Abstract
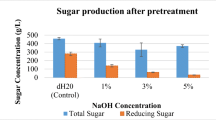
Agricultural and industrial wastes are available in massive amounts, and the disposal of those biowastes causes environmental and health problems. Xylanase plays an essential role in nature and has various industrial applications. The objective of the present work was to evaluate the utilization of some agro-industrial biowastes, including sugarcane bagasse, corn cob, corn stalk, rice straw, wheat straw, wheat bran, and rice bran in the cost-effective production of xylanase enzyme using Aspergillus niger strain AUMC 14230 by applying corn cob (1%) as the main component in fermentation media. Among the agriculture residues used, the results showed that the corn cob was the preferable optimized carbon source for xylanase production with activity (100 Uml−1), followed by wheat bran (84 Uml−1), while corn steep liquor (3% concentration) was the ideal nitrogen source. Furthermore, the findings revealed that the optimum levels of various parameters for the xylanase production were inoculum size 0.50% (v/mL), initial pH 4.5, incubation temperature 50 °C, aeration 1:5 (Vm: Vf), agitation speed 175 rpm, and time course 120 h. Based on the obtained findings, it could be concluded that the corn cob and wheat bran, inexpensive and abundant sources, could be applied as a carbon source for large-scale industrial xylanase production, which ultimately lowers the production cost of xylanase enzyme, besides, helps to reduce the environmental problems linked with the disposal of agro-industrial biowastes.







Similar content being viewed by others
Data availability
Not applicable.
References
Ahmed SA, Saleh SAA, Mostafa FA, Abd El Aty AA, Ammar HAM (2016) Characterization and valuable applications of xylanase from endophytic fungus Aspergillus terreus KP900973 isolated from Corchorus olitorius. Biocatal Agric Biotechnol 7:134–144
Alani F, Anderson WA, Moo-Young M (2008) New isolate of Streptomyces sp. with novel thermo alkalo tolerant cellulases. Biotechn Lett 30:123–126
Ashour A, Amer M, Marzouk A, Shimizu K, Kondo R, El-Sharkawy S (2013) Corncobs as a potential source of functional chemicals. Molecules 18(11):13823–13830
Atalla SMM, Ahmed NE, Awad HM, El Gamal NG, El Shamy AR (2020) Statistical optimization of xylanase production, using different agricultural wastes by Aspergillus oryzae MN894021, as a biological control of faba bean root diseases. Egypt J Biol Pest Control 30:125
Bajar S, Singh A, Bishnoi NR (2020) Exploration of low-cost agro-industrial waste substrate for cellulase and xylanase production using Aspergillus heteromorphus. Appl Water Sci 10:153. https://doi.org/10.1007/s13201-020-01236-w
Bedade D, Berezina O, Singhal R, Deska J, Shamekh S (2017) Extracellular xylanase production from a new xylanase producer Tuber maculatum mycelium under submerged fermentation and its characterization. Biocatal Agric Biotechnol 11:288–293
Bhardwaj N, Kumar B, Verma P (2019) A detailed overview of xylanases: an emerging biomolecule for current and future prospective. Bioresour Bioprocess. https://doi.org/10.1186/s40643-019-0276-2
Chopra S, Kumar D (2023) Characterization and biodegradation of paracetamol by biomass of Bacillus licheniformis strain PPY-2 isolated from wastewater. Rend Fis Acc Lincei. https://doi.org/10.1007/s12210-023-01140-w
da Silva-López RE, de Araujo TAA, Monteiro HJJ et al (2022) Study of protease activity from Aspergillus awamori INCQS2B.361U2/1 extracellular fraction and modification of culture medium composition to isolate a novel aspartic protease. Braz J Microbiol 53:1599–1611. https://doi.org/10.1007/s42770-022-00750-0
Das A, Ray L (2016) Production of crude xylanase using an isolated fungal strain Aspergillus sp. S6 by solid state fermentation. Mater Today Proc 3(10):3343–3360
Deguchi S, Tsudome M, Shen Y, Kanish S, Tsujii K, Horikoshi K (2007) Preparation and characterization of nanofibrous cellulose plate as a new solid support for microbial culture. Soft Matt 3:1170–1175
Devi S, Dwivedi D, Bhatt AK (2022) Utilization of Agroresidues for the production of xylanase by Bacillus safensis XPS7 and optimization of production parameters. Fermentation 8:221. https://doi.org/10.3390/fermentation8050221
Dhulappa L, Siddalingeshwara KG (2015) Effect of nitrogen source for the biosynthesis of xylanase from Aspergillus tamarii. J Drug Deliv 5:18–22
Elimam DM, Ramadan MF, Elshazly AM, Farag MA (2022) Introduction to Mediterranean Fruits Bio-wastes Chemistry Functionality and TechnoApplications. In: Ramadan MF, Farag MA (eds) Mediterranean Fruits Bio-wastes. Springer, Cham. https://doi.org/10.1007/978-3-030-84436-3_1
Gawande PV, Kamat MY (1999) Production of Aspergillus xylanase by lignocellulosic waste fermentation and its application. J Appl Microbiol 87:511–519
Ghoshal G, Banerjee UC, Shivhare US (2015) Utilization of agrowaste and xylanase production in solid state fermentation. J Biochem Technol 6:1013–1024
Gowdhaman D, Ponnusami V (2019) Xylanases: a biotechnological potential enzyme. Phytopharmaceuticals and Drug Delivery Approaches. Avid Science, pp 02–15
Haltrich, D.; Prenner, E.; Preil, M. and Steiner, W. (1996). Production of extremely high values of xylanase activity by Schizophyllum commune. In Biotechnology in the Pulp and Paper Industry. Proc. 5th Intern. Conf. Biotechnol. Pulp Paper Ind., ed. M. Kuwahara & M. Shimada. Uni Publishers, Tokyo, pp. 123–128.
Ho HH (2015) Xylanase production by Bacillus subtilis using carbon source of inexpensive agricultural wastes in two different approaches of submerged fermentation (smf) and solid state fermentation. J Food Process Technol 4:4–7
Irfan M, Nadeem M, Q Syed, J., (2014) Radiation Res. and Appl. Sci. 7 317–326.
Irfan M, Muhammad U, Nadeem R, Syed Q (2016) Optimization of process parameters for xylanase production by Bacillus sp. in submerged fermentation. J Radiat Res Appl Sci 9:139–147
Kadowaki MK, Souza CGM, Simão RCG, PeraltaRM, (1997) Xylanase production by Aspergillus tamarii. Appl Biochem Biotechnol - Part A Enzyme Eng Biotechnol 66(2):97–106
Katapodis P, Christakopoulou V, Kekos D, Christakopoulos P (2007) Optimization of xylanase production by Chaetomium thermophilum in wheat straw using response surface methodology. Biochem Eng J 35(2):136–141
Khasin A, Alchanati IRIS, Al Shoham YUV (1993) Purification and characterization of a thermostable xylanase from Bacillus stearo thermophilus T-6. Appl Environ Microbiol. https://doi.org/10.1128/aem.59.6.1725-1730.1993
Mehnati-Najafabadi V, Taheri-Kafrani A, Bordbar AK (2018) Xylanase immobilization on modified superparamagnetic graphene oxide nanocomposite Effect of PEGylation on activity and stability. Int J Biol Macromol 107(PartA):418–425
Mehta N, Yadava S, Tilak P, Jayaramu M, Siddalingeshwara KG (2014) Role of nitrogen source for the production of xylanase from Aspergillus sp. J Drug Deliv 4:147–149
Monclaro AV, Fontes PR, Recalde GL et al (2022) Evaluation of endoglucanase and xylanase production by Aspergillus tamarii cultivated in agro-industrial lignocellulosic biomasses. Folia Microbiol 67:721–732. https://doi.org/10.1007/s12223-022-00971-8
Nitin KS, Vivek KT, Santosh KM (2017). The production of xylanase enzyme (E.C. Number=3.2.1.8) using solid substrate fermentation. Biotechnol An Ind J, 134–145.
Pirota RDPB, Tonelotto M, da Delabona PS, Fonseca RF, Paixão DAA, Baleeiro FCF, Bertucci Neto V, Farinas CS (2013) Enhancing xylanases production by a new Amazon Forest strain of Aspergillus oryzae using solid-state fermentation under controlled operation conditions. Ind Crops Products 45:465–471
Ritthibut N, Lim ST, Oh SJ (2022) In vitro cosmeceutical activity of alcoholic extract from chestnut inner shell fermented with Aspergillus sojae. Food Sci Biotechnol 31:443–450. https://doi.org/10.1007/s10068-022-01044-9
Sanghi A, Garg N, Sharma J, Kuhar K, Kuhad RC, Gupta VK (2008) Optimization of xylanase production using inexpensive agro-residues by alkalophilic Bacillus subtilis ASH in solid-state fermentation. World J Microbiol Biotechnol 24:633–640
Seyis I, Aksoz N (2005) Effect of carbon and nitrogen sources on xylanase production by Trichoderma harzianum 1073 D3. Int Biodeterior Biodegradation 55(2):115–119
Somogyi M (1952) Notes on sugar determination. J Biol Chem 195:19–23
Sonia KG, Chadha BS, Saini HS (2005) Sorghum straw for xylanase hyperproduction by Thermomyces lanuginosus (D2W3) under solid-state fermentation. Bioresour Technol 96:1561e1569
Uday USP, Majumdar R, Tiwari ON, Mishra U, Mondal A, Bandyopadhyay TK, Bhunia B (2017) Isolation, screening and characterization of a novel extracellular xylanase from Aspergillus niger (KP874102.1) and its application in orange peel hydrolysis. Int J Biol Macromol 105:401–409
Funding
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that they do not have any conflict of interest.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abdelaliem, Y.F., Mahmoud, M.H., Elkassem, N.A. et al. Utilization of agro-industrial biowastes to produce xylanase using Aspergillus niger AUMC 14230: optimization of production parameters. Rend. Fis. Acc. Lincei 34, 941–951 (2023). https://doi.org/10.1007/s12210-023-01180-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12210-023-01180-2