalupa LM, editors. The new visual neurosciences. 1st ed. Cambridge: The MIT Press; 2013. 1696 p
Derrington AM, Allen HA, Delicato LS. Visual mechanisms of motion analysis and motion perception. Annu Rev Psychol. 2004;55(1):181–205.
Vaina LM, Soloviev S. First-order and second-order motion: neurological evidence for neuroanatomically distinct systems. Prog Brain Res. 2004;144:197–212.
Chen KJ, Sheliga BM, Fitzgibbon EJ, Miles FA. Initial ocular following in humans depends critically on the fourier components of the motion stimulus. Ann N Y Acad Sci. 2005;1039:260–71.
Nagao S, Kitamura T, Nakamura N, Hiramatsu T, Yamada J. Differences of the primate flocculus and ventral paraflocculus in the mossy and climbing fiber input organization. J Comp Neurol. 1997;382(4):480–98.
Rambold H, Churchland A, Selig Y, Jasmin L, Lisberger SG. Partial ablations of the Flocculus and ventral Paraflocculus in monkeys cause linked deficits in smooth pursuit eye movements and adaptive modification of the VOR. J Neurophysiol. 2002;87(2):912–24.
Heinen SJ, Keller EL. The function of the cerebellar uvula in monkey during optokinetic and pursuit eye movements: single-unit responses and lesion effects. Exp Brain Res. 1996;110(1):1–14.
Kase M, Noda H, Suzuki DA, Miller DC. Target velocity signals of visual tracking in vermal Purkinje cells of the monkey. Science. 1979;205(4407):717–20.
Suzuki DA, Noda H, Kase M. Visual and pursuit eye movement-related activity in posterior vermis of monkey cerebellum. J Neurophysiol. 1981;46(5):1120–39.
Suzuki DA, Keller EL. The role of the posterior vermis of monkey cerebellum in smooth-pursuit eye movement control. II. Target velocity-related Purkinje cell activity. J Neurophysiol. 1988;59(1):19–40.
Ohtsuka K, Enoki T. Transcranial magnetic stimulation over the posterior cerebellum during smooth pursuit eye movements in man. Brain J Neurol. 1998;121(Pt 3):429–35.
Shinmei Y, Yamanobe T, Fukushima J, Fukushima K. Purkinje cells of the cerebellar dorsal vermis: simple-spike activity during pursuit and passive whole-body rotation. J Neurophysiol. 2002;87(4):1836–49.
Fuchs AF, Robinson FR, Straube A. Participation of the caudal fastigial nucleus in smooth-pursuit eye movements. I. Neuronal activity. J Neurophysiol. 1994;72(6):2714–28.
Vahedi K, Rivaud S, Amarenco P, Pierrot-Deseilligny C. Horizontal eye movement disorders after posterior vermis infarctions. J Neurol Neurosurg Psychiatry. 1995;58(1):91–4.
Laurens J, Angelaki DE. The functional significance of velocity storage and its dependence on gravity. Exp Brain Res. 2011;210(3–4):407–22.
Zee DS, Leigh RJ, Mathieu-Millaire F. Cerebellar control of ocular gaze stability. Ann Neurol. 1980;7(1):37–40.
Leigh RJ, Robinson DA, Zee DS. A hypothetical explanation for periodic alternating nystagmus: instability in the optokinetic-vestibular system. Ann N Y Acad Sci. 1981;374:619–35.
Stell R, Bronstein AM, Plant GT, Harding AE. Ataxia telangiectasia: a reappraisal of the ocular motor features and their value in the diagnosis of atypical cases. Mov Disord Off J Mov Disord Soc. 1989;4(4):320–9.
Shaikh AG, Marti S, Tarnutzer AA, Palla A, Crawford TO, Straumann D, et al. Ataxia telangiectasia: a “disease model” to understand the cerebellar control of vestibular reflexes. J Neurophysiol. 2011;105(6):3034–41.
Waespe W, Cohen B, Raphan T. Dynamic modification of the vestibulo-ocular reflex by the nodulus and uvula. Science. 1985;228(4696):199–202.
Ito M. Neurophysiological aspects of the cerebellar motor control system. Int J Neurol. 1970;7(2):162–76.
Schultheis LW, Robinson DA. Directional plasticity of the vestibuloocular reflex in the cat. Ann N Y Acad Sci. 1981;374:504–12.
Walker MF, Zee DS. Cerebellar disease alters the axis of the high-acceleration vestibuloocular reflex. J Neurophysiol. 2005;94(5):3417–29.
Walker MF, Zee DS. Directional abnormalities of vestibular and optokinetic responses in cerebellar disease. Ann N Y Acad Sci. 1999;871:205–20.
Lisberger SG. The latency of pathways containing the site of motor learning in the monkey vestibulo-ocular reflex. Science. 1984;225(4657):74–6.
Fernandez C, Goldberg JM, Abend WK. Response to static tilts of peripheral neurons innervating otolith organs of the squirrel monkey. J Neurophysiol. 1972;35(6):978–87.
Angelaki DE, Shaikh AG, Green AM, Dickman JD. Neurons compute internal models of the physical laws of motion. Nature. 2004;430(6999):560–4.
Shaikh AG, Ghasia FF, Dickman JD, Angelaki DE. Properties of cerebellar fastigial neurons during translation, rotation, and eye movements. J Neurophysiol. 2005;93(2):853–63.
Shaikh AG, Green AM, Ghasia FF, Newlands SD, Dickman JD, Angelaki DE. Sensory convergence solves a motion ambiguity problem. Curr Biol CB. 2005;15(18):1657–62.
Yakusheva TA, Shaikh AG, Green AM, Blazquez PM, Dickman JD, Angelaki DE. Purkinje cells in posterior cerebellar vermis encode motion in an inertial reference frame. Neuron. 2007;54(6):973–85.
Angelaki DE, Hess BJ. Inertial representation of angular motion in the vestibular system of rhesus monkeys. II. Otolith-controlled transformation that depends on an intact cerebellar nodulus. J Neurophysiol. 1995;73(5):1729–51.
Sheliga BM, Yakushin SB, Silvers A, Raphan T, Cohen B. Control of spatial orientation of the angular vestibulo-ocular reflex by the nodulus and uvula of the vestibulocerebellum. Ann N Y Acad Sci. 1999;871(1):94–122.
Robinson DA. Adaptive gain control of vestibuloocular reflex by the cerebellum. J Neurophysiol. 1976;39(5):954–69.
Wearne S, Raphan T, Cohen B. Effects of tilt of the gravito-inertial acceleration vector on the angular vestibuloocular reflex during centrifugation. J Neurophysiol. 1999;81(5):2175–90.
Robinson DA. The use of matrices in analyzing the three-dimensional behavior of the vestibulo-ocular reflex. Biol Cybern. 1982;46(1):53–66.
Raphan T, Matsuo V, Cohen B. Velocity storage in the vestibulo-ocular reflex arc (VOR). Exp Brain Res. 1979;35(2):229–48.
Ramat S, Leigh RJ, Zee DS, Optican LM. What clinical disorders tell us about the neural control of saccadic eye movements. Brain J Neurol. 2007;130(Pt 1):10–35.
Funding
This work was supported by the Dystonia Coalition Career Development Award (AS), Dystonia Medical Research Foundation Research Grant (AS), and the American Academy of Neurology Career Development Award (AS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tang, S.Y., Shaikh, A.G. Past and Present of Eye Movement Abnormalities in Ataxia-Telangiectasia. Cerebellum 18, 556–564 (2019). https://doi.org/10.1007/s12311-018-0990-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-018-0990-x