Abstract
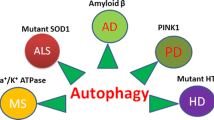
Autophagy is a catabolic pathway by which misfolded proteins or damaged organelles are engulfed by autophagosomes and then transported to lysosomes for degradation. Recently, a great improvement has been done to explain the molecular mechanisms and roles of autophagy in several important cellular metabolic processes. Besides being a vital clearance pathway or a cell survival pathway in response to different stresses, autophagy dysfunction, either upregulated or down-regulated, has been suggested to be linked with numerous neurodegenerative disorders like Alzheimer’s disease, Parkinson’s disease, Huntington’s disease, and Amyotrophic lateral sclerosis. Impairment at different stages of autophagy results in the formation of large protein aggregates and damaged organelles, which leads to the onset and progression of different neurodegenerative disorders. This article elucidates the recent progress about the role of autophagy in neurodegenerative disorders and explains how autophagy dysfunction is linked with the pathogenesis of such disorders as well as the novel potential autophagy-associated therapies for treating them.
Graphical abstract



Similar content being viewed by others
Data availability
Not applicable.
References
Corti O, Blomgren K, Poletti A, Beart PM (2020) Autophagy in neurodegeneration: new insights underpinning therapy for neurological diseases. J Neurochem 154:354–371
Rodolfo C, Di Bartolomeo S, Cecconi F (2016) Autophagy in stem and progenitor cells. Cell Mol Life Sci 73:475–496. https://doi.org/10.1007/s00018-015-2071-3
Klionsky DJ, Codogno P, Cuervo AM, Deretic V, Elazar Z, Fueyo-Margareto J, Gewirtz DA, Kroemer G, Levine B, Mizushima N et al (2010) A comprehensive glossary of autophagy-related molecules and processes. Autophagy 6:438–448
Lin Y, Huang Y, Chen L, Chu P (2015) Autophagy in cancer stem/progenitor cells. Cancer Chemother Pharmacol 75:879–886. https://doi.org/10.1007/s00280-014-2634-2
Guan J, Simon A, Prescott M, Menendez J, Liu F, Wang F, Wang C, Wolvetang E, Vazquez-Martin A, Zhang J (2013) Autophagy in stem cells. Autophagy 9:830–849. https://doi.org/10.4161/auto.24132
Lei Y, Zhang D, Yu J, Dong H, Zhang J, Yang S (2017) Targeting autophagy in cancer stem cells as an anticancer therapy. Cancer Lett 393:33–39. https://doi.org/10.1016/j.canlet.2017.02.012
Guo JY, White E (2017) Autophagy, metabolism, and cancer. Cold Spring Harb Symp Quant Biol 81:73–78. https://doi.org/10.1101/sqb.2016.81.030981
Hua F, Shang S, Hu ZW (2017) Seeking new anti-cancer agents from autophagy-regulating natural products. J Asian Nat Prod Res 19:305–313. https://doi.org/10.1080/10286020.2017.1304385
Jacob JA, Salmani JM, Jiang Z, Feng L, Song J, Jia X, Chen B (2017) Autophagy: an overview and its roles in cancer and obesity. Clin Chim Acta 468:85–89. https://doi.org/10.1016/j.cca.2017.01.028
Chen K, Yuan R, Geng S, Zhang Y, Ran T, Kowalski E, Liu J, Li L (2017) Toll-interacting protein deficiency promotes neurodegeneration via impeding autophagy completion in high-fat diet-fed ApoE-/- mouse model. Brain Behav Immun 59:200–210. https://doi.org/10.1016/j.bbi.2016.10.002
Hwang CJ, Kim YE, Son DJ, Park MH, Choi DY, Park PH, Hellstrom M, Han SB, Oh KW, Park EK, Hong JT (2017) Parkin deficiency exacerbate ethanol-induced dopaminergic neurodegeneration by P38 pathway dependent inhibition of autophagy and mitochondrial function. Redox Biol 11:456–468. https://doi.org/10.1016/j.redox.2016.12.008
Menzies FM, Fleming A, Caricasole A, Bento CF, Andrews SP, Ashkenazi A et al (2017) Autophagy and neurodegeneration: pathogenic mechanisms and therapeutic opportunities. Neuron 93:1015–1034. https://doi.org/10.1016/j.neuron.2017.01.022
Plaza-Zabala A, Sierra-Torre V, Sierra A (2017) Autophagy and microglia: novel partners in neurodegeneration and aging. Int J Mol Sci 18:E598. https://doi.org/10.3390/ijms18030598
Yuan B, Shen H, Lin L, Su T, Zhong L, Yang Z (2017) Autophagy promotes microglia activation through Beclin-1-Atg5 pathway in intracerebral hemorrhage. Mol Neurobiol 54:115–124. https://doi.org/10.1007/s12035-015-9642-z
Zhong Z, Sanchez-Lopez E, Karin M (2016) Autophagy, NLRP3 inflammasome and auto-inflammatory/immune diseases. Clin Exp Rheumatol 34:12–16
Suh HW, Kim JK, Kim TS, Jo EK (2017) New insights into vitamin D and autophagy in inflammatory bowel diseases. Curr Med Chem 24:898–910. https://doi.org/10.2174/0929867323666161202151856
Miettinen TP, Bjorklund M (2016) The mevalonate pathway as a metabolic requirement for autophagy-implications for growth control, proteostasis, and disease. Mol Cell Oncol 3:e1143546. https://doi.org/10.1080/23723556.2016.1143546
Jia G, Sowers JR (2015) Autophagy: a housekeeper in cardiorenal metabolic health and disease. Biochim Biophys Acta 1852:219–224. https://doi.org/10.1016/j.bbadis.2014.06.025
Wang F, Jia J, Rodrigues B (2017) Autophagy, metabolic disease, and pathogenesis of heart dysfunction. Can J Cardiol 33:850–859. https://doi.org/10.1016/j.cjca.2017.01.002
Zhang S, Lin X, Li G, Shen X, Niu D, Lu G, Fu X, Chen Y, Cui M, Bai Y (2017) Knockout of Eva1a leads to rapid development of heart failure by impairing autophagy. Cell Death Dis 8:e2586. https://doi.org/10.1038/cddis.2017.17
Ren SY, Xu X (2015) Role of autophagy in metabolic syndrome-associated heart disease. Biochim Biophys Acta 1852:225–231. https://doi.org/10.1016/j.bbadis.2014.04.029
Rubinsztein David C, Marino G, Kroemer G (2011) Autophagy and aging. Cell 146:682–695. https://doi.org/10.1016/j.cell.2011.07.030
Mizushima N, Levine B (2010) Autophagy in mammalian development and differentiation. Nat Cell Biol 12:823–830. https://doi.org/10.1038/ncb0910-823
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147:728–741. https://doi.org/10.1016/j.cell.2011.10.026
He L, Zhang J, Zhao J, Ma N, Kim SW, Qiao S et al (2018) Autophagy: the last defense against cellular nutritional stress. Adv Nutr Bethesda Md 9:493–504
Doherty J, Baehrecke EH (2018) Life, death and autophagy. Nat Cell Biol 20:1110–1117
Xilouri M, Stefanis L (2010) Autophagy in the central nervous system: implications for neurodegenerative disorders. CNS Neurol Disord Drug Targets 9:701–719. https://doi.org/10.2174/187152710793237421
Cai Z, Zeng W, Tao K, Wang ZEB, Yang Q (2015) Chaperone mediated autophagy: roles in neuroprotection. Neurosci Bull 31:452–458. https://doi.org/10.1007/s12264-015-1540-x
Kenney DL, Benarroch EE (2015) The autophagy-lysosomal pathway: general concepts and clinical implications. Neurology 85:634–645
Suzuki K, Ohsumi Y (2007) Molecular machinery of autophagosome formation in yeast, Saccharomyces cerevisiae. FEBS Lett 581:2156–2161
Tsukada M, Ohsumi Y (1993) Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae. FEBS Lett 333:169–174
Ogura K, Wicky C, Magnenat L, Tobler H, Mori I, Muller F et al (1994) Caenorhabditis elegans unc-51 gene required for axonal elongation encodes a novel serine/threonine kinase. Genes Dev 8:2389–2400
Wang JL, Xu CJ (2020) Astrocytes autophagy in aging and neurodegenerative disorders. Biomed Pharmacother 122:109691
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R et al (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889
Komatsu M, Waguri S, Chiba T, Murata S, Iwata J, Tanida I et al (2006) Loss of autophagy in the central nervous system causes neurodegeneration in mice. Nature 441:880–884
Dice JF (1990) Peptide sequences that target cytosolic proteins for lysosomal proteolysis. Trends Biochem Sci 15:305–309
Kaushik S, Cuervo AM (2012) Chaperone-mediated autophagy: a unique way to enter the lysosome world. Trends Cell Biol 22:407–417
Parzych KR, Klionsky DJ (2014) An overview of autophagy: morphology, mechanism, and regulation. Antioxid Redox Signal 20:460–473
Bauer PO, Goswami A, Wong HK, Okuno M, Kurosawa M, Yamada M et al (2010) Harnessing chaperone-mediated autophagy for the selective degradation of mutant huntingtin protein. Nat Biotechnol 28:256–263
Wang Y, Martinez-Vicente M, Kruger U, Kaushik S, Wong E, Mandelkow E-M et al (2009) Tau fragmentation, aggregation and clearance: the dual role of lysosomal processing. Hum Mol Genet 18:4153–4170
Cuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D (2004) Impaired degradation of mutant alpha-synuclein by chaperone-mediated autophagy. Science 305:1292–1295
Martinez-Vicente M, Talloczy Z, Kaushik S, Massey AC, Mazzulli J, Mosharov EV et al (2008) Dopamine-modified alpha-synuclein blocks chaperone-mediated autophagy. J Clin Invest 118:777–788
Kabuta T, Furuta A, Aoki S, Furuta K, Wada K (2008) Aberrant interaction between Parkinson disease-associated mutant UCH-L1 and the lysosomal receptor for chaperone-mediated autophagy. J Biol Chem 283:23731–23738
Orenstein SJ, Kuo S-H, Tasset I, Arias E, Koga H, Fernandez-Carasa I et al (2013) Interplay of LRRK2 with chaperone-mediated autophagy. Nat Neurosci 16:394–406
Sahu R, Kaushik S, Clement CC, Cannizzo ES, Scharf B, Follenzi A et al (2011) Microautophagy of cytosolic proteins by late endosomes. Dev Cell 20:131–139
Sharma M, Burre J, Bronk P, Zhang Y, Xu W, Sudhof TC (2012) CSPα knockout causes neurodegeneration by impairing SNAP-25 function. EMBO J 31:829–841
Uytterhoeven V, Lauwers E, Maes I, Miskiewicz K, Melo MN, Swerts J et al (2015) Hsc70–4 Deforms membranes to promote synaptic protein turnover by endosomal microautophagy. Neuron 88:735–748
Kim DH, Sarbassov DD, Ali SM, King JE, Latek RR, Erdjument-Bromage H et al (2002) mTOR interacts with raptor to form a nutrient-sensitive complex that signals to the cell growth machinery. Cell 110:163–175
Hosokawa N, Hara T, Kaizuka T, Kishi C, Takamura A, Miura Y et al (2009) Nutrient dependent mTORC1 association with the ULK1-Atg13-FIP200 complex required for autophagy. Mol Biol Cell 20:1981–1991
Mercer CA, Kaliappan A, Dennis PB (2009) A novel, human Atg13 binding protein, Atg101, interacts with ULK1 and is essential for macroautophagy. Autophagy 5:649–662
Russell RC, Tian Y, Yuan H, Park HW, Chang Y-Y, Kim J et al (2013) ULK1 induces autophagy by phosphorylating Beclin-1 and activating VPS34 lipid kinase. Nat Cell Biol 15:741–750
Kabeya Y, Mizushima N, Yamamoto A, Oshitani-Okamoto S, Ohsumi Y, Yoshimori T (2004) LC3, GABARAP and GATE16 localize to autophagosomal membrane depending on form-II formation. J Cell Sci 117:2805–2812
Romanov J, Walczak M, Ibiricu I, Schuchner S, Ogris E, Kraft C et al (2012) Mechanism and functions of membrane binding by the Atg5-Atg12/Atg16 complex during autophagosome formation. EMBO J 31:4304–4317
Alemu EA, Lamark T, Torgersen KM, Birgisdottir AB, Larsen KB, Jain A et al (2012) ATG8 family proteins act as scaffolds for assembly of the ULK complex: sequence requirements for LC3-interacting region (LIR) motifs. J Biol Chem 287:39275–39290
Rogov V, Dotsch V, Johansen T, Kirkin V (2014) Interactions between autophagy receptors and ubiquitin-like proteins form the molecular basis for selective autophagy. Mol Cell 53:167–178
Sardiello M, Palmieri M, di Ronza A, Medina DL, Valenza M, Gennarino VA, Di Malta C, Donaudy F, Embrione V, Polishchuk RS, Banfi S, Parenti G, Cattaneo E, Ballabio A (2009) A gene network regulating lysosomal biogenesis and function. Science 325:473–477
Settembre C, Di Malta C, Polito VA, Garcia Arencibia M, Vetrini F, Erdin S, Erdin SU, Huynh T, Medina D, Colella P, Sardiello M, Rubinsztein DC, Ballabio A (2011) TFEB links autophagy to lysosomal biogenesis. Science 332:1429–1433. https://doi.org/10.1126/science.1204592
Kilpatrick K, Zeng Y, Hancock T, Segatori L (2015) Genetic and chemical activation of TFEB mediates clearance of aggregated α-synuclein. PLoS ONE 10:e0120819. https://doi.org/10.1371/journal.pone.0120819
Martina JA, Diab HI, Lishu L, Jeong AL, Patange S, Raben N, Puertollano R (2014) The nutrient-responsive transcription factor TFE3 promotes autophagy, lysosomal biogenesis, and clearance of cellular debris. Sci Signal 7:ra9
Medina DL, Fraldi A, Bouche V, Annunziata F, Mansueto G, Spampanato C, Puri C, Pignata A, Martina JA, Sardiello M, Palmieri M, Polishchuk R, Puertollano R, Ballabio A (2011) Transcriptional activation of lysosomal exocytosis promotes cellular clearance. Dev Cell 21:421–430
Cheli Y, Ohanna M, Ballotti R, Bertolotto C (2011) Fifteen-year quest for microphthalmia-associated transcription factor target genes. Pigment Cell Melanoma Res 23:27–40
Ploper D, Taelman VF, Robert L, Perez BS, Titz B, Chen HW, Graeber TG, von Euw E, Ribas A, De Robertis EM (2015) MITF drives endolysosomal biogenesis and potentiates Wnt signaling in melanoma cells. Proc Natl Acad Sci U S A 112:E420–E429
Perera RM, Stoykova S, Nicolay BN, Ross KN, Fitamant J, Boukhali M et al (2015) Transcriptional control of autophagy-lysosome function drives pancreatic cancer metabolism. Nature 524:361–365. https://doi.org/10.1038/nature14587
Palmieri M, Impey S, Kang H, di Ronza A, Pelz C, Sardiello M, Ballabio A (2011) Characterization of the CLEAR network reveals an integrated control of cellular clearance pathways. Hum Mol Genet 20:3852–3866
Chen S, Guo D, Lei B, Bi J, Yang H (2020) Biglycan protects human neuroblastoma cells from nitric oxide-induced death by inhibiting AMPK-mTOR mediated autophagy and intracellular ROS level. Biotechnol Lett 42:657–668
Ganley IG, Lam DH, Wang J, Ding X, Chen S, Jiang X (2009) ULK1·ATG13·FIP200 Complex mediates mTOR signaling and is essential for autophagy. J Biol Chem 284:12297–12305. https://doi.org/10.1074/jbc.M900573200
Hosokawa N, Hara T, Kaizuka T, Kishi C, Takamura A, Miura Y, Iemura S-i Natsume T, Takehana K, Yamada N, Guan J-L, Oshiro N, Mizushima N (2009) Nutrient-dependent mTORC1 association with the ULK1–Atg13–FIP200 complex required for autophagy. Mol Biol Cell 20:1981–1991. https://doi.org/10.1091/mbc.E08-12-1248
Jung CH, Jun CB, Ro S-H, Kim Y-M, Otto NM, Cao J, Kundu M, Kim D-H (2009) ULK-Atg13-FIP200 complexes mediate mTOR signaling to the autophagy machinery. Mol Biol Cell 20:1992–2003. https://doi.org/10.1091/mbc.E08-12-1249
Laplante M, Sabatini DM (2012) mTOR signaling in growth control and disease. Cell 149:274–293. https://doi.org/10.1016/j.cell.2012.03.017
Loffler AS, Alers S, Dieterle AM, Keppeler H, Franz-Wachtel M, Kundu M, Campbell DG, Wesselborg S, Alessi DR, Stork B (2011) Ulk1-mediated phosphorylation of AMPK constitutes a negative regulatory feedback loop. Autophagy 7:696–706. https://doi.org/10.4161/auto.7.7.15451
Kim J, Kundu M, Viollet B, Guan K-L (2011) AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nat Cell Biol 13:132–141. https://doi.org/10.1038/ncb2152
Hara T, Nakamura K, Matsui M, Yamamoto A, Nakahara Y, Suzuki-Migishima R, Yokoyama M, Mishima K, Saito I, Okano H, Mizushima N (2006) Suppression of basal autophagy in neural cells causes neurodegenerative disease in mice. Nature 441:885–889. https://doi.org/10.1038/nature04724
Dooley Hannah C, Razi M, Polson Hannah E, Girardin Stephen E, Wilson Michael I, Tooze Sharon A (2014) WIPI2 Links LC3 conjugation with PI3P, autophagosome formation, and pathogen clearance by recruiting Atg12–5-16L1. Mol Cell 55:238–252. https://doi.org/10.1016/j.molcel.2014.05.021
Furuya N, Yu J, Byfield M, Pattingre S, Levine B (2005) The evolutionarily conserved domain of Beclin 1 is required for Vps34 binding, autophagy and tumor suppressor function. Autophagy 1:46–52. https://doi.org/10.4161/auto.1.1.1542
Russell RC, Tian Y, Yuan H, Park HW, Chang Y-Y, Kim J, Kim H, Neufeld TP, Dillin A, Guan K-L (2013) ULK1 induces autophagy by phosphorylating Beclin-1 and activating Vps34 lipid kinase. Nat Cell Biol 15:741–750. https://doi.org/10.1038/ncb2757
Dolzhenko E, van Vugt J, Shaw RJ, Bekritsky MA, van Blitterswijk M, Narzisi G, Ajay SS, Rajan V et al (2017) Detection of long repeat expansions from PCR-free whole-genome sequence data. Genome Res 27:1895–1903. https://doi.org/10.1101/gr.225672.117
Wang S, Xia P, Rehm M, Fan Z (2015) Autophagy and cell reprogramming. Cell Mol Life Sci 72:1699–1713. https://doi.org/10.1007/s00018-014-1829-3
Bento CF, Renna M, Ghislat G, Puri C, Ashkenazi A, Vicinanza M, Menzies FM, Rubinsztein DC (2016) Mammalian autophagy: how does it work? Annu Rev Biochem 85:685–713. https://doi.org/10.1146/annurev-biochem-060815-014556
Pankiv S, Clausen TH, Lamark T, Brech A, Bruun J-A, Outzen H, Overvatn A, Bjorkoy G, Johansen T (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282:24131–24145. https://doi.org/10.1074/jbc.m702824200
Bjorkoy G, Lamark T, Brech A, Outzen H, Perander M, Overvatn A, Stenmark H, Johansen T (2005) p62/SQSTM1 forms protein aggregates degraded by autophagy and has a protective effect on huntingtin-induced cell death. J Cell Biol 171:603. https://doi.org/10.1083/jcb.200507002
Wild P, Farhan H, McEwan DG, Wagner S, Rogov VV, Brady NR, Richter B, Korac J, Waidmann O, Choudhary C, Dotsch V, Bumann D, Dikic I (2011) Phosphorylation of the autophagy receptor optineurin restricts Salmonella growth. Science 333:228–233. https://doi.org/10.1126/science.1205405
Thurston TLM, Ryzhakov G, Bloor S, von Muhlinen N, Randow F (2009) The TBK1 adaptor and autophagy receptor NDP52 restricts the proliferation of ubiquitin-coated bacteria. Nat Immunol 10:1215-U1103. https://doi.org/10.1038/ni.1800
Kirkin V, Lamark T, Sou Y-S, Bjorkoy G, Nunn JL, Bruun J-A, Shvets E et al (2009) A role for NBR1 in autophagosomal degradation of ubiquitinated substrates. Mol Cell 33:505–516. https://doi.org/10.1016/j.molcel.2009.01.020
Filimonenko M, Isakson P, Finley KD, Anderson M, Jeong H, Melia TJ, Bartlett BJ et al (2010) The selective macroautophagic degradation of aggregated proteins requires the PI3P-binding protein Alfy. Mol Cell 38:265–279. https://doi.org/10.1016/j.molcel.2010.04.007
Mandell Michael A, Jain A, Arko-Mensah J, Chauhan S, Kimura T, Dinkins C, Silvestri G, Munch J, Kirchhoff F et al (2014) TRIM proteins regulate autophagy and can target autophagic substrates by direct recognition. Dev Cell 30:394–409. https://doi.org/10.1016/j.devcel.2014.06.013
Jager S, Bucci C, Tanida I, Ueno T, Kominami E, Saftig P, Eskelinen E-L (2004) Role for Rab7 in maturation of late autophagic vacuoles. J Cell Sci 117:4837. https://doi.org/10.1242/jcs.01370
Tanaka Y, Guhde G, Suter A, Eskelinen EL, Hartmann D, Lullmann-Rauch R, Janssen PM, Blanz J, von Figura K, Saftig P (2000) Accumulation of autophagic vacuoles and cardiomyopathy in LAMP-2-deficient mice. Nature 406:902–906. https://doi.org/10.1038/35022595
Epple UD, Suriapranata I, Eskelinen E-L, Thumm M (2001) Aut5/Cvt17p, a putative lipase essential for disintegration of autophagic bodies inside the vacuole. J Bacteriol 183:5942–5955. https://doi.org/10.1128/JB.183.20.5942-5955.2001
Tanida I, Minematsu-Ikeguchi N, Ueno T, Kominami E (2005) Lysosomal turnover, but not a cellular level, of endogenous LC3 is a marker for autophagy. Autophagy 1:84–91. https://doi.org/10.4161/auto.1.2.1697
Nah J, Yuan J, Jung YK (2015) Autophagy in neurodegenerative diseases: from mechanism to therapeutic approach. Mol Cells 38:381–389. https://doi.org/10.14348/molcells.2015.0034
Ravikumar B, Duden R, Rubinsztein DC (2002) Aggregate-prone proteins with polyglutamine and polyalanine expansions are degraded by autophagy. Hum Mol Genet 11:1107–1117. https://doi.org/10.1093/hmg/11.9.1107
Menzies FM, Fleming A, Rubinsztein DC (2015) Compromised autophagy and neurodegenerative diseases. Nat Rev Neurosci 16:345. https://doi.org/10.1038/nrn3961
Puyal J, Ginet V, Grishchuk Y, Truttmann AC, Clarke PG (2012) Neuronal autophagy as a mediator of life and death: contrasting roles in chronic neurodegenerative and acute neural disorders. Neuroscientist 18:224–236. https://doi.org/10.1177/1073858411404948
Levine B, Kroemer G (2008) Autophagy in the pathogenesis of disease. Cell 132:27–42. https://doi.org/10.1016/j.cell.2007.12.018
Nikoletopoulou V, Markaki M, Palikaras K, Tavernarakis N (2013) Crosstalk between apoptosis, necrosis and autophagy. Biochim Biophys Acta 1833:3448–3459. https://doi.org/10.1016/j.bbamcr.2013.06.001
Son JH, Shim JH, Kim KH, Ha JY, Han JY (2012) Neuronal autophagy and neurodegenerative diseases. Exp Mol Med 44:89–98. https://doi.org/10.3858/emm.2012.44.2.031
Martinet W, Agostinis P, Vanhoecke B, Dewaele M, De Meyer GR (2009) Autophagy in disease: a double-edged sword with therapeutic potential. Clin Sci 116:697–712. https://doi.org/10.1042/CS20080508
Renna M, Jimenez-Sanchez M, Sarkar S, Rubinsztein DC (2010) Chemical inducers of autophagy that enhance the clearance of mutant proteins in neurodegenerative diseases. J Biol Chem 285:11061–11067. https://doi.org/10.1074/jbc.R109.072181
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451:1069–1075. https://doi.org/10.1038/nature06639
He C, Levine B (2010) The beclin 1 interactome. Curr Opin Cell Biol 22:140–149. https://doi.org/10.1016/j.ceb.2010.01.001
Markossian KA, Kurganov BI (2004) Protein folding, misfolding, and aggregation. Formation of inclusion bodies and aggresomes. Biochemistry 69:971–984. https://doi.org/10.1023/b%3Abiry.0000043539.07961.4c
Ross CA, Poirier MA (2004) Protein aggregation and neurodegenerative disease. Nat Med 10:S10–S17. https://doi.org/10.1038/nm1066
Alzheimer’s A, (2016) 2016 Alzheimer’s disease facts and figures. Alzheimers Dement 12:459–509. https://doi.org/10.1016/j.jalz.2016.03.001
Liu X, Hou D, Lin F, Luo J, Xie J, Wang Y et al (2019) The role of neurovascular unit damage in the occurrence and development of Alzheimer’s disease. Rev Neurosci 30:477–484
Yoon S-Y, Kim D-H (2016) Alzheimer’s disease genes and autophagy. Brain Res 1649:201–209. https://doi.org/10.1016/j.brainres.2016.03.018
Yu WH, Cuervo AM, Kumar A, Peterhoff CM, Schmidt SD, Lee JH et al (2005) Macroautophagy–a novel Beta-amyloid peptide-generating pathway activated in Alzheimer’s disease. J Cell Biol 171:87–98. https://doi.org/10.1083/jcb.200505082
Hung SY, Huang WP, Liou HC, Fu WM (2009) Autophagy protects neuron from Abeta-induced cytotoxicity. Autophagy 5:502–510. https://doi.org/10.4161/auto.5.4.8096
Wang H, Ma J, Tan Y, Wang Z, Sheng C, Chen S, Ding J (2010) Amyloid-beta1-42 induces reactive oxygen species-mediated autophagic cell death in U87 and SH-SY5Y cells. JAD 21:597–610. https://doi.org/10.3233/jad-2010-091207
Nixon RA, Wegiel J, Kumar A, Yu WH, Peterhoff C, Cataldo A, Cuervo AM (2005) Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J Neuropathol Exp Neurol 64:113–122. https://doi.org/10.1093/jnen/64.2.113
Nilsson P, Loganathan K, Sekiguchi M, Matsuba Y, Hui K, Tsubuki S, Tanaka M, Iwata N, Saito T, Saido Takaomi C (2013) Aβ secretion and plaque formation depend on autophagy. Cell Rep 5:61–69. https://doi.org/10.1016/j.celrep.2013.08.042
Boland B, Kumar A, Lee S, Platt FM, Wegiel J, Yu WH, Nixon RA (2008) Autophagy induction and autophagosome clearance in neurons: relationship to autophagic pathology in Alzheimer’s disease. J Neurosci 28:6926–6937. https://doi.org/10.1523/JNEUROSCI.0800-08.2008
Spilman P, Podlutskaya N, Hart MJ, Debnath J, Gorostiza O, Bredesen D, Richardson A, Strong R, Galvan V (2010) Inhibition of mTOR by rapamycin abolishes cognitive deficits and reduces amyloid-β levels in a mouse model of Alzheimer’s disease. PLoS ONE 5:e9979. https://doi.org/10.1371/journal.pone.0009979
Tian Y, Bustos V, Flajolet M, Greengard P (2011) A small-molecule enhancer of autophagy decreases levels of Aβ and APP-CTF via Atg5-dependent autophagy pathway. FASEB J 25:1934–1942. https://doi.org/10.1096/fj.10-175158
Vingtdeux V, Chandakkar P, Zhao H, d’Abramo C, Davies P, Marambaud P (2011) Novel synthetic small-molecule activators of AMPK as enhancers of autophagy and amyloid-β peptide degradation. FASEB J 5:219–231. https://doi.org/10.1096/fj.10-167361
Lee VMY, Goedert M, Trojanowski JQ (2001) Neurodegenerative tauopathies. Annu Rev Neurosci 24:1121–1159. https://doi.org/10.1146/annurev.neuro.24.1.1121
Majid T, Ali YO, Venkitaramani DV, Jang M-K, Lu H-C, Pautler RG (2014) In vivo axonal transport deficits in a mouse model of fronto-temporal dementia. Neuroimage Clin 4:711–717. https://doi.org/10.1016/j.nicl.2014.02.005
Butzlaff M, Hannan SB, Karsten P, Lenz S, Ng J, Voßfeldt H, Prußing K, Pflanz R, Schulz JB, Rasse T, Voigt A (2015) Impaired retrograde transport by the Dynein/Dynactin complex contributes to Tau-induced toxicity. Hum Mol Genet 24:3623–3637. https://doi.org/10.1093/hmg/ddv107
Caccamo A, Majumder S, Richardson A, Strong R, Oddo S (2010) Molecular interplay between mammalian target of rapamycin (mTOR), amyloid-β, and Tau. J Biol Chem 285:13107–13120. https://doi.org/10.1074/jbc.m110.100420
Berger Z, Ravikumar B, Menzies FM, Oroz LG, Underwood BR, Pangalos MN, Schmitt I, Wullner U, Evert BO, O’Kane CJ, Rubinsztein DC (2006) Rapamycin alleviates toxicity of different aggregate-prone proteins. Hum Mol Genet 15:433–442. https://doi.org/10.1093/hmg/ddi458
Majumder S, Richardson A, Strong R, Oddo S (2011) Inducing autophagy by rapamycin before, but not after, the formation of plaques and tangles ameliorates cognitive deficits. PLoS ONE 6:e25416. https://doi.org/10.1371/journal.pone.0025416
Rodríguez-Navarro JA, Rodríguez L, Casarejos MJ, Solano RM, Gómez A, Perucho J, Cuervo AM, García de Yébenes J, Mena MA (2010) Trehalose ameliorates dopaminergic and tau pathology in parkin deleted/tau overexpressing mice through autophagy activation. Neurobiol Dis 39:423–438. https://doi.org/10.1016/j.nbd.2010.05.014
Schaeffer V, Lavenir I, Ozcelik S, Tolnay M, Winkler DT, Goedert M (2012) Stimulation of autophagy reduces neurodegeneration in a mouse model of human tauopathy. Brain 135:2169–2177. https://doi.org/10.1093/brain/aws143
Perez SE, He B, Nadeem M, Wuu J, Ginsberg SD, Ikonomovic MD, Mufson EJ (2015) Hippocampal endosomal, lysosomal and autophagic dysregulation in mild cognitive impairment: correlation with Aβ and Tau pathology. J Neuropathol Exp Neurol 74:345–358. https://doi.org/10.1097/NEN.0000000000000179
Collin L, Bohrmann B, Göpfert U, Oroszlan-Szovik K, Ozmen L, Grüninger F (2014) Neuronal uptake of tau/pS422 antibody and reduced progression of tau pathology in a mouse model of Alzheimer‘s disease. Brain 137:2834–2846. https://doi.org/10.1093/brain/awu213
Lee J-H, Yu WH, Kumar A, Lee S, Mohan PS, Peterhoff CM, Wolfe DM, Martinez-Vicente M, Massey AC, Sovak G, Uchiyama Y, Westaway D, Sisodia SS, Cuervo AM, Nixon RA (2010) Lysosomal proteolysis and autophagy require presenilin 1 and are disrupted by Alzheimer-related PS1 mutations. Cell 141:1146–1158. https://doi.org/10.1016/j.cell.2010.05.008
Bustos V, Pulina MV, Bispo A, Lam A, Flajolet M, Gorelick FS, Greengard P (2017) Phosphorylated Presenilin 1 decreases β-amyloid by facilitating autophagosome–lysosome fusion. Proc Natl Acad Sci USA 114:7148–7153. https://doi.org/10.1073/pnas.1705240114
Yang D-S, Stavrides P, Saito M, Kumar A, Rodriguez-Navarro JA, Pawlik M, Huo C, Walkley SU, Saito M, Cuervo AM, Nixon RA (2014) Defective macroautophagic turnover of brain lipids in the TgCRND8 Alzheimer mouse model: prevention by correcting lysosomal proteolytic deficits. Brain 137:3300–3318. https://doi.org/10.1093/brain/awu278
Tammineni P, Ye X, Feng T, Aikal D, Cai Q (2017) Impaired retrograde transport of axonal autophagosomes contributes to autophagic stress in Alzheimer’s disease neurons. Elife 6:e21776. https://doi.org/10.7554/eLife.21776
Pickford F, Masliah E, Britschgi M, Lucin K, Narasimhan R, Jaeger PA, Small S, Spencer B, Rockenstein E, Levine B, Wyss-Coray T (2008) The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid β accumulation in mice. J Clin Invest 118:2190–2199. https://doi.org/10.1172/JCI33585
Small Scott A, Kent K, Pierce A, Leung C, Kang Min S, Okada H, Honig L, Vonsattel JP, Kim TW (2005) Model-guided microarray implicates the retromer complex in Alzheimer’s disease. Ann Neurol 58:909–919. https://doi.org/10.1002/ana.20667
Rohn TT, Wirawan E, Brown RJ, Harris JR, Masliah E, Vandenabeele P (2011) Depletion of Beclin-1 due to proteolytic cleavage by caspases in the Alzheimer’s disease brain. Neurobiol Dis 43:68–78. https://doi.org/10.1016/j.nbd.2010.11.003
Pajares M, Jiménez-Moreno N, García-Yagüe ÁJ, Escoll M, de Ceballos ML, Van Leuven F, Rábano A, Yamamoto M, Rojo AI, Cuadrado A (2016) Transcription factor NFE2L2/NRF2 is a regulator of macroautophagy genes. Autophagy 12:1902–1916. https://doi.org/10.1080/15548627.2016.1208889
Jo C, Gundemir S, Pritchard S, Jin YN, Rahman I, Johnson GVW (2014) Nrf2 reduces levels of phosphorylated tau protein by inducing autophagy adaptor protein NDP52. Nat Commun 5:3496–3496. https://doi.org/10.1038/ncomms4496
Harold D, Abraham R, Hollingworth P, Sims R, Gerrish A, Hamshere M, Singh Pahwa J, Moskvina V, Dowzell K, Williams A, Jones N, Thomas C et al (2009) Genome-wide association study identifies variants at CLU and PICALM associated with Alzheimer’s disease, and shows evidence for additional susceptibility genes. Nat Genet 41:1088–1093. https://doi.org/10.1038/ng.440
Jun G, Naj AC, Beecham GW, Wang L-S, Buros J, Gallins PJ, Buxbaum JD, Ertekin-Taner N, Fallin MD, Friedland R et al (2010) Meta-analysis confirms CR1, CLU, and PICALM as Alzheimer’s disease risk loci and reveals interactions with APOE genotypes. Arch Neurol 67:1473–1484. https://doi.org/10.1001/archneurol.2010.201
Ando K, Brion J-P, Stygelbout V, Suain V, Authelet M, Dedecker R, Chanut A et al (2013) Clathrin adaptor CALM/PICALM is associated with neurofibrillary tangles and is cleaved in Alzheimer’s brains. Acta Neuropathol 125:861–878. https://doi.org/10.1007/s00401-013-1111-z
Ando K, Tomimura K, Sazdovitch V, Suain V, Yilmaz Z, Authelet M, Ndjim M, Vergara C, Belkouch M, Potier M-C, Duyckaerts C, Brion J-P (2016) Level of PICALM, a key component of clathrin-mediated endocytosis, is correlated with levels of phosphotau and autophagy-related proteins and is associated with tau inclusions in AD, PSP and Pick disease. Neurobiol Dis 94:32–43. https://doi.org/10.1016/j.nbd.2016.05.017
Moreau K, Fleming A, Imarisio S, Lopez Ramirez A, Mercer JL, Jimenez-Sanchez M, Bento CF, Puri C, Zavodszky E, Siddiqi F, Lavau CP et al (2014) PICALM modulates autophagy activity and tau accumulation. Nat Commun 5:4998. https://doi.org/10.1038/ncomms5998
Tian Y, Chang JC, Fan EY, Flajolet M, Greengard P (2013) Adaptor complex AP2/PICALM, through interaction with LC3, targets Alzheimer’s APP-CTF for terminal degradation via autophagy. Proc Natl Acad Sci USA 110:17071–17076. https://doi.org/10.1073/pnas.1315110110
Chen P, Wang Y, Chen L, Song N, Xie J (2020) Apelin-13 protects dopaminergic neurons against rotenone—induced neurotoxicity through the AMPK/mTOR/ULK-1 mediated autophagy activation. Int J Mol Sci 21:8376
Rocha EM, De Miranda B, Sanders LH (2018) Alpha-synuclein: pathology, mitochondrial dysfunction and neuroinflammation in Parkinson’s disease. Neurobiol Dis 109:249–257
Hewitt VL, Whitworth AJ (2017) Mechanisms of Parkinson’s disease: lessons from Drosophila. Curr Top Dev Biol 121:173–200
Dauer W, Przedborski S (2003) Parkinson’s disease: mechanisms and models. Neuron 39:889–909. https://doi.org/10.1016/s0896-6273(03)00568-3
Moloudizargari M, Asghari MH, Ghobadi E, Fallah M, Rasouli S, Abdollahi M (2017) Autophagy, its mechanisms and regulation: Implications in neurodegenerative diseases. Ageing Res Rev 40:64–74. https://doi.org/10.1016/j.arr.2017.09.005
Dehay B, Bové J, Rodríguez-Muela N, Perier C, Recasens A, Boya P, Vila M (2010) Pathogenic lysosomal depletion in Parkinson’s disease. J Neurosci 30:12535. https://doi.org/10.1523/jneurosci.1920-10.2010
Bellomo G, Paciotti S, Gatticchi L, Parnetti L (2020) The vicious cycle between α- synuclein aggregation and autophagic-lysosomal dysfunction. Mov Disord Off J Mov Disord Soc 35:34–44
Lin KJ, Lin KL, Chen SD, Liou CW, Chuang YC, Lin HY, Lin TK (2019) The overcrowded crossroads: mitochondria, alpha-synuclein, and the endo-lysosomal system interaction in Parkinson’s disease. Int J Mol Sci 20:5312
Kalia LV, Kalia SK, McLean PJ, Lozano AM, Lang AE (2013) α-Synuclein oligomers and clinical implications for Parkinson disease. Ann Neurol 73:155–169. https://doi.org/10.1002/ana.23746
Webb JL, Ravikumar B, Atkins J, Skepper JN, Rubinsztein DC (2003) Alpha-Synuclein is degraded by both autophagy and the proteasome. J Biol Chem 278:25009–25013. https://doi.org/10.1074/jbc.M300227200
Mak SK, McCormack AL, Manning-Boğ AB, Cuervo AM, Di Monte DA (2010) Lysosomal degradation of α-synuclein in vivo. J Biol Chem 285:13621–13629
Vogiatzi T, Xilouri M, Vekrellis K, Stefanis L (2008) Wild type α-synuclein is degraded by chaperone-mediated autophagy and macroautophagy in neuronal cells. J Biol Chem 283:23542–23556
Xilouri M, Brekk OR, Landeck N, Pitychoutis PM, Papasilekas T, Papadopoulou-Daifoti Z et al (2013) Boosting chaperone-mediated autophagy in vivo mitigates α-synuclein-induced neurodegeneration. Brain 136:2130–2146
Alvarez-Erviti L, Rodriguez-Oroz MC, Cooper JM, Caballero C, Ferrer I, Obeso JA, Schapira AH (2010) Chaperone-mediated autophagy markers in Parkinson disease brains. Arch Neurol 67:1464–1472
Sala G, Marinig D, Arosio A, Ferrarese C (2016) Role of chaperone-mediated autophagy dysfunctions in the pathogenesis of Parkinson’s disease. Front Mol Neurosci 9:157
Tang FL, Erion JR, Tian Y, Liu W, Yin DM, Ye J et al (2015) VPS35 in dopamine neurons is required for endosome-to-Golgi retrieval of Lamp2a, a receptor of chaperone-mediated autophagy that is critical for α-synuclein degradation and prevention of pathogenesis of Parkinson’s disease. J Neurosci 35:10613–10628
Murphy KE, Gysbers AM, Abbott SK, Tayebi N, Kim WS, Sidransky E et al (2014) Reduced glucocerebrosidase is associated with increased α-synuclein in sporadic Parkinson’s disease. Brain 137:834–848
Zhao L, Wang Z (2019) MicroRNAs: game changers in the regulation of α-Synuclein in Parkinson’s disease. Parkinson’s Disease 2019:1–10
Luan Y, Ren X, Zheng W, Zeng Z, Guo Y, Hou Z et al (2018) Chronic caffeine treatment protects against α-synucleinopathy by reestablishing autophagy activity in the mouse striatum. Frontiers in neuroscience 12:301
Parekh P, Sharma N, Gadepalli A, Shahane A, Sharma M, Khairnar A (2019) A cleaning crew: the pursuit of autophagy in Parkinson’s disease. ACS chemical neuroscience 10:3914–3926
Spencer B, Potkar R, Trejo M, Rockenstein E, Patrick C, Gindi R et al (2009) Beclin 1 gene transfer activates autophagy and ameliorates the neurodegenerative pathology in α-synuclein models of Parkinson’s and Lewy body diseases. J Neurosci 29:13578–13588
Yan JQ, Yuan YH, Chu SF, Li GH, Chen NH (2018) E46K mutant α-synuclein is degraded by both proteasome and macroautophagy pathway. Molecules 23:2839
Salvador N, Aguado C, Horst M, Knecht E (2000) Import of a cytosolic protein into lysosomes by chaperone-mediated autophagy depends on its folding state. J Biol Chem 275:27447–27456
Tanik SA, Schultheiss CE, Volpicelli-Daley LA, Brunden KR, Lee VM (2013) Lewy body-like α-synuclein aggregates resist degradation and impair macroautophagy. J Biol Chem 288:15194–15210
Ahmed I, Liang Y, Schools S, Dawson VL, Dawson TM, Savitt JM (2012) Development and characterization of a new Parkinson’s disease model resulting from impaired autophagy. J Neurosci 32:16503–16509
Friedman LG, Lachenmayer ML, Wang J, He L, Poulose SM, Komatsu M et al (2012) Disrupted autophagy leads to dopaminergic axon and dendrite degeneration and promotes presynaptic accumulation of α-synuclein and LRRK2 in the brain. J Neurosci 32:7585–7593
Oueslati A, Schneider BL, Aebischer P, Lashuel HA (2013) Polo-like kinase 2 regulates selective autophagic α-synuclein clearance and suppresses its toxicity in vivo. Proc Natl Acad Sci 110:E3945–E3954
Tenreiro S, Reimao-Pinto MM, Antas P, Rino J, Wawrzycka D, Macedo D et al (2014) Phosphorylation modulates clearance of alpha-synuclein inclusions in a yeast model of Parkinson’s disease. PLoS Genet 10:e1004302
Shahpasandzadeh H, Popova B, Kleinknecht A, Fraser PE, Outeiro TF, Braus GH (2014) Interplay between sumoylation and phosphorylation for protection against α-synuclein inclusions. J Biol Chem 289:31224–31240
Sevlever D, Jiang P, Yen S-HC (2008) Cathepsin D is the main lysosomal enzyme involved in the degradation of α- synuclein and generation of its carboxy-terminally truncated species. Biochemistry 47:9678–9687
Cullen V, Lindfors M, Ng J, Paetau A, Swinton E, Kolodziej P et al (2009) Cathepsin D expression level affects alpha-synuclein processing, aggregation, and toxicity in vivo. Molecular brain 2:1–17
Miura E, Hasegawa T, Konno M, Suzuki M, Sugeno N, Fujikake N et al (2014) VPS35 dysfunction impairs lysosomal degradation of α-synuclein and exacerbates neurotoxicity in a Drosophila model of Parkinson’s disease. Neurobiol Dis 71:1–13
Zimprich A, Benet-Pagès A, Struhal W, Graf E, Eck SH, Offman MN et al (2011) A mutation in VPS35, encoding a subunit of the retromer complex, causes late-onset Parkinson disease. Am J Hum Genet 89:168–175
Zavodszky E, Seaman MN, Moreau K, Jimenez-Sanchez M, Breusegem SY, Harbour ME, Rubinsztein DC (2014) Mutation in VPS35 associated with Parkinson’s disease impairs WASH complex association and inhibits autophagy. Nature Comm 5:1–16
Winslow AR, Chen C-W, Corrochano S, Acevedo-Arozena A, Gordon DE, Peden AA, Lichtenberg M, Menzies FM et al (2010) α-Synuclein impairs macroautophagy: implications for Parkinson’s disease. J Cell Biol 190:1023–1037. https://doi.org/10.1083/jcb.201003122
Vekrellis K, Xilouri M, Emmanouilidou E, Rideout HJ, Stefanis L (2011) Pathological roles of α-synuclein in neurological disorders. Lancet Neurol 10:1015–1025
Hou X, Watzlawik JO, Fiesel FC, Springer W (2020) Autophagy in Parkinson’s disease. J Mol Biol 432:2651–2672
Plowey ED, Cherra SJ III, Liu YJ, Chu CT (2008) Role of autophagy in G2019S-LRRK2-associated neurite shortening in differentiated SH-SY5Y cells. J Neurochem 105:1048–1056
Li JQ, Tan L, Yu JT (2014) The role of the LRRK2 gene in Parkinsonism. Mol Neurodegener 9:1–17
Xilouri M, Vogiatzi T, Vekrellis K, Park D, Stefanis L (2009) Abberant α-Synuclein confers toxicity to neurons in part through inhibition of chaperone-mediated autophagy. PLoS ONE 4:e5515. https://doi.org/10.1371/journal.pone.0005515
Decressac M, Mattsson B, Weikop P, Lundblad M, Jakobsson J, Björklund A (2013) TFEB-mediated autophagy rescues midbrain dopamine neurons from α-synuclein toxicity. Proc Natl Acad Sci U S A 110:E1817–E1826. https://doi.org/10.1073/pnas.1305623110
Karabiyik C, Lee MJ, Rubinsztein DC (2017) Autophagy impairment in Parkinson’s disease. Essays Biochem 61:711–720. https://doi.org/10.1042/EBC20170023
Duda JE, Giasson BI, Mabon ME, Lee VM, Trojanowski JQ (2002) Novel antibodies to synuclein show abundant striatal pathology in Lewy body diseases. Ann Neurol 52:205–210. https://doi.org/10.1002/ana.10279\
Narendra D, Tanaka A, Suen DF, Youle RJ (2008) Parkin is recruited selectively to impaired mitochondria and promotes their autophagy. J Cell Biol 183:795–803. https://doi.org/10.1083/jcb.200809125
Limanaqi F, Biagioni F, Busceti CL, Ryskalin L, Polzella M, Frati A, Fornai F (2019) Phytochemicals bridging autophagy induction and Alpha-Synuclein degradation in Parkinsonism. Int J Mol Sci 20:3274
Li G, Luo W, Wang B, Qian C, Ye Y, Li Y, Zhang S (2021) HMGA1 Induction of miR-103/107 forms a negative feedback loop to regulate autophagy in MPTP model of Parkinson’s disease. Front Cell Neurosci 14:464
Gil JM, Rego AC (2008) Mechanisms of neurodegeneration in Huntington’s disease. Eur J Neurosci 27:2803–2820. https://doi.org/10.1111/j.1460-9568.2008.06310.x
Martin JB, Gusella JF (1986) Huntington’s disease. Pathogenesis and management. N Engl J Med 315:1267–1276. https://doi.org/10.1056/NEJM198611133152006
Imarisio S, Carmichael J, Korolchuk V, Chen C-W, Saiki S, Rose C, Krishna G, Davies Janet E, Ttofi E, Underwood Benjamin R, Rubinsztein David C (2008) Huntington’s disease: from pathology and genetics to potential therapies. Biochem J 412:191. https://doi.org/10.1042/BJ20071619
Jimenez-Sanchez M, Licitra F, Underwood BR, Rubinsztein DC (2017) Huntington’s disease: mechanisms of pathogenesis and therapeutic strategies. Cold Spring Harb Perspect Med 7:a024240. https://doi.org/10.1101/cshperspect.a024240
Dayalu P, Albin RL (2015) Huntington disease: pathogenesis and treatment. Neurol Clin 33:101–114. https://doi.org/10.1016/j.ncl.2014.09.003
Tellez-Nagel I, Johnson AB, Terry RD (1974) Studies on brain biopsies of patients with Huntington’s chorea. J Neuropathol Exp Neurol 33:308–332. https://doi.org/10.1097/00005072-197404000-00008
Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, Scaravilli F, Easton DF, Duden R, O’Kane CJ, Rubinsztein DC (2004) Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 36:585–595. https://doi.org/10.1038/ng1362
Martinez-Vicente M, Talloczy Z, Wong E, Tang GM, Koga H, Kaushik S, de Vries R, Arias E, Harris S, Sulzer D, Cuervo AM (2010) Cargo recognition failure is responsible for inefficient autophagy in Huntington’s disease. Nat Neurosci 13:567-U574. https://doi.org/10.1038/nn.2528
Zheng S, Clabough EBD, Sarkar S, Futter M, Rubinsztein DC, Zeitlin SO (2010) Deletion of the Huntingtin polyglutamine stretch enhances neuronal autophagy and longevity in mice. PLoS Genet 6:e1000838. https://doi.org/10.1371/journal.pgen.1000838
Atwal RS, Truant R (2008) A stress sensitive ER membrane-association domain in Huntingtin protein defines a potential role for Huntingtin in the regulation of autophagy. Autophagy 4:91–93. https://doi.org/10.4161/auto.5201
Rui Y-N, Xu Z, Patel B, Chen Z, Chen D, Tito A, David G, Sun Y, Stimming EF, Bellen HJ, Cuervo AM, Zhang S (2015) Huntingtin functions as a scaffold for selective macroautophagy. Nat Cell Biol 17:262–275. https://doi.org/10.1038/ncb3101
Walter C, Clemens LE, Muller AJ, Fallier-Becker P, Proikas-Cezanne T, Riess O, Metzger S, Nguyen HP (2016) Activation of AMPK-induced autophagy ameliorates Huntington disease pathology in vitro. Neuropharmacology 108:24–38. https://doi.org/10.1016/j.neuropharm.2016.04.041
Squitieri F, Gellera C, Cannella M, Mariotti C, Cislaghi G, Rubinsztein DC, Almqvist EW, Turner D et al (2003) Homozygosity for CAG mutation in Huntington disease is associated with a more severe clinical course. Brain 126:946–955. https://doi.org/10.1093/brain/awg077
Ashkenazi A, Bento CF, Ricketts T, Vicinanza M, Siddiqi F, Pavel M, Squitieri F, Hardenberg MC, Imarisio S, Menzies FM, Rubinsztein DC (2017) Polyglutamine tracts regulate beclin 1-dependent autophagy. Nature 545:108–111. https://doi.org/10.1038/nature22078
Sarkar S, Rubinsztein DC (2008) Huntington’s disease: degradation of mutant Huntingtin by autophagy. FEBS J 275:4263–4270. https://doi.org/10.1111/j.1742-4658.2008.06562.x
Wong YC, Holzbaur EL (2014) The regulation of autophagosome dynamics by Huntingtin and HAP1 is disrupted by expression of mutant huntingtin, leading to defective cargo degradation. J Neurosci 34:1293–1305. https://doi.org/10.1523/jneurosci.1870-13.2014
Martin DD, Ladha S, Ehrnhoefer DE, Hayden MR (2015) Autophagy in Huntington disease and huntingtin in autophagy. Trends Neurosci 38:26–35. https://doi.org/10.1016/j.tins.2014.09.003
Lee H, Noh JY, Oh Y, Kim Y, Chang JW, Chung CW, Lee ST, Kim M, Ryu H, Jung YK (2012) IRE1 plays an essential role in ER stress-mediated aggregation of mutant huntingtin via the inhibition of autophagy flux. Hum Mol Genet 21:101–114. https://doi.org/10.1093/hmg/ddr445
Ashkenazi A, Bento CF, Ricketts T, Vicinanza M, Siddiqi F, Pavel M, Squitieri F, Hardenberg MC, Imarisio S, Menzies FM, Rubinsztein DC (2017) Polyglutamine tracts regulate autophagy. Autophagy 13:1613–1614. https://doi.org/10.1080/15548627.2017.1336278
Matsumoto G, Wada K, Okuno M, Kurosawa M, Nukina N (2011) Serine 403 phosphorylation of p62/SQSTM1 regulates selective autophagic clearance of ubiquitinated proteins. Mol Cell 44:279–289. https://doi.org/10.1016/j.molcel.2011.07.039
Al-Ramahi I, Giridharan S, Chen YC, Patnaik S, Safren N, Hasegawa J, de Haro M et al (2017) Inhibition of PIP4Kgamma ameliorates the pathological effects of mutant huntingtin protein. Elife 6:e29123. https://doi.org/10.7554/eLife.29123
Croce KR, Yamamoto A (2018) A role for autophagy in Huntington’s disease. Neurobiol Dis 122:16–22. https://doi.org/10.1016/j.nbd.2018.08.010
Sarkar S, Davies JE, Huang Z, Tunnacliffe A, Rubinsztein DC (2007) Trehalose, a novel mTOR-independent autophagy enhancer, accelerates the clearance of mutant Huntingtin and α-Synuclein. J Biol Chem 282:5641–5652. https://doi.org/10.1074/jbc.M609532200
Sarkar S, Floto RA, Berger Z, Imarisio S, Cordenier A, Pasco M, Cook LJ, Rubinsztein DC (2005) Lithium induces autophagy by inhibiting inositol monophosphatase. J Cell Biol 170:1101–1111. https://doi.org/10.1083/jcb.200504035
Jeong H, Then F, Melia TJ, Mazzulli JR, Cui L, Savas JN, Voisine C, Paganetti P, Tanese N, Hart AC, Yamamoto A, Krainc D (2009) Acetylation targets mutant Huntingtin to autophagosomes for degradation. Cell 137:60–72. https://doi.org/10.1016/j.cell.2009.03.018
Qi L, Zhang XD, Wu JC, Lin F, Wang J, DiFiglia M, Qin ZH (2012) The role of chaperone-mediated autophagy in huntingtin degradation. PLoS One 7:e46834. https://doi.org/10.1371/journal.pone.0046834
Hardiman O, van den Berg LH, Kiernan MC (2011) Clinical diagnosis and management of amyotrophic lateral sclerosis. Nat Rev Neurol 7:639–649. https://doi.org/10.1038/nrneurol.2011.153
Andersen PM, Al-Chalabi A (2011) Clinical genetics of amyotrophic lateral sclerosis: what do we really know? Nat Rev Neurol 7:603. https://doi.org/10.1038/nrneurol.2011.150
Blokhuis AM, Groen EJN, Koppers M, van den Berg LH, Pasterkamp RJ (2013) Protein aggregation in amyotrophic lateral sclerosis. Acta Neuropathol 125:777–794. https://doi.org/10.1007/s00401-013-1125-6
Boillee S, Vande Velde C, Cleveland DW (2006) ALS: A disease of motor neurons and their nonneuronal neighbors. Neuron 52:39–59. https://doi.org/10.1016/j.neuron.2006.09.018
Sasaki S (2011) Autophagy in spinal cord motor neurons in sporadic amyotrophic lateral sclerosis. J Neuropathol Exp Neurol 70:349–359. https://doi.org/10.1097/NEN.0b013e3182160690
Li L, Zhang X, Le W (2008) Altered macroautophagy in the spinal cord of SOD1 mutant mice. Autophagy 4:290–293. https://doi.org/10.4161/auto.5524
Morimoto N, Nagai M, Ohta Y, Miyazaki K, Kurata T, Morimoto M, Murakami T, Takehisa Y, Ikeda Y, Kamiya T, Abe K (2007) Increased autophagy in transgenic mice with a G93A mutant SOD1 gene. Brain Res 1167:112–117. https://doi.org/10.1016/j.brainres.2007.06.045
Sheng YW, Chattopadhyay M, Whitelegge J, Valentine JS (2012) SOD1 Aggregation and ALS: role of metallation states and disulfide status. Curr Top Med Chem 12:2560–2572. https://doi.org/10.2174/15680266112129990079
An T, Shi P, Duan W, Zhang S, Yuan P, Li Z, Wu D, Xu Z, Li C, Guo Y (2014) Oxidative stress and autophagic alteration in brainstem of SOD1-G93A mouse model of ALS. Mol Neurobiol 49:1435–1448. https://doi.org/10.1007/s12035-013-8623-3
Rudnick ND, Griffey CJ, Guarnieri P, Gerbino V, Wang X, Piersaint JA, Tapia JC, Rich MM, Maniatis T (2017) Distinct roles for motor neuron autophagy early and late in the SOD1G93A mouse model of ALS. Proc Natl Acad Sci U S A 114:E8294–E8303. https://doi.org/10.1073/pnas.1704294114
Hetz C, Thielen P, Matus S, Nassif M, Court F, Kiffin R, Martinez G, Cuervo AM, Brown RH, Glimcher LH (2009) XBP-1 deficiency in the nervous system protects against amyotrophic lateral sclerosis by increasing autophagy. Genes Dev 23:2294–2306. https://doi.org/10.1101/gad.1830709
Crippa V, Sau D, Rusmini P, Boncoraglio A, Onesto E, Bolzoni E, Galbiati M, Fontana E, Marino M, Carra S, Bendotti C, De Biasi S, Poletti A (2010) The small heat shock protein B8 (HspB8) promotes autophagic removal of misfolded proteins involved in amyotrophic lateral sclerosis (ALS). Hum Mol Genet 19:3440–3456. https://doi.org/10.1093/hmg/ddq257
Gal J, Ström A-L, Kwinter DM, Kilty R, Zhang J, Shi P, Fu W, Wooten MW, Zhu H (2009) Sequestosome 1/p62 links familial ALS mutant SOD1 to LC3 via an ubiquitin-independent mechanism. J Neurochem 111:1062–1073. https://doi.org/10.1111/j.1471-4159.2009.06388.x
Goode A, Butler K, Long J, Cavey J, Scott D, Shaw B, Sollenberger J, Gell C, Johansen T, Oldham NJ, Searle MS, Layfield R (2016) Defective recognition of LC3B by mutant SQSTM1/p62 implicates impairment of autophagy as a pathogenic mechanism in ALS-FTLD. Autophagy 12:1094–1104. https://doi.org/10.1080/15548627.2016.1170257
Brady OA, Meng P, Zheng Y, Mao Y, Hu F (2011) Regulation of TDP-43 aggregation by phosphorylation andp62/SQSTM1. J Neurochem 116:248–259. https://doi.org/10.1111/j.1471-4159.2010.07098.x
Li F, Xie X, Wang Y, Liu J, Cheng X, Guo Y, Gong Y, Hu S, Pan L (2016) Structural insights into the interaction and disease mechanism of neurodegenerative disease-associated optineurin and TBK1 proteins. Nat Commun 7:12708. https://doi.org/10.1038/ncomms12708
Pilli M, Arko-Mensah J, Ponpuak M, Roberts E, Master S, Mandell MA, Dupont N et al (2012) TBK-1 promotes autophagy-mediated antimicrobial defense by controlling autophagosome maturation. Immunity 37:223–234. https://doi.org/10.1016/j.immuni.2012.04.015
Moore AS, Erika LFH (2016) Dynamic recruitment and activation of ALS-associated TBK1 with its target optineurin are required for efficient mitophagy. Proc Natl Acad Sci U S A 113:E3349. https://doi.org/10.1073/pnas.1523810113
Sundaramoorthy V, Walker AK, Tan V, Fifita JA, Mccann EP, Williams KL, Blair IP, Guillemin GJ, Farg MA, Atkin JD (2015) Defects in optineurin- and myosin VI-mediated cellular trafficking in amyotrophic lateral sclerosis. Hum Mol Genet 24:3830–3846. https://doi.org/10.1093/hmg/ddv126
Tumbarello DA, Waxse BJ, Arden SD, Bright NA, Kendrick-Jones J, Buss F (2012) Autophagy receptors link myosin VI to autophagosomes to mediate Tom1-dependent autophagosome maturation and fusion with the lysosome. Nat Cell Biol 14:1024–1035. https://doi.org/10.1038/ncb2589
Wong YC, Erika LFH (2014) Optineurin is an autophagy receptor for damaged mitochondria in parkin-mediated mitophagy that is disrupted by an ALS-linked mutation. Proc Natl Acad Sci U S A 111:E4439. https://doi.org/10.1073/pnas.1405752111
Wong YC, Holzbaur ELF (2015) Temporal dynamics of PARK2/parkin and OPTN/optineurin recruitment during the mitophagy of damaged mitochondria. Autophagy 11:422–424. https://doi.org/10.1080/15548627.2015.1009792
Jantrapirom S, Lo PL, Yoshida H, Yamaguchi M (2018) Depletion of ubiquilin induces an augmentation in soluble ubiquitinated Drosophila TDP-43 to drive neurotoxicity in the fly. Biochim Biophys Acta Mol Basis Dis 1864:3038–3049. https://doi.org/10.1016/j.bbadis.2018.06.017
Deng HX, Chen W, Hong ST, Boycott KM, Gorrie GH, Siddique N, Yang Y et al (2011) Mutations in UBQLN2 cause dominant X-linked juvenile and adult-onset ALS and ALS/dementia. Nature 477:211–215. https://doi.org/10.1038/nature10353
Le NT, Chang L, Kovlyagina I, Georgiou P, Safren N, Braunstein KE, Kvarta MD, Van Dyke AM, LeGates TA et al (2016) Motor neuron disease, TDP-43 pathology, and memory deficits in mice expressing ALS-FTD-linked UBQLN2 mutations. Proc Natl Acad Sci U S A 113:E7580–E7589. https://doi.org/10.1073/pnas.1608432113
Chang L, Monteiro MJ (2015) Defective proteasome delivery of polyubiquitinated proteins by ubiquilin-2 proteins containing ALS mutations. PLoS One 10:e0130162. https://doi.org/10.1371/journal.pone.0130162
Osaka M, Ito D, Suzuki N (2016) Disturbance of proteasomal and autophagic protein degradation pathways by amyotrophic lateral sclerosis-linked mutations in ubiquilin 2. Biochem Biophys Res Commun 472:324–331. https://doi.org/10.1016/j.bbrc.2016.02.107
Todd TW, Petrucelli L (2016) Insights into the pathogenic mechanisms of chromosome 9 open reading frame 72 (C9orf72) repeat expansions. J Neurochem 138:145–162. https://doi.org/10.1111/jnc.13623
Webster CP, Smith EF, Bauer CS, Moller A, Hautbergue GM, Ferraiuolo L, Myszczynska MA, Higginbottom A et al (2016) The C9orf72 protein interacts with Rab1a and the ULK1 complex to regulate initiation of autophagy. EMBO J 35:1656–1676. https://doi.org/10.15252/embj.201694401
Sellier C, Campanari ML, Julie Corbier C, Gaucherot A, Kolb Cheynel I, Oulad Abdelghani M, Ruffenach F, Page A et al (2016) Loss of C9ORF72 impairs autophagy and synergizes with polyQ Ataxin-2 to induce motor neuron dysfunction and cell death. EMBO J 35:1276–1297. https://doi.org/10.15252/embj.201593350
Ji YJ, Ugolino J, Brady NR, Hamacher-Brady A, Wang J (2017) Systemic deregulation of autophagy upon loss of ALS- and FTD-linked C9orf72. Autophagy 13:1254–1255. https://doi.org/10.1080/15548627.2017.1299312
Chang MC, Srinivasan K, Friedman BA, Suto E, Modrusan Z, Lee WP, Kaminker JS, Hansen DV, Sheng M (2017) Progranulin deficiency causes impairment of autophagy and TDP-43 accumulation. J Exp Med 214:2611–2628. https://doi.org/10.1084/jem.20160999
Ser A, Vollrath JT, Sechi A, Johann S, Roos A, Yamoah A, Katona I, Bohlega S, Wiemuth D, Tian Y et al (2017) The ALS-linked E102Q mutation in Sigma receptor-1 leads to ER stress-mediated defects in protein homeostasis and dysregulation of RNA-binding proteins. Cell Death Differ 24:1655–1671. https://doi.org/10.1038/cdd.2017.88
Johnson JO, Mandrioli J, Benatar M, Abramzon Y, Van Deerlin VM, Trojanowski JQ, Gibbs JR et al (2010) Exome sequencing reveals VCP mutations as a cause of familial ALS. Neuron 68:857–864. https://doi.org/10.1016/j.neuron.2010.11.036
Watts GDJ, Wymer J, Kovach MJ, Mehta SG, Mumm S, Darvish D, Pestronk A, Whyte MP, Kimonis VE (2004) Inclusion body myopathy associated with Paget disease of bone and frontotemporal dementia is caused by mutant valosin-containing protein. Nat Genet 36:377–381. https://doi.org/10.1038/ng1332
Johnson AE, Shu H, Hauswirth AG, Tong A, Davis GW (2015) VCP-dependent muscle degeneration is linked to defects in a dynamic tubular lysosomal network in vivo. Elife 4:e07366. https://doi.org/10.7554/eLife.07366
Kustermann M, Manta L, Paone C, Kustermann J, Lausser L, Wiesner C, Eichinger L et al (2018) Loss of the novel Vcp (valosin containing protein) interactor Washc4 interferes with autophagy-mediated proteostasis in striated muscle and leads to myopathy in vivo. Autophagy 14:1911–1927. https://doi.org/10.1080/15548627.2018.1491491
Ritson GP, Custer SK, Freibaum BD, Guinto JB, Geffel D, Moore J et al (2010) TDP-43 mediates degeneration in a novel Drosophila model of disease caused by mutations in VCP/p97. J Neurosci 30:7729–7739. https://doi.org/10.1523/jneurosci.5894-09.2010
Caccamo A, Maldonado MA, Majumder S, Medina DX, Holbein W, Magrí A, Oddo S (2011) Naturally secreted amyloid-beta increases mammalian target of rapamycin (mTOR) activity via a PRAS40-mediated mechanism. J Biol Chem 286:8924–8932. https://doi.org/10.1074/jbc.M110.180638
Tanji K, Miki Y, Ozaki T, Maruyama A, Yoshida H, Mimura J, Wakabayashi K (2014) Phosphorylation of serine 349 of p62 in Alzheimer’s disease brain. Acta Neuropathol Commun 2:50. https://doi.org/10.1186/2051-5960-2-50
Isakson P, Holland P, Simonsen A (2013) The role of ALFY in selective autophagy. Cell Death Differ 20:12–20. https://doi.org/10.1038/cdd.2012.66
Lim J, Lachenmayer ML, Wu S, Liu W, Kundu M, Wang R, Yue Z (2015) Proteotoxic stress induces phosphorylation of p62/SQSTM1 by ULK1 to regulate selective autophagic clearance of protein aggregates. PLoS Genetics 11:e1004987. https://doi.org/10.1371/journal.pgen.1004987
Xilour M, Vogiatzi T, Stefanis L (2008) Alpha-synuclein degradation by autophagic pathways: a potential key to Parkinson’s disease pathogenesis. Autophagy 4:917–919. https://doi.org/10.4161/auto.6685
Ramirez A, Heimbach A, Grundemann J, Stiller B, Hampshire D, Cid LP, Kubisch C (2006) Hereditary parkinsonism with dementia is caused by mutations in ATP13A2, encoding a lysosomal type 5 P-type ATPase. Nat. Genet 38:1184–1191. https://doi.org/10.1038/ng1884
Di Fonzo A, Chien HF, Socal M, Giraudo S, Tassorelli C, Iliceto G, Bonifati V (2007) ATP13A2 missense mutations in juvenile parkinsonism and young onset Parkinson disease. Neurology 68:1557–1562. https://doi.org/10.1212/01.wnl.0000260963.08711.08
Nixon RA, Yang DS (2012) Autophagy and neuronal cell death in neurological disorders. Cold Spring Harb Perspect Biol 4(10):a008839. https://doi.org/10.1101/cshperspect.a008839
Gasser T (2009) Molecular pathogenesis of Parkinson disease: insights from genetic studies. Expert Rev Mol Med 11:e22. https://doi.org/10.1017/S1462399409001148
Kim YC, Guan KL (2015) mTOR: a pharmacologic target for autophagy regulation. J Clin Invest 125:25–32
Du J, Liang Y, Xu F, Sun B, Wang Z (2013) Trehalose rescues Alzheimer’s disease phenotypes in APP/PS1 transgenic mice. J Pharm Pharmacol 65:1753–1756
Son SM, Shin H-J, Byun J, Kook SY, Moon M, Chang YJ et al (2016) Metformin facilitates amyloid-β generation by β- and γ-secretases via autophagy activation. J Alzheimers Dis JAD 51:1197–1208
Castillo K, Nassif M, Valenzuela V, Rojas F, Matus S, Mercado G et al (2013) Trehalose delays the progression of amyotrophic lateral sclerosis by enhancing autophagy in motoneurons. Autophagy 9:1308–1320
Li Y, Guo Y, Wang X, Yu X, Duan W, Hong K et al (2015) Trehalose decreases mutant SOD1 expression and alleviates motor deficiency in early but not end-stage amyotrophic lateral sclerosis in a SOD1-G93A mouse model. Neuroscience 298:12–25
Zhang X, Chen S, Song L, Tang Y, Shen Y, Jia L et al (2014) MTOR-independent, autophagic enhancer trehalose prolongs motor neuron survival and ameliorates the autophagic flux defect in a mouse model of amyotrophic lateral sclerosis. Autophagy 10:588–602
Ma TC, Buescher JL, Oatis B, Funk JA, Nash AJ, Carrier RL et al (2007) Metformin therapy in a transgenic mouse model of Huntington’s disease. Neurosci Lett 411:98–103
Suresh SN, Chavalmane AK, Pillai M, Ammanathan V, Vidyadhara DJ, Yarreiphang H et al (2018) Modulation of autophagy by a small molecule inverse agonist of ERRα is neuroprotective. Front Mol Neurosci 11:109
Rose C, Menzies FM, Renna M, Acevedo-Arozena A, Corrochano S, Sadiq O et al (2010) Rilmenidine attenuates toxicity of polyglutamine expansions in a mouse model of Huntington’s disease. Hum Mol Genet 19:2144–2153
Williams A, Sarkar S, Cuddon P, Ttofi EK, Saiki S, Siddiqi FH et al (2008) Novel targets for Huntington’s disease in an mTOR-independent Autophagy pathway. Nat Chem Biol 4:295–305
Hirano K, Fujimaki M, Sasazawa Y, Yamaguchi A, Ishikawa K-I, Miyamoto K et al (2019) Neuroprotective effects of memantine via enhancement of autophagy. Biochem Biophys Res Commun 518:161–170
Chu C, Zhang X, Ma W, Li L, Wang W, Shang L, Fu P (2013) Induction of autophagy by a novel small molecule improves abeta pathology and ameliorates cognitive deficits. PLoS One 8:e65367. https://doi.org/10.1371/journal.pone.0065367
Guo X, Lv J, Lu J, Fan L, Huang X, Hu L, Wang J, Shen X (2018) Protopanaxadiol derivative DDPU improves behavior and cognitive deficit in AD mice involving regulation of both ER stress and autophagy. Neuropharmacology 130:77–91. https://doi.org/10.1016/j.neuropharm.2017.11.033
Huang M, Jiang X, Liang Y, Liu Q, Chen S, Guo Y (2017) Berberine improves cognitive impairment by promoting autophagic clearance and inhibiting production of beta-amyloid in APP/tau/PS1 mouse model of Alzheimer’s disease. Exp Gerontol 91:25–33. https://doi.org/10.1016/j.exger.2017.02.004
Li LS, Lu YL, Nie J, Xu YY, Zhang W, Yang WJ, Gong QH, Lu YF, Lu Y, Shi JS (2017) Dendrobium nobile Lindl alkaloid, a novel autophagy inducer, protects against axonal degeneration induced by Abeta25-35 in hippocampus neurons in vitro. CNS Neurosci Ther 23:329–340. https://doi.org/10.1111/cns.12678
Jiang T-F, Zhang Y-J, Zhou H-Y, Wang H-M, Tian L-P, Liu J, Ding J-Q, Chen S-D (2013) Curcumin ameliorates the neurodegenerative pathology in A53T α-synuclein cell model of Parkinson’s disease through the downregulation of mTOR/p70S6K signaling and the recovery of macroautophagy. J Neuroimmune Pharmacol 8:356–369. https://doi.org/10.1007/s11481-012-9431-7
Wu Y, Li X, Zhu JX, Xie W, Le W, Fan Z, Jankovic J, Pan T (2011) Resveratrol-activated AMPK/SIRT1/autophagy in cellular models of Parkinson’s disease. Neurosignals 19:163–174. https://doi.org/10.1159/000328516
Lan D-M, Liu F-T, Zhao J, Chen Y, Wu J-J, Ding Z-T, Yue Z-Y, Ren H-M, Jiang Y-P, Wang J (2012) Effect of trehalose on PC12 cells overexpressing wild-type or A53T mutant α-synuclein. Neurochem Res 37:2025–2032. https://doi.org/10.1007/s11064-012-0823-0
Hebron ML, Lonskaya I, Moussa CEH (2013) Nilotinib reverses loss of dopamine neurons and improves motor behavior via autophagic degradation of α-synuclein in Parkinson’s disease models. Hum Mol Genet 22:3315–3328. https://doi.org/10.1093/hmg/ddt192
Liu J, Chen M, Wang X, Wang Y, Duan C, Gao G, Lu L, Wu X, Wang X, Yang H (2016) Piperine induces autophagy by enhancing protein phosphotase 2A activity in a rotenone-induced Parkinson’s disease model. Oncotarget 7:60823–60843. https://doi.org/10.18632/oncotarget.11661
Hou YS, Guan JJ, Xu HD, Wu F, Sheng R, Qin ZH (2015) Sestrin2 protects dopaminergic cells against rotenone toxicity through AMPK-dependent autophagy activation. Mol Cell Biol 35:2740–2751. https://doi.org/10.1128/MCB.00285-15
Yang G, Li J, Cai Y, Yang Z, Li R, Fu W (2018) Glycyrrhizic acid alleviates 6-hydroxydopamine and corticosterone-induced neurotoxicity in SH-SY5Y cells through modulating autophagy. Neurochem Res 43:1914–1926. https://doi.org/10.1007/s11064-018-2609-5
Jang W, Ju Kim H, Li H, Jo K, Kyu Lee M, Hong Song S, Ok Yang H (2014) 1,25-Dyhydroxyvitamin D-3 attenuates rotenone-induced neurotoxicity in SH-SY5Y cells through induction of autophagy. Biochem Biophys Res Commun 451:142–147. https://doi.org/10.1016/j.bbrc.2014.07.081
Jia H, Kast RJ, Steffan JS, Thomas EA (2012) Selective histone deacetylase (HDAC) inhibition imparts beneficial effects in Huntington’s disease mice: implications for the ubiquitin–proteasomal and autophagy systems. Hum Mol Genet 21:5280–5293. https://doi.org/10.1093/hmg/dds379
Abd-Elrahman KS, Hamilton A, Hutchinson SR, Liu F, Russell RC, Ferguson SSG (2017) mGluR5 antagonism increases autophagy and prevents disease progression in the zQ175 mouse model of Huntington’s disease. Sci Signal 10:eaan6387. https://doi.org/10.1126/scisignal.aan6387
Chang C-C, Lin T-C, Ho H-L, Kuo C-Y, Li H-H, Korolenko TA, Chen W-J, Lai T-J, Ho Y-J, Lin C-L (2018) GLP-1 analogue liraglutide attenuates mutant Huntingtin-induced neurotoxicity by restoration of neuronal insulin signaling. Int J Mol Sci 19:2505. https://doi.org/10.3390/ijms19092505
Wong VKW, Wu AG, Wang JR, Liu L, Law BY-K (2015) Neferine attenuates the protein level and toxicity of mutant huntingtin in PC-12 cells via induction of autophagy. Molecules 20:3496–3514. https://doi.org/10.3390/molecules20033496
Wang IF, Guo BS, Liu YC, Wu CC, Yang CH, Tsai KJ, Shen CKJ (2012) Autophagy activators rescue and alleviate pathogenesis of a mouse model with proteinopathies of the TAR DNA-binding protein 43. Proc Natl Acad Sci U S A 109:15024–15029. https://doi.org/10.1073/pnas.1206362109
Wang Y, Chen S, Wang Z, Zhang X, Yang D, Zhang X, Li L, Le W (2011) Rapamycin treatment augments motor neuron degeneration in SOD1G93A mouse model of amyotrophic lateral sclerosis. Autophagy 7:412–425
Feng HL, Leng Y, Ma CH, Zhang J, Ren M, Chuang DM (2008) Combined lithium and valproate treatment delays disease onset, reduces neurological deficits and prolongs survival in an amyotrophic lateral sclerosis mouse model. Neuroscience 155:567–572. https://doi.org/10.1016/j.neuroscience.2008.06.040
Fornai F, Longone P, Cafaro L, Kastsiuchenka O, Ferrucci M, Manca ML, Lazzeri G et al (2008) Lithium delays progression of amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A 105:2052–2057. https://doi.org/10.1073/pnas.0708022105
Chang C-F, Lee Y-C, Lee K-H, Lin H-C, Chen C-L, Shen C-KJ, Huang C-C (2016) Therapeutic effect of berberine on TDP-43-related pathogenesis in FTLD and ALS. J Biomed Sci 23:72–72. https://doi.org/10.1186/s12929-016-0290-z
Sun J, Mu Y, Jiang Y, Song R, Yi J, Zhou J, Sun J, Jiao X, Prinz RA, Li Y, Xu X (2018) Inhibition of p70 S6 kinase activity by A77 1726 induces autophagy and enhances the degradation of superoxide dismutase 1 (SOD1) protein aggregates. Cell Death Dis 9:407–407. https://doi.org/10.1038/s41419-018-0441-0
Zhou Q-M, Zhang J-J, Li S, Chen S, Le W-D (2017) n-butylidenephthalide treatment prolongs life span and attenuates motor neuron loss in SOD1G93A mouse model of amyotrophic lateral sclerosis. CNS Neurosci Ther 23:375–385. https://doi.org/10.1111/cns.12681
Acknowledgements
The authors would like to thank Chitkara University, Punjab, India for providing the basic facilities for the completion of this article.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All the authors have given consent for final publication of the current manuscript.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Autophagy is an evolutionary conserved cellular process and is crucial for maintaining cellular homeostasis.
• Autophagy performs an essential role in the pathophysiology of several neurodegenerative disorders.
• Autophagy is regulated by important nutrient-sensing pathways including the mTORC1 and AMPK.
• Autophagy is a potential therapeutic target to ameliorate neurodegenerative disorders.
Rights and permissions
About this article
Cite this article
Rana, T., Behl, T., Sehgal, A. et al. Exploring the Role of Autophagy Dysfunction in Neurodegenerative Disorders. Mol Neurobiol 58, 4886–4905 (2021). https://doi.org/10.1007/s12035-021-02472-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02472-0