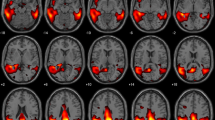
Abstract
Objective
We propose an innovative approach for 18F-FDG PET analysis based on an interval-valued reconstruction of 18F-FDG brain distribution. Its diagnostic performance for Alzheimer’s disease (AD) diagnosis with comparison to a validated post-processing software was assessed.
Methods
Brain 18F-FDG PET data from 26 subjects were acquired in a clinical routine setting. Raw data were reconstructed using an interval-valued version of the ML–EM algorithm called NIBEM that stands for Non-Additive interval-based expectation maximization. Subject classification was obtained via interval-based statistical comparison (intersection ratio, IR) between cortical regions of interest (ROI) including parietal, temporal, and temporo-mesial cortices and a reference region, the sub-cortical grey nuclei, known not to be affected by AD. In parallel, PET images were post-processed using a validated automated software based on the computation of ROI normalized uptake ratios standard deviation (SUVr SD) with reference to a healthy control database (Siemens Scenium). Clinical diagnosis made during follow-up was considered as the gold-standard for patient classification (16 healthy controls and 10 AD patients).
Results
Both methods provided cortical ROI indices that were significantly different between controls and AD patients. The area under the ROC curve for control/AD classification was statistically identical (0.96 for NIBEM IR and 0.95 for Scenium SUVr SD). At the optimal threshold, the sensitivity, specificity, accuracy, positive predictive value, and negative predictive value were, respectively, 100%, 88%, 92%, 83%, and 100% for both Scenium SUVr SD and NIBEM IR methods.
Conclusion
This preliminary study shows that interval-valued reconstruction allows self-consistent analysis of brain 18F-FDG PET data, yielding diagnostic performances that seem promising with respect to those of a commercial post-processing software based on SUVr SD analysis.







Similar content being viewed by others
Notes
Available at: https://www.gin.cnrs.fr/AAL2.
References
Prince M, Wimo AGM, Ali GC, Wu YT, Prina M. World Alzheimer Report 2015: the global impact of dementia: an analysis of prevalence, incidence, cost and trends. London: Alzheimer’s Disease International; 2015.
Castro DM, Dillon C, Machnicki G, Allegri RF. The economic cost of Alzheimer’s disease: family or public health burden? Dement Neuropsychol. 2010;4(4):262–7.
Bloudek LM, Spackman DE, Blankenburg M, Sullivan SD. Review and meta-analysis of biomarkers and diagnostic imaging in Alzheimer’s disease. J Alzheimers Dis. 2011;26(4):627–45.
Perani D, Cerami C, Caminiti SP, Santangelo R, Coppi E, Ferrari L, et al. Cross-validation of biomarkers for the early differential diagnosis and prognosis of dementia in a clinical setting. Eur J Nucl Med Mol Imaging. 2016;43(3):499–508.
Siderowf A, Aarsland D, Mollenhauer B, Goldman JG, Ravina B. Biomarkers for cognitive impairment in Lewy body disorders: btatus and relevance for clinical trials: biomarkers of cognitive impairment. Mov Disord. 2018;33(4):528–36.
Petrella JR. Neuroimaging and the search for a cure for Alzheimer disease. Radiology. 2013;269(3):671–91.
Nasrallah IM, Wolk DA. Multimodality imaging of Alzheimer disease and other neurodegenerative dementias. J Nucl Med. 2014;55(12):2003–111.
Bohnen NI, Djang DS, Herholz K, Anzai Y, Minoshima S. Effectiveness and safety of 18F-FDG PET in the evaluation of dementia: a review of the recent literature. J Nucl Med. 2012;53(1):59–71.
Kato T, Inui Y, Nakamura A, Ito K. Brain fluorodeoxyglucose (FDG) PET in dementia. Ageing Res Rev. 2016;30:73–84.
Nestor PJ, Altomare D, Festari C, Drzezga A, Rivolta J, Walker Z, Bouwman F, Orini S, Law I, Agosta F, Arbizu J, Boccardi M, Nobili F, Frisoni GB. Clinical utility of FDG-PET for the differential diagnosis among the main forms of dementia. Eur J Nucl Med Mol Imaging. 2018;45(9):1509–25.
Ng S, Villemagne VL, Berlangieri S, Lee ST, Cherk M, Gong SJ, et al. Visual assessment versus quantitative assessment of 11C-PIB PET and 18F-FDG PET for detection of Alzheimer’s disease. J Nucl Med. 2007;48(4):547–52.
Mosconi L, Mistur R, Switalski R, Tsui WH, Glodzik L, Li Y, et al. FDG-PET changes in brain glucose metabolism from normal cognition to pathologically verified Alzheimer’s disease. Eur J Nucl Med Mol Imaging. 2009;36(5):811–22.
Varrone A, Asenbaum S, Vander Borght T, Booij J, Nobili F, Någren K, et al. EANM procedure guidelines for PET brain imaging using 18F-FDG, version 2. Eur J Nucl Med Mol Imaging. 2009;12:2103–10.
Giovacchini G, Squitieri F, Esmaeilzadeh M, Milano A, Mansi L, Ciarmiello A. PET translates neurophysiology into images: a review to stimulate a network between neuroimaging and basic research. J Cell Physiol. 2011;226(4):948–61.
Anchisi D, Borroni B, Franceschi M, Kerrouche N, Kalbe E, Beuthien-Beumann B, et al. Heterogeneity of brain glucose metabolism in mild cognitive impairment and clinical progression to Alzheimer disease. Arch Neurol. 2005;62(11):1728–33.
Silverman DH, Small GW, Chang CY, Lu CS, De Kung AMA, Chen W, et al. Positron emission tomography in evaluation of dementia: regional brain metabolism and long-term outcome. JAMA. 2001;286(17):2120–7.
Grimmer T, Wutz C, Alexopoulos P, Drzezga A, Förster S, Förstl H, et al. Visual versus fully automated analyses of 18F-FDG and amyloid PET for prediction of dementia due to Alzheimer disease in mild cognitive impairment. J Nucl Med. 2016;57(2):204–7.
Lehman VT, Carter RE, Claassen DO, Murphy RC, Lowe V, Petersen RC, et al. Visual assessment versus quantitative three-dimensional stereotactic surface projection fluorodeoxyglucose positron emission tomography for detection of mild cognitive impairment and Alzheimer disease. Clin Nucl Med. 2012;37(8):721–6.
Morbelli S, Brugnolo A, Bossert I, Buschiazzo A, Frisoni GB, Galluzzi S, et al. Visual versus semi-quantitative analysis of 18F-FDG-PET in amnestic MCI: an European Alzheimer’s Disease Consortium (EADC) project. J Alzheimers Dis. 2015;44(3):815–26.
Ding Y, Sohn JH, Kawczynski MG, Trivedi H, Harnish R, Jenkins NW, et al. A deep learning model to predict a diagnosis of Alzheimer disease by using 18F-FDG PET of the brain. Radiology. 2018;290(2):456–64.
Liu M, Cheng D, Wang K, Wang Y. Multi-modality cascaded convolutional neural networks for Alzheimer’s disease diagnosis. Neuroinformatics. 2018;16:295–308.
De Carli F, Nobili F, Pagani M, Bauckneht M, Massa F, Grazzini M, et al. Accuracy and generalization capability of an automatic method for the detection of typical brain hypometabolism in prodromal Alzheimer disease. Eur J Nucl Med Mol Imaging. 2019;46(2):334–47.
Cerami C, Della Rosa PA, Magnani G, Santangelo R, Marcone A, Cappa SF, et al. Brain metabolic maps in mild cognitive impairment predict heterogeneity of progression to dementia. Neuroimage Clin. 2014;5(7):187–94.
Perani D, Della Rosa PA, Cerami C, Gallivanone F, Fallanca F, Vanoli EG, Panzacchi A, Nobili F, Pappata S, Marcone A, Garibotto V, Castiglioni I, Magnani G, Cappa SF, Gianolli L. Validation of an optimized SPM procedure for FDG-PET in dementia diagnosis in a clinical setting. Neuroimage Clin. 2014;6:445–54.
Yamane T, Ikari Y, Nishio T, Ishii K, Ishii K, Kato T, et al. Visual-statistical interpretation of (18)F-FDG-PET images for characteristic Alzheimer patterns in a multicenter study: inter-rater concordance and relationship to automated quantitative evaluation. AJNR Am J Neuroradiol. 2014;35(2):244–9.
Caminiti SP, Ballarini T, Sala A, Cerami C, Presotto L, Santangelo R, et al. FDG-PET and CSF biomarker accuracy in prediction of conversion to different dementias in a large multicentre MCI cohort. Neuroimage Clin. 2018;28(18):167–77.
Brugnolo A, De Carli F, Pagani M, Morbelli S, Jonsson C, Chincarini A, et al. Head-to-Head comparison among semi-quantification tools of brain FDG-PET to aid the diagnosis of prodromal Alzheimer’s disease. J Alzheimers Dis. 2019;68(1):383–94.
Kucharczak F, Loquin K, Buvat I, Strauss O, Mariano-Goulart D. Interval-based reconstruction for uncertainty quantification in PET. Phys Med Biol. 2018;63(3):035014.
Kucharczak F, Ben BF, Strauss O, Mariano-Goulart D. Confidence interval constraint-based regularization framework for PET quantization. IEEE Trans Med Imaging. 2019;38(6):1513–23.
Jena A, Taneja S, Goel R, Renjen P, Negi P. Reliability of semiquantitative 18F-FDG PET parameters derived from simultaneous brain PET/MRI: a feasibility study. Eur J Radiol. 2014;83(7):1269–74.
Dubois B, Feldman HH, Jacova C, DeKosky ST, Barberger-Gateau P, Cummings J, et al. Research criteria for the diagnosis of Alzheimer’s disease: revising the NINCDS-ADRDA criteria. Lancet Neurol. 2007;6:734–46.
Shepp L, Vardi Y. Maximum likelihood reconstruction for emission tomography. IEEE Trans Medical Imaging. 1982;1(2):113–22.
Lange K, Carson R. EM reconstruction algorithms for emission and transmission tomography. J Comput Assist Tomogr. 1984;8(2):306–16.
Defrise M, Kinahan PE, Michel DT, Sibomana C, Newport MD. Exact and approximate rebinning algorithms for 3-D PET data. IEEE Trans Med Imaging. 1997;16(2):194–204.
Rolls ET, Joliot M, Tzourio-Mazoyer N. Implementation of a new parcellation of the orbitofrontal cortex in the automated anatomical labeling atlas. NeuroImage. 2015;122:1–5.
Mosconi L. Brain glucose metabolism in the early and specific diagnosis of Alzheimer’s disease. FDG-PET studies in MCI and AD. Eur J Nucl Med Mol Imaging. 2005;32:486–510.
Perolat J, Couso I, Loquin K, Strauss O. Generalizing the Wilcoxon rank-sum test for interval data. J Approx Reason. 2015;56:108–21.
Smets P. Analyzing the combination of conflicting belief functions. Inf Fus. 2007;8(4):387–412.
Hanley JA, McNeil BJ. The meaning and use of the area under a Receiver Operating Characteristic (ROC) curve. Radiology. 1982;143:29–36.
Petersen RC, Aisen PS, Beckett LA, et al. Alzheimer’s Disease Neuroimaging Initiative (ADNI): clinical characterization. Neurology. 2010;74(3):201–9.
Acknowledgements
The authors are grateful to the nuclear medicine staff of University Hospital Gui de Chauliac in Montpellier for their help regarding the constitution of the database used in this study. The first author was the recipient of a grant funded by the Siemens Healthineers company. There is no other potential conflict of interest relevant to this article.
Funding
Florentin Kucharczak was the recipient of a grant funded by the Siemens Healthineers company. There is no other potential conflict of interest relevant to this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed were in accordance with the ethical standards of the institutional and/or national research committee and with the principles of the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. The authors received a favorable opinion of the Institutional Review Board of University Hospital Center of Montpellier under Grant number 2018-IRB-MTP-09-08.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kucharczak, F., Suau, M., Strauss, O. et al. Brain 18F-FDG PET analysis via interval-valued reconstruction: proof of concept for Alzheimer’s disease diagnosis. Ann Nucl Med 34, 565–574 (2020). https://doi.org/10.1007/s12149-020-01490-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-020-01490-7