Abstract
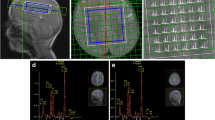
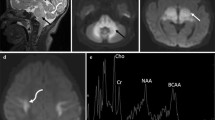
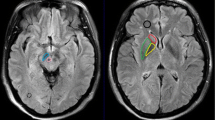
This study aimed to use chemical exchange saturation transfer (CEST) and magnetic resonance spectroscopy (MRS) at 7T-MRI for early detection of intracerebral lactate in a mitochondrial disease model without brain lesions. We considered Ndufs4-knockout (KO) mice as Leigh syndrome models and wild-type (WT) mice as control mice. Brain MRI and 1H-MRS were performed. T2WI data acquired with the Rapid Acquisition with Refocused Echoes (RARE) sequence were used for evaluation of brain lesions. CEST imaging of mice brains was performed using RARE with a magnetization transfer (MT) pulse. The MT ratio (MTR) asymmetry curves and five MTR asymmetry maps at 0.5, 1.0, 2.0, 3.0, and 3.5 ppm were calculated using these CEST images. Metabolite concentrations were measured by MRS. T2WI MRI revealed no obvious abnormal findings in KO and WT mice brains at 6 weeks of age. The MTR asymmetry maps at 0.5 ppm, 1.0 ppm, and 2.0 ppm of the KO mice were higher than those of the control mice. Brain 1H MRS revealed a significant increase in lactate levels in all KO mice in comparison with those in the control mice. Additionally, creatine levels in the KO mice were slightly higher than those in the control mice. The levels of the other four metabolites—mIns, NAA + NAAG, GPC + PCh, and Glu + Gln—did not change significantly. We propose that CEST imaging can be used as a biomarker of intracerebral elevated lactate levels in mitochondrial disease.






Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- CEST:
-
Chemical exchange saturation transfer
- CSI:
-
Chemical shift imaging
- FASTMAP:
-
Fast automated shimming technique by mapping along projections
- KO:
-
Knock out
- mM:
-
Mmol/L
- MTR:
-
Magnetization transfer ratio
- MR:
-
Magnetic resonance
- MRI:
-
Magnetic resonance imaging
- NOE:
-
Nuclear overhauser enhancements
- PRESS:
-
Point resolved spectroscopy
- RARE:
-
Rapid acquisition with refocused echoes
- SD:
-
Standard deviation
- TE:
-
Echo time
- TR:
-
Repetition time
- T2WI:
-
T2-weighted images
- VAPOR:
-
Variable power RF pulses with optimized relaxation delays
- WT:
-
Wild type
- WASSR:
-
Water saturation shift referencing
- 1H MRS:
-
Proton magnetic resonance spectroscopy
References
Gorman GS, Chinnery PF, DiMauro S, Hirano M, Koga Y, McFarland R, Suomalainen A, Thorburn DR, Zeviani M, Turnbull DM. Mitochondrial diseases. Nat Rev Dis Primers. 2016;2:16080.
Vafai SB, Mootha VK. Mitochondrial disorders as windows into an ancient organelle. Nature. 2012;491(7424):374–83.
Schapira AH. Mitochondrial diseases. Lancet. 2012;379(9828):1825–34.
Lake NJ, Bird MJ, Isohanni P, Paetau A. Leigh syndrome: neuropathology and pathogenesis. J Neuropathol Exp Neurol. 2015;74(6):482–92.
Lee JS, Kim H, Lim BC, Hwang H, Choi J, Kim KJ, Hwang YS, Chae JH. Leigh syndrome in childhood: neurologic progression and functional outcome. J Clin Neurol. 2016;12(2):181–7.
Sofou K, De Coo IF, Isohanni P, Ostergaard E, Naess K, De Meirleir L, Tzoulis C, Uusimaa J, De Angst IB, Lonnqvist T, Pihko H, Mankinen K, Bindoff LA, Tulinius M, Darin N. A multicenter study on Leigh syndrome: disease course and predictors of survival. Orphanet J Rare Dis. 2014;9:52.
Lee HF, Tsai CR, Chi CS, Lee HJ, Chen CC. Leigh syndrome: clinical and neuroimaging follow-up. Pediatr Neurol. 2009;40(2):88–93.
Torraco A, Diaz F, Vempati UD, Moraes CT. Mouse models of oxidative phosphorylation defects: powerful tools to study the pathobiology of mitochondrial diseases. Biochim Biophys Acta. 2009;1793(1):171–80.
Lin DD, Crawford TO, Barker PB. Proton MR spectroscopy in the diagnostic evaluation of suspected mitochondrial disease. AJNR Am J Neuroradiol. 2003;24(1):33–41.
Kaufmann P, Shungu DC, Sano MC, Jhung S, Engelstad K, Mitsis E, Mao X, Shanske S, Hirano M, DiMauro S. De Vivo DC. Cerebral lactic acidosis correlates with neurological impairment in MELAS. Neurology. 2004;62(8):1297–302.
Boddaert N, Romano S, Funalot B, Rio M, Sarzi E, Lebre AS, Bahi-Buisson N, Valayannopoulos V, Desguerre I, Seidenwurm D, Brunelle F, Brami-Zylberberg F, Rotig A, Munnich A de. Lonlay P. 1H MRS spectroscopy evidence of cerebellar high lactate in mitochondrial respiratory chain deficiency. Mol Genet Metab. 2008;93(1):85–8.
Cakmakci H, Pekcevik Y, Yis U, Unalp A, Kurul S. Diagnostic value of proton MR spectroscopy and diffusion-weighted MR imaging in childhood inherited neurometabolic brain diseases and review of the literature. Eur J Radiol. 2010;74(3):e161-71.
Ward KM, Aletras AH. Balaban RS. A new class of contrast agents for MRI based on proton chemical exchange dependent saturation transfer (CEST). J Magn Reson. 2000;143(1):79–87.
Kogan F, Hariharan H, Reddy R. Chemical exchange saturation transfer (CEST) imaging: description of technique and potential clinical applications. Curr Radiol Rep. 2013;1(2):102–14.
Cai K, Haris M, Singh A, Kogan F, Greenberg JH, Hariharan H, Detre JA, Reddy R. Magnetic resonance imaging of glutamate. Nat Med. 2012;18(2):302–6.
Saito S, Mori Y, Tanki N, Yoshioka Y, Murase K. Factors affecting the chemical exchange saturation transfer of Creatine as assessed by 11.7 T MRI. Radiol Phys Technol. 2015;8(1):146–52.
Cai K, Singh A, Poptani H, Li W, Yang S, Lu Y, Hariharan H, Zhou XJ. Reddy R. CEST signal at 2 ppm (CEST@2 ppm) from Z-spectral fitting correlates with creatine distribution in brain tumor. NMR Biomed. 2015;28(1):1–8.
DeBrosse C, Nanga RP, Bagga P, Nath K, Haris M, Marincola F, Schnall MD, Hariharan H, Reddy R. Lactate chemical exchange saturation transfer (LATEST) imaging in vivo A biomarker for LDH activity. Sci Rep. 2016;6:19517.
DeBrosse C, Nanga RP, Wilson N, D’Aquilla K, Elliott M, Hariharan H, Yan F, Wade K, Nguyen S, Worsley D, Parris-Skeete C, McCormick E, Xiao R, Cunningham ZZ, Fishbein L, Nathanson KL, Lynch DR, Stallings VA, Yudkoff M, Falk MJ, Reddy R. McCormack SE. Muscle oxidative phosphorylation quantitation using creatine chemical exchange saturation transfer (CrCEST) MRI in mitochondrial disorders. JCI Insight. 2016;1(18):e88207.
Kruse SE, Watt WC, Marcinek DJ, Kapur RP, Schenkman KA, Palmiter RD. Mice with mitochondrial complex I deficiency develop a fatal encephalomyopathy. Cell Metab. 2008;7(4):312–20.
Quintana A, Kruse SE, Kapur RP, Sanz E, Palmiter RD. Complex I deficiency due to loss of Ndufs4 in the brain results in progressive encephalopathy resembling Leigh syndrome. Proc Natl Acad Sci USA. 2010;107(24):10996–1001.
Jain IH, Zazzeron L, Goli R, Alexa K, Schatzman-Bone S, Dhillon H, Goldberger O, Peng J, Shalem O, Sanjana NE, Zhang F, Goessling W, Zapol WM. Mootha VK. Hypoxia as a therapy for mitochondrial disease. Science. 2016;352(6281):54–61.
Ferrari M, Jain IH, Goldberger O, Rezoagli E, Thoonen R, Cheng KH, Sosnovik DE, Scherrer-Crosbie M, Mootha VK. Zapol WM. Hypoxia treatment reverses neurodegenerative disease in a mouse model of Leigh syndrome. Proc Natl Acad Sci USA. 2017;114(21):E4241-E50.
Kim M, Gillen J, Landman BA, Zhou J, van Zijl PC. Water saturation shift referencing (WASSR) for chemical exchange saturation transfer (CEST) experiments. Magn Reson Med. 2009;61(6):1441–50.
Liu G, Gilad AA, Bulte JW, van Zijl PC, McMahon MT. High-throughput screening of chemical exchange saturation transfer MR contrast agents. Contrast Media Mol Imaging. 2010;5(3):162–70.
Provencher SW. Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med. 1993;30(6):672–9.
Haris M, Singh A, Cai K, Kogan F, McGarvey J, Debrosse C, Zsido GA, Witschey WR, Koomalsingh K, Pilla JJ, Chirinos JA, Ferrari VA, Gorman JH, Hariharan H, Gorman RC, Reddy R. A technique for in vivo mapping of myocardial creatine kinase metabolism. Nat Med. 2014;20(2):209–14.
Chi CS, Lee HF, Tsai CR, Chen WS, Tung JN, Hung HC. Lactate peak on brain MRS in children with syndromic mitochondrial diseases. J Chin Med Assoc. 2011;74(7):305–9.
Lindroos MM, Borra RJ, Parkkola R, Virtanen SM, Lepomaki V, Bucci M, Virta JR, Rinne JO, Nuutila P, Majamaa K. Cerebral oxygen and glucose metabolism in patients with mitochondrial m.3243A> G mutation. Brain. 2009;132(Pt 12):3274–84.
Kaufmann P, Engelstad K, Wei Y, Kulikova R, Oskoui M, Sproule DM, Battista V, Koenigsberger DY, Pascual JM, Shanske S, Sano M, Mao X, Hirano M, Shungu DC, Dimauro S, De Vivo DC. Natural history of MELAS associated with mitochondrial DNA m.3243A> G genotype. Neurology. 2011;77(22):1965–71.
Thompson Legault J, Strittmatter L, Tardif J, Sharma R, Tremblay-Vaillancourt V, Aubut C, Boucher G, Clish CB, Cyr D, Daneault C, Waters PJ, Consortium L, Vachon L, Morin C, Laprise C, Rioux JD, Mootha VK. Des Rosiers C. A metabolic signature of mitochondrial dysfunction revealed through a monogenic form of leigh syndrome. Cell Rep. 2015;13(5):981–9.
Steele HE, Horvath R, Lyon JJ. Chinnery PF. Monitoring clinical progression with mitochondrial disease biomarkers. Brain. 2017;140(10):2530–40.
Jin T, Wang P, Zong X, Kim SG. MR imaging of the amide-proton transfer effect and the pH-insensitive nuclear overhauser effect at 9.4 T. Magn Reson Med. 2013;69(3):760–70.
Haris M, Cai K, Singh A, Hariharan H, Reddy R. In vivo mapping of brain myo-inositol. Neuroimage. 2011;54(3):2079–85.
Chen SQ, Wang PJ, Ten GJ, Zhan W, Li MH, Zang FC. Role of myo-inositol by magnetic resonance spectroscopy in early diagnosis of Alzheimer’s disease in APP/PS1 transgenic mice. Dement Geriatr Cogn Disord. 2009;28(6):558–66.
Wallimann T, Dolder M, Schlattner U, Eder M, Hornemann T, O’Gorman E, Ruck A, Brdiczka D. Some new aspects of creatine kinase (CK): compartmentation, structure, function and regulation for cellular and mitochondrial bioenergetics and physiology. Biofactors. 1998;8(3–4):229–34.
Pajares S, Arias A, Garcia-Villoria J, Briones P, Ribes A. Role of creatine as biomarker of mitochondrial diseases. Mol Genet Metab. 2013;108(2):119–24.
Zu Z, Janve VA, Li K, Does MD, Gore JC, Gochberg DF. Multi-angle ratiometric approach to measure chemical exchange in amide proton transfer imaging. Magn Reson Med. 2012;68(3):711–9.
Zhang XY, Wang F, Afzal A, Xu J, Gore JC, Gochberg DF. Zu Z. A new NOE-mediated MT signal at around – 1.6 ppm for detecting ischemic stroke in rat brain. Magn Reson Imaging. 2016;34(8):1100–6.
Zhang XY, Wang F, Jin T, Xu J, Xie J, Gochberg DF, Gore JC. Zu Z. MR imaging of a novel NOE-mediated magnetization transfer with water in rat brain at 9.4 T. Magn Reson Med. 2017;78(2):588–97.
Sun PZ, Cheung JS, Wang E, Benner T, Sorensen AG. Fast multislice pH-weighted chemical exchange saturation transfer (CEST) MRI with Unevenly segmented RF irradiation. Magn Reson Med. 2011;65(2):588–94.
Acknowledgements
We thank Dr. Rikita Araki (Bruker Biospin) for CEST calculation used in the current study.
Funding
This work was supported by Grants-in-Aid for Scientific Research (Kakenhi, No. 16K090170 and 15K21774) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no financial or commercial conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
About this article
Cite this article
Saito, S., Takahashi, Y., Ohki, A. et al. Early detection of elevated lactate levels in a mitochondrial disease model using chemical exchange saturation transfer (CEST) and magnetic resonance spectroscopy (MRS) at 7T-MRI. Radiol Phys Technol 12, 46–54 (2019). https://doi.org/10.1007/s12194-018-0490-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12194-018-0490-1