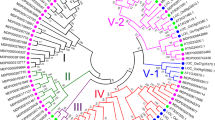
Abstract
The conserved BURP-containing proteins are specific to plants and play a crucial role in plant growth, development, and response to abiotic stresses. However, less is known about the systematic characterization of BURP-containing proteins in apple. This study aimed to identify and analyze all BURP-containing genes in the apple genome, as well as to examine their expression patterns through various bioinformatics methods. Eighteen members of BURP-containing genes were identified in apple, six members lacked signal peptides, and the secondary structure was mainly a Random coil of BURP-containing genes. Gene structure and Motif analysis showed that proteins have similar structures and are conserved at the C-terminal. Cis-acting element analysis revealed that the proteins contain phytohormone and stress response elements, and chromosomal localization revealed that the family is unevenly distributed across eight chromosomes, with duplication of fragments leading to the expansion of family proteins. Tissue expression showed that MdPG3 and MdPG4 were expressed in different tissues and different varieties, MdRD2 and MdRD7 were highly expressed in ‘M74’ fruits and MdRD7 in ‘M49’ leaves, while MdUSP1 was highly expressed in ‘GD’ roots. The quantitative real-time PCR analysis showed that the expressions of six and seven genes were significantly up-regulated under NaCl and PEG treatments, respectively, whereas MdRD7 was significantly up-regulated under NaCl and PEG treatment over time. This study offers a comprehensive identification and expression analysis of BURP-containing proteins in apple. The findings provide a theoretical foundation for further exploration of the functions of this protein family.










Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available in the Phytozome 13 repository (https://phytozome-next.jgi.doe.gov/) and the apple repository (https://iris.angers.inra.fr/gddh13/).
References
Bassüner R, Bäumlein H, Huth A, Jung R, Wobus U, Rapoport TA, Saalbach G, Müntz K (1998) Abundant embyonic mRNA in field bean (Vicia faba L.) codes for a new class of seed proteins: cDNA cloning and characterization of primary translation product. Plant Mol Biol 11:321–334. https://doi.org/10.1007/BF00027389
Batchelor AK, Boutilier K, Miller SS, Hattori J, Bowman LA, Hu M, Lantin S, Johnson DA, Miki BLA (2002) SCB1, a BURP-domain protein gene, from developing soybean seed coats. Planta 215(4):523–532. https://doi.org/10.1007/s00425-002-0798-1
Boutilier KA, Ginés MJ, Huang DJM, B, Baszczynski CL, et al (1994) Expression of the BnmNAP subfamily of napin genes coincides with the induction of Brassica microspore embryogenesis. Plant Mol Biol 26:1711–1723. https://doi.org/10.1007/BF00019486
Cannon SB, Mitra A, Baumgarten A, Young ND, May G (2004) The roles of segmental and tandem gene duplication in the evolution of large gene families in Arabidopsis thaliana. BMC Plant Biol 4:10. https://doi.org/10.1186/1471-2229-4-10
Capella-Gutierrez S, Silla-Martinez JM, Gabaldon T (2009) trimAl: a tool for automated alignment trimming in large-scale phylogenetic analyses. Bioinformatics 25:1972–1973. https://doi.org/10.1093/bioinformatics/btp348
Chenna R, Sugawara H, Koike T, Lopez R, Gibson TJ, Higgins DG, Thompson JD (2003) Multiple sequence alignment with the Clustal series of programs. Nucleic Acids Res 31:3497–3500. https://doi.org/10.1093/nar/gkg500
Daccord N, Celton JM, Linsmith G, Becker C, Choisne N (2017) High-quality de novo assembly of the apple genome and methylome dynamics of early fruit development. Nat Genet 49(7):1099–1106. https://doi.org/10.1038/ng.3886
Ding XP, Hou X, Xie KB, Xiong LZ (2009) Genome-wide identification of BURP domain-containing genes in rice reveals a gene family with diverse structures and responses to abiotic stresses. Planta 230:149–163. https://doi.org/10.1007/s00425-009-0929-z
Du H, Yang SS, Liang Z, Feng BR, Liu L, Huang YB, Tang YX (2012) Genome-wide analysis of the MYB transcription factor superfamily in soybean. BMC Plant Biol 12:106. https://doi.org/10.1186/1471-2229-12-106
Fernandez L, Torregrosa L, Terrier N, Sreekantan L, Grimplet J, Davies C, Thomas MR, Romieu C, Ageorges A (2007) Identification of genes associated with flesh morphogenesis during grapevine fruit development. Plant Mol Biol 63(3):307–323. https://doi.org/10.1007/s11103-006-9090-2
Finn RD, Mistry J, Schuster-Böckler B, Griffiths-Jones S, Hollich V (2006) Pfam: clans, web tools and services. Nucleic Acids Res 34:D247–D251. https://doi.org/10.1093/nar/gkj149
Freeling M (2009) Bias in plant gene content following different sorts of duplication: tandem, whole-genome, segmental, or by transposition. Annu Rev Plant Biol 60:433–453. https://doi.org/10.1146/annurev.arplant.043008.092122
Fu LF, Zhang ZJ, Wang H, Zhao XJ, Su L, Geng LF, Lu YZ, Tong BQ, Liu QH, Jiang XQ (2022) Genome-wide analysis of BURP genes and identification of a BURP-V gene RcBURP4 in Rosa chinensis. Plant Cell Rep 41(2):395–413. https://doi.org/10.1007/s00299-021-02815-0
Gan DF, Jiang HY, Zhang J, Zhao Y, Zhu SW, Ceng BJ (2011) Genome-wide analysis of BURP domain-containing genes in maize and sorghum. Mol Biol Rep 38(7):4553–4563. https://doi.org/10.1007/s11033-010-0587-z
Geng MT, Yao Y, Wang YL, Wu XH, Sun C et al (2017) Structure, expression, and functional analysis of the hexokinase gene family in cassava. Int J Mol Sci 18(5):1041–1059. https://doi.org/10.3390/ijms18051041
Gou H, Nai G, Lu S, Ma W, Chen B, Mao J (2022) Genome-wide identification and expression analysis of PIN gene family under phytohormone and abiotic stresses in Vitis Vinifera L. Physiol Mol Biol Plants 28(10):1905–1919. https://doi.org/10.1007/s12298-022-01239-8
Granger C, Coryell V, Khanna A, Keim P, Vodkin L, Shoemaker RC (2002) Identification, structure, and differential expression of members of a BURP domain containing protein family in soybean. Genome 45:693–701. https://doi.org/10.1139/g02-032
Harshavardhan VT, Van Son L, Seiler C, Junker A, Weigelt-Fischer K, Klukas C, Altmann T, Sreenivasulu N, Bäumlein H, Kuhlmann M (2014) AtRD22 and AtUSPL1, members of the plant-specifc BURP domain family involved in Arabidopsis thaliana drought tolerance. PLoS ONE 9(10):e110065. https://doi.org/10.1371/journal.pone.0110065
Hattori J, Boutilier KA, van Lookeren Campagne MM, Miki BL (1998) A conserved BURP domain defines a novel group of plant proteins with unusual primary structures. Mol Gen Genet 259:424–428. https://doi.org/10.1007/s004380050832
Jamoussi RJ, Elabbassi MM, Ben Jouira H, Hanana M, Zoghlami N, Ghorbel A, Mliki A (2014) Physiological responses of transgenic tobacco plants expressing the dehydration-responsive RD22 gene of Vitis vinifera to salt stress. Turk J Bot 38(2):268–280. https://doi.org/10.3906/bot-1301-53
Kavas M, Yıldırım K, Seçgin Z, Abdulla MF, Gökdemir G (2021) Genome-wide identification of the BURP domain-containing genes in Phaseolus vulgaris. Physiol Mol Biol Plants 27(9):1885–1902. https://doi.org/10.1007/s12298-021-01052-9
Lee JJ, Woodward AW, Chen ZJ (2007) Gene expression changes and early events in cotton fibre development. Ann Bot 100:1391–1401. https://doi.org/10.1093/aob/mcm232
Li Y, Chen X, Chen Z, Cai RH, Zhang HM, Xiang Y (2016) Identification and expression analysis of BURP domain-containing genes in Medicago truncatula. Front Plant Sci 7:485. https://doi.org/10.3389/fpls.2016.00485
Li K, Zhai L, Fu S, Wu T, Zhang X, Xu X, Han Z, Wang Y (2023) Genome-wide analysis of the MdZR gene family revealed MdZR2.2-induced salt and drought stress tolerance in apple rootstock. Plant Sci 334:111755. https://doi.org/10.1016/j.plantsci.2023.111755
Liu HH, Ma Y, Chen N, Guo SY, Liu HL, Guo XY, Chong K, Xu YY (2014) Overexpression of stress-inducible OsBURP16, the β subunit of polygalacturonase 1, decreases pectin content and cell adhesion and increases abiotic stress sensitivity in rice. Plant Cell Environ 37(5):1144–1158. https://doi.org/10.1111/pce.12223
Liu XJ, Gong Q, Zhao CN, Wang DL, Ye XM, Zheng GX, Wang Y, Cao JP, Sun CD (2023) Genome-wide analysis of cytochrome P450 genes in Citrus clementina and characterization of a CYP gene encoding flavonoid 3’-hydroxylase. Hortic Res 10(2):uhac283. https://doi.org/10.1093/hr/uhac283
Lu YY, Fricke W (2023) Salt stress-regulation of root water uptake in a whole-plant and diurnal context. Int J Mol Sci 24:8070. https://doi.org/10.3390/ijms24098070
Ma BQ, Liao L, Fang T, Qian P, Collins O, Hui Z, Ma FW, Han YP (2019) A Ma10 gene encoding P-type ATPase is involved in fruit organic acid accumulation in apple. Plant Biotechnol J 17(3):674–686. https://doi.org/10.1111/pbi.13007
Matus JT, Aquea F, Espinoza C, Vega A, Cavallini E, Dal Santo S, Cañón P, de la Guardia AR-H, Serrano J, Tornielli GB, ArceJohnson P (2014) Inspection of the grapevine BURP superfamily highlights an expansion of RD22 genes with distinctive expression features in berry development and ABA-mediated stress responses. PLoS ONE 9:e110372. https://doi.org/10.1371/journal.pone.0110372
Nguyen LT, Schmidt HA, von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32(1):268–274. https://doi.org/10.1093/molbev/msu300
Pickett FB, Meeks-Wagner DR (1995) Seeing double: appreciating genetic redundancy. Plant Cell 7:1347–1356. https://doi.org/10.1105/tpc.7.9.1347
Ren JX, Li YM, Ma WF, Wu Z, Mao J (2023a) Identification of apple 14–3-3 gene family and functional analysis of MdGRF13 gene in apple. J Fruit Sci 40:405–421. https://doi.org/10.13925/j.cnki.gsxb.20220368
Ren JX, Zhang P, Dai YB, Liu XH, Lu SX, Guo LL, Gou HM, Mao J (2023b) Evolution of the 14–3-3 gene family in monocotyledons and dicotyledons and validation of MdGRF13 function in transgenic Arabidopsis thaliana. Plant Cell Rep 42:1345–1364. https://doi.org/10.1007/s00299-023-03035-4
Rosero A, Granda L, Berdugo-Cely JA, Šamajová O, Šamaj J, Cerkal R (2020) A dual strategy of breeding for drought tolerance and introducing drought-tolerant, underutilized crops into production systems to enhance their resilience to water defciency. Plants 9:1263. https://doi.org/10.3390/plants9101263
Shao YH, Wei G, Wang L, Dong Q, Zhao Y, Chen BJ (2011) Genome-wide analysis of BURP domain-containing genes in Populus trichocarpa. J Integr Plant Biol 53(9):743–755. https://doi.org/10.1111/j.1744-7909.2011.01068.x
Sun HR, Wei HL, Wang HT, Hao PB, Gu LJ, Liu GY, Ma L, Su ZZ, Yu SX (2019) Genome-wide identification and expression analysis of the BURP domain-containing genes in Gossypium hirsutum. BMC Genomics 20(1):558. https://doi.org/10.1186/s12864-019-5948-y
Tang YL, Cao Y, Qiu JB, Gao Z, Ou ZH, Wang YJ, Zheng YZ (2014) Expression of a vacuole-localized BURP-domain protein from soybean (SALI3-2) enhances tolerance to cadmium and copper stresses. PLoS ONE 9(6):e98830. https://doi.org/10.1371/journal.pone.0098830
Teerawanichpan P, Xia Q, Caldwell SJ, Datla R, Selvaraj G (2009) Protein storage vacuoles of Brassica napus zygotic embryos accumulate a BURP domain protein and perturbation of its production distorts the PSV. Plant Mol Biol 71:331–343. https://doi.org/10.1007/s11103-009-9541-7
Van Son L, Tiedemann J, Rutten T, Hillmer S, Hinz G, Zank T, Manteuffel R, Bäumlein S (2009) The BURP domain protein AtUSPL1 of Arabidopsis thaliana is destined to the protein storage vacuoles and overexpression of the cognate gene distorts seed development. Plant Mol Biol 71:319–329. https://doi.org/10.1007/s11103-009-9526-6
Vera Hernández PF, Mendoza Onofre LE, Rosas Cárdenas FF (2023) Responses of sorghum to cold stress: a review focused on molecular breeding. Front Plant Sci 14:1124335. https://doi.org/10.3389/fpls.2023.1124335
Wang AM, Xia Q, Xie WS, Datla R, Selvaraj G (2003) The classical Ubisch bodies carry a sporophytically produced structural protein (RAFTIN) that is essential for pollen development. PNAS 100(24):14487–14492. https://doi.org/10.1073/pnas.2231254100
Wang K, Li M, Hakonarson H (2010) ANNOVAR: Functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res 38:e164. https://doi.org/10.1093/nar/gkq603
Wang HM, Zhou L, Fu YP, Cheung MY, Wong FL, Phang TH, Sun ZX, Lam HM (2012) Expression of an apoplast-localized BURP-domain protein from soybean (GmRD22) enhances tolerance towards abiotic stress. Plant Cell Environ 35(11):1932–1947. https://doi.org/10.1111/j.1365-3040.2012.02526.x
Wang WZ, Zhang Z, Li XG (2022) Identification and expression analysis of BURP domain-containing genes in jujube and their involvement in low temperature and drought response. BMC Genomics 23:692–707. https://doi.org/10.1186/s12864-022-08907-9
Watson CF, Zheng LS, DellaPenna D (1994) Reduction of tomato polygalacturonase β subunit expression affects pectin solubilization and degradation during fruit ripening. Plant Cell 6(11):1623–1634. https://doi.org/10.1105/tpc.6.11.1623
Xu HL, Li YX, Yan YM, Wang K, Gao Y, Hu YK (2010) Genome-scale identification of soybean BURP domain-containing genes and their expression under stress treatments. BMC Plant Biol 10:197. https://doi.org/10.1186/1471-2229-10-197
Yamaguchi-Shinozaki K, Shinozaki K (1994) A novel cis-acting element in an Arabidopsis gene is involved in responsiveness to drought, low-temperature, or high-salt stress. Plant Cell 6:251–264. https://doi.org/10.1105/tpc.6.2.251
Yang YM, Ding YD, Chen LJ, Tian XT, Yin WJ, Peng HH, Du W, Liang LP, Ren XL (2021) Identification and expression analysis of the vacuolar iron transporter gene family in apple. Acta Hortic Sin 48(2):205–218. https://doi.org/10.16420/j.issn.0513-353x.2020-0272
Yu SW, Yang FW, Zou YQ, Yang YN, Li TF, Chen SJ, Wang YL, Xu K, Xia H, Luo LJ (2022) Overexpressing PpBURP2 in rice increases plant defense to abiotic stress and bacterial leaf blight. Front Plant Sci 13:812279. https://doi.org/10.3389/fpls.2022.812279
Zheng LS, Heupel RC, DellaPenna D (1992) The β subunit of tomato fruit polygalacturonase isoenzyme 1: isolation, characterization, and identification of unique structural features. Plant Cell 4:1147–1156. https://doi.org/10.1105/tpc.4.9.1147
Acknowledgements
We express our gratitude to Shanghai Tengyun Biotechnology Co., Ltd. for their development of the Hiplot Pro platform (https://hiplot.com.cn/) and their provision of technical assistance and valuable data analysis and visualization tools.
Funding
This work was supported by the National Key Research and Development Program of China (2022YFD1602106) and Science and Technology Major Project of Gansu Province (22ZD6NA045). The funding bodies had no roles in the experimental design or collection, analysis and interpretation of data in writing the manuscript.
Author information
Authors and Affiliations
Contributions
JM conceived and designed the research. JR performed the experiments, and drafted the manuscript. LF, LG, HG and SL analyzed data and helped in the preparation of the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no confict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ren, J., Feng, L., Guo, L. et al. Genome-wide identification and expression analysis of the BURP domain-containing genes in Malus domestica. Physiol Mol Biol Plants 29, 1717–1731 (2023). https://doi.org/10.1007/s12298-023-01393-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-023-01393-7